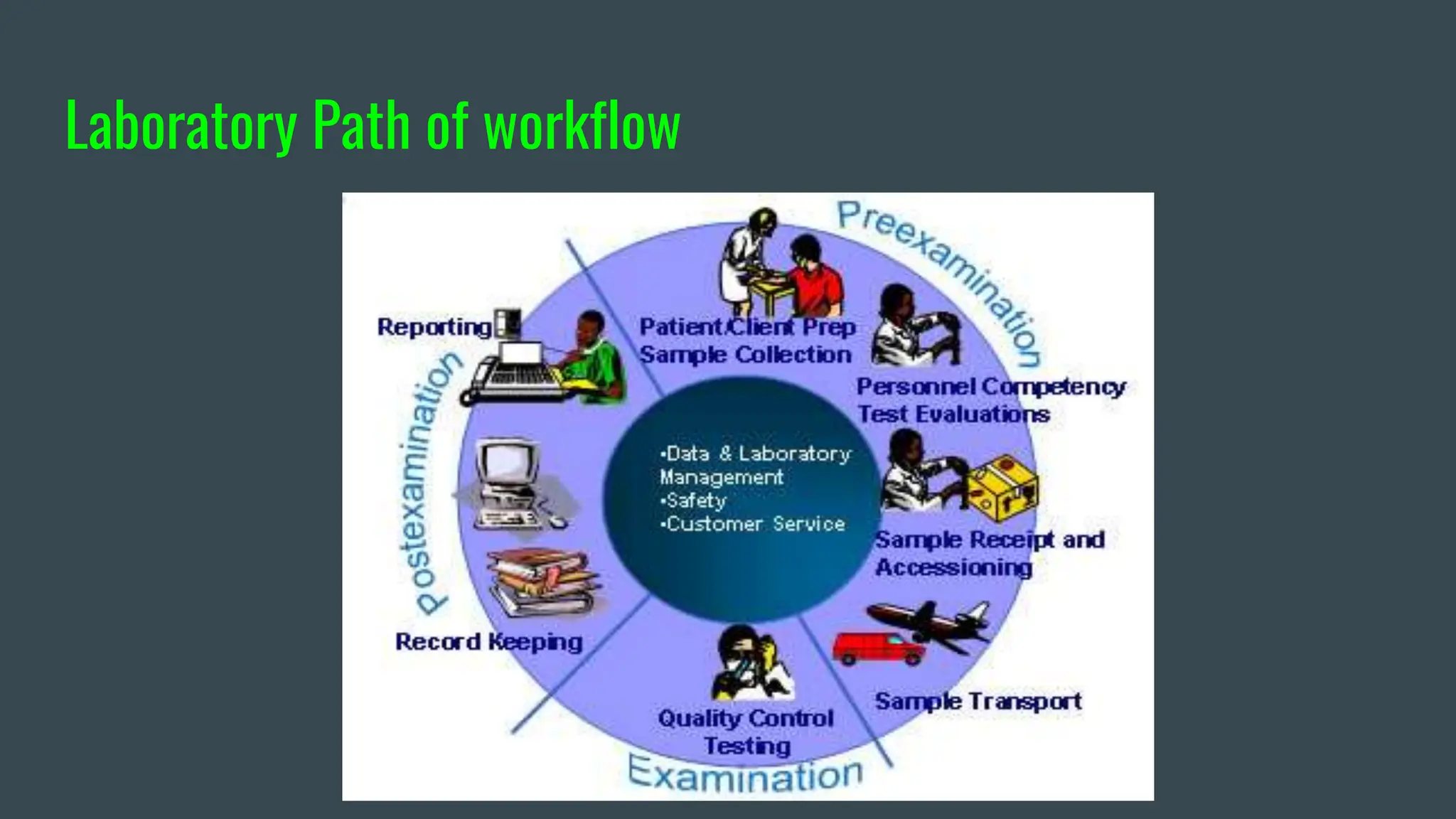
The document provides a comprehensive guide on quality control in medical laboratories, detailing its importance, the types of quality control materials, and the statistical processes used to ensure accurate and reliable laboratory results. It covers aspects such as control product selection, handling, and best practices for minimizing errors in patient testing. The guide emphasizes the significance of continuous education and adherence to quality standards for effective laboratory performance.












































































