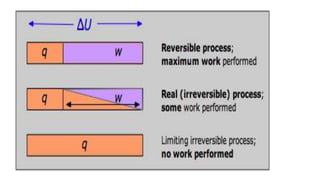
1) A reversible process is one that can proceed in either direction, returning both the system and surroundings to their original states. It occurs through infinitesimally small steps and involves no friction.
2) An irreversible process cannot reverse direction and results in a permanent change to the system or surroundings. Examples include tearing paper or free expansion of a gas into a vacuum.
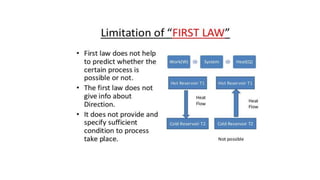
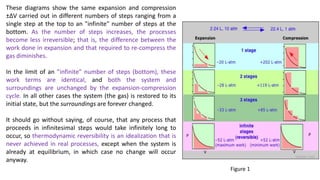
3) Reversible processes are idealizations since real processes involve some irreversibility. However, the concept is important because it establishes the minimum heat and maximum work values for a given change between states.