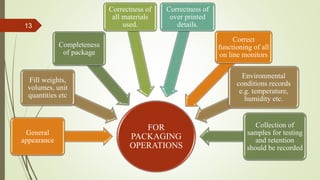
The document outlines essential practices and requirements for manufacturing operations and quality assurance in the pharmaceutical industry, emphasizing the importance of good manufacturing practices to ensure product identity, strength, safety, and purity. It covers various aspects including sanitation, contamination control, intermediate and bulk product processing, packaging operations, in-process quality checks, finished product release, and compliance with regulatory standards. The document stresses that all manufacturing processes must be conducted under the supervision of qualified personnel and that rigorous documentation and inspection processes must be in place to maintain quality.