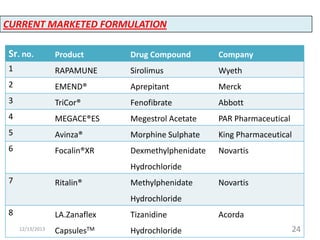
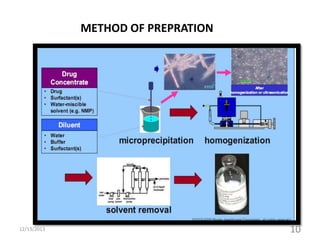
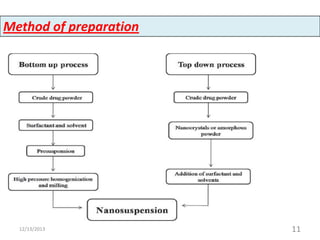
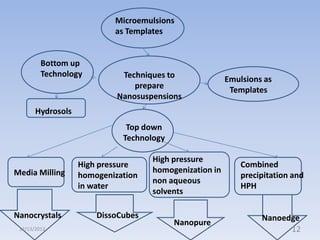
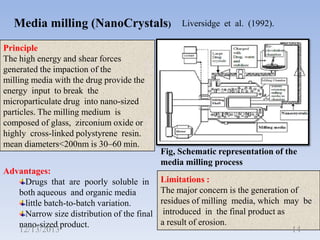
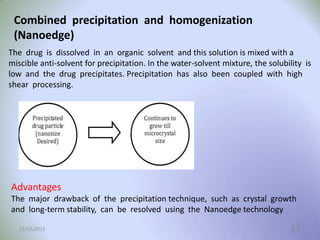
This document provides an introduction and overview of nanosuspensions for pharmaceutical applications. It was prepared by Mayuri B. Yadav and guided by Prof. Dr. S. N. Dhole of Modern College of Pharmacy in Pune, India. The document defines nanosuspensions and discusses their advantages such as improved bioavailability. It also outlines various preparation methods including media milling, high pressure homogenization, and the use of emulsions or microemulsions as templates. Evaluation parameters for nanosuspensions and some currently marketed formulations are also mentioned. The applications and conclusion state that nanosuspensions can effectively deliver poorly soluble drugs.














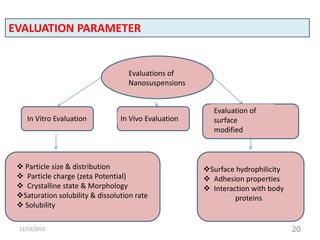
![In vitro Evaluations
Mean particle size and size distribution:
The mean particle size and the width of particle size distribution called
Polydidpersity Index are determined by Photon Correlation Spectroscopy (PCS).
Laser Diffractometry (LD) particles ranging from 0.05- 80μm upto 2000µm.
Atomic Force microscop is used for visualization of particle shape.
Particle charge (Zeta Potential):
Particle charge determines the stability of nanosuspension. For
electrostatically stabilized nanosuspension a minimum zeta potential of ±30mV
Crystalline state and particle morphology:
Differential Scanning colorimeter (DSC) determines the crystalline
structure.. The X-Ray Diffraction[30] (XRD) is also used for determining change
in physical state and extent of amorphous drug. Scanning electron microscopy
is also used to get exact information about particle morphology.
12/13/2013
21](https://image.slidesharecdn.com/newmicrosoftofficepowerpointpresentation-131213033403-phpapp01/85/New-microsoft-office-power-point-presentation-21-320.jpg)