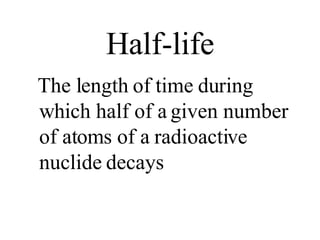Nuclear forces keep nucleons together through powerful short-range attraction between protons and neutrons. The mass of a nucleus is less than the total mass of its constituent particles due to the nuclear mass defect. This mass defect is converted to nuclear binding energy, which is released when the nucleus forms. Nuclei with intermediate atomic weights have the greatest stability and binding energy per particle, while the lightest and heaviest nuclei are less stable.