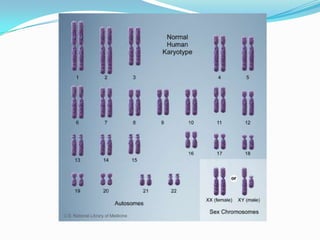
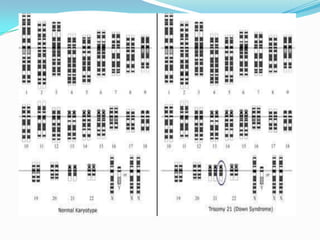
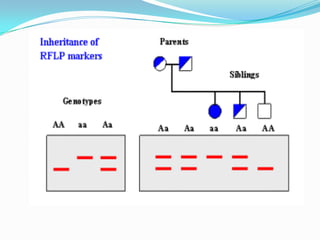
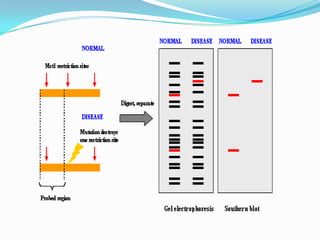
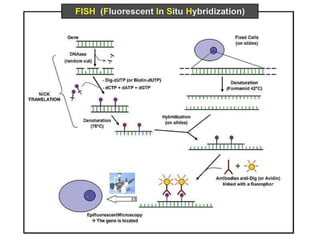
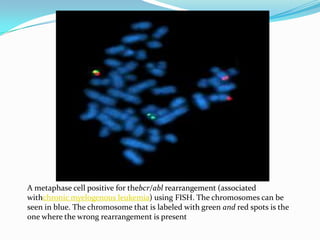
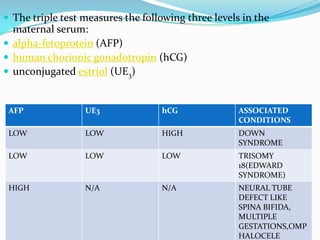
Prenatal diagnosis employs techniques like amniocentesis and chorionic villus sampling to determine the health of the unborn fetus. It is helpful for managing the pregnancy, determining outcomes, and planning for complications. It allows decisions about continuing the pregnancy and identifying conditions that could affect future pregnancies. Maternal serum screening measures markers like alpha-fetoprotein to screen for neural tube defects and other abnormalities by detecting higher than normal levels that cross the placenta.