QBD.pptx
•Download as PPTX, PDF•
0 likes•34 views
Quality by Design (QBD) is a systematic approach to pharmaceutical development that begins with predefined objectives and emphasizes product and process understanding and control based on sound science and quality risk management. It provides benefits like less validation burden, better product design, fewer manufacturing problems, enabling continuous improvement, and lower costs. The key elements of QBD include its definition, benefits, and components like defining objectives, understanding variability, and setting in-process controls.
Report
Share
Report
Share
Recommended
Good Warehousing Practice.pptx-1.pdf

This document discusses good warehousing practices, including warehouse layout and design, safety and security measures, quality control and inspection, and environmental considerations. Proper inventory management, efficient warehouse layout, safety protocols, quality checks, and sustainability efforts are important for maintaining product quality, delivering goods efficiently, and reducing costs and environmental impact. Implementing good warehousing practices provides benefits like increased productivity, profitability, reputation