![Coefficient of Variation [CV]
The Coefficient of Variation [CV] is the ratio of the standard deviation to
the mean and is expressed as a percentage.
● The CV allows the laboratory professional to make easier comparisons
of the overall precision.
● Since standard deviation typically increases as the concentration of
the analyte increases, the CV can be regarded as a statistical
equalizer.
FORMULA: CV = (SD / MEAN) X 100%](https://image.slidesharecdn.com/qualitycontrolforclinicallaboratories-231215094718-0415196b/75/Master-Quality-control-for-clinical-laboratories-pptx-78-2048.jpg)
![Coefficient of Variation [CV]-Scenario
● For example, a comparison between hexokinase and glucose oxidase
(two methods for assaying glucose) is required
● The standard deviation for the hexokinase method is 4.8 and it is 4.0
for glucose oxidase
● If the comparison only uses standard deviation, it can be incorrectly
assumed that the glucose oxidase method is more precise than the
hexokinase method.
If, however, a CV is calculated, it might show that both methods are
equally precise](https://image.slidesharecdn.com/qualitycontrolforclinicallaboratories-231215094718-0415196b/75/Master-Quality-control-for-clinical-laboratories-pptx-79-2048.jpg)
![Coefficient of Variation [CV]-Scenario
Assume
the mean for the hexokinase method is 120 and the glucose oxidase
mean is 100. The CV then, for both methods, is 4%. They are equally
precise
Hope you get hexokinase =4.8/120 x 100% = 4%
Then oxidase = 4/100 x 100% = 4%
● NB The Coefficient of Variation can also be used when comparing
instrument performance.](https://image.slidesharecdn.com/qualitycontrolforclinicallaboratories-231215094718-0415196b/75/Master-Quality-control-for-clinical-laboratories-pptx-80-2048.jpg)
![Coefficient of Variation [CV]-Scenario
● The CV allows you to look at imprecision across multiple control levels,
even compare imprecision between methods and instruments, and
compare them against the manufacturer’s expectations.
● Many accrediting organizations have requirements that the CV must be
monitored monthly for all quantitative assays.](https://image.slidesharecdn.com/qualitycontrolforclinicallaboratories-231215094718-0415196b/75/Master-Quality-control-for-clinical-laboratories-pptx-81-2048.jpg)
![Coefficient of Variation [CV]-Scenario
● The lower the CV, the lower the test system’s imprecision](https://image.slidesharecdn.com/qualitycontrolforclinicallaboratories-231215094718-0415196b/75/Master-Quality-control-for-clinical-laboratories-pptx-82-2048.jpg)




![How to set ranges?
❖ Since it comprises multiple instruments, even multiple labs, the SD will
be larger, possibly MUCH larger,than any individual laboratory’s SD.
❖ Therefore, it is important to switch to your own SD as soon as possible
after you have derived it from the data you have captured
❖ The longer you use the package insert SD, the longer you are at risk of
having your control limits too wide and missing significant errors.
Okay, that’s not so fun.]](https://image.slidesharecdn.com/qualitycontrolforclinicallaboratories-231215094718-0415196b/75/Master-Quality-control-for-clinical-laboratories-pptx-87-2048.jpg)









































![R4s
● Level I is +2.8s above the mean and Level II is -
1.3s below the mean. The total difference between
the two control levels is greater than 4s.
(e.g. [+2.8s – (-1.3s)] = 4.1s).](https://image.slidesharecdn.com/qualitycontrolforclinicallaboratories-231215094718-0415196b/75/Master-Quality-control-for-clinical-laboratories-pptx-129-2048.jpg)









