Embed presentation
Download to read offline




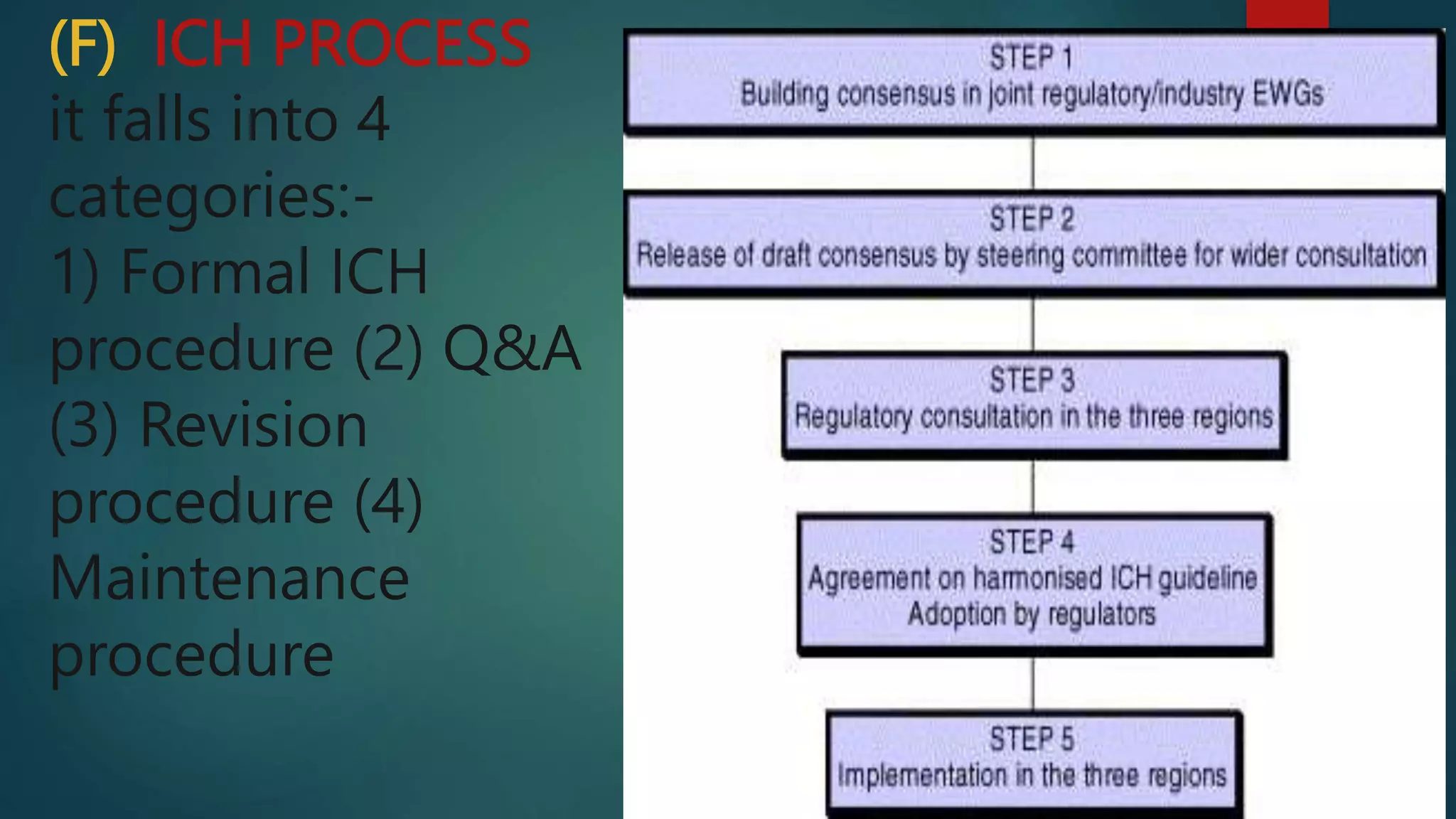
The document discusses the goals and process of the International Conference on Harmonization (ICH). The ICH aims to harmonize technical requirements for pharmaceutical registration across regions to increase efficiency and access to new medicines while maintaining quality and safety. The ICH process involves developing guidelines through formal procedures, answering questions, revising existing guidelines, and maintaining established standards.




