This document discusses minerals in the human body. It classifies minerals as either macronutrients or micronutrients based on daily intake levels. Calcium and phosphorus, which are important for bone mineralization, make up the majority of mineral content in the body. Hormones like PTH and calcitriol precisely control blood calcium and phosphorus levels by regulating absorption and excretion. Imbalances can lead to deficiencies or disorders.

![Classification of minerals in
human body:
According to body needs, minerals are divided:According to body needs, minerals are divided:
1-1-Major [MacronutrientsMajor [Macronutrients ]:]:
They are required in amountsThey are required in amounts > 100 mg / day> 100 mg / day..
They include:They include: calcium, phosphorus, magnesium,calcium, phosphorus, magnesium,
sodium, potassium, chloride and sulphur.sodium, potassium, chloride and sulphur.
2-2-Trace elements [Micronutrients]Trace elements [Micronutrients] ::
They are required in amountsThey are required in amounts < 100 mg / day< 100 mg / day..
They include:They include:
iron, copper, zinc, selenium, chromium, cobalt, iodine,iron, copper, zinc, selenium, chromium, cobalt, iodine,
fluoride, manganese and molybdenumfluoride, manganese and molybdenum..](https://image.slidesharecdn.com/minerals-asnan-2013-150511131507-lva1-app6891/85/Minerals-2-320.jpg)


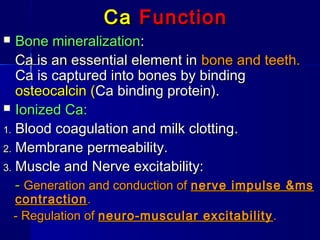


![PhPh
FunctionFunction
Bone and teethBone and teeth mineralization.mineralization.
Phosphate esterPhosphate ester compounds forcompounds for
transfer and storage of energy.transfer and storage of energy.
PhosphatePhosphate buffersbuffers system in blood.system in blood.
Nucleic acidsNucleic acids formation.formation.
Coenzymes [Coenzymes [TPP, NADP].TPP, NADP].](https://image.slidesharecdn.com/minerals-asnan-2013-150511131507-lva1-app6891/85/Minerals-9-320.jpg)




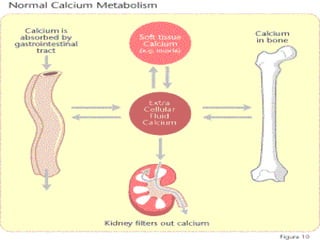
![Blood calcium & phosphorusBlood calcium & phosphorus
CaCa PhPh
Plasma level isPlasma level is
9-11 mg/dl9-11 mg/dl
diffusible:diffusible:
ionized: 50% (active)ionized: 50% (active)
non ionized: 5%non ionized: 5%
(inactive Ca citrate)(inactive Ca citrate)
non diffusible:non diffusible:
protein bound [45%].protein bound [45%].
Plasma level isPlasma level is
4-7 mg/dl in child &4-7 mg/dl in child &
3-4.5 mg/dl in adult3-4.5 mg/dl in adult
Ca : P ratioCa : P ratio is importantis important
in ossificationin ossification
In serum, the ionic productIn serum, the ionic product
of Caof Ca ×× P is about 50 mg/dlP is about 50 mg/dl
in children (in children (↓↓ in rickets)in rickets)](https://image.slidesharecdn.com/minerals-asnan-2013-150511131507-lva1-app6891/85/Minerals-14-320.jpg)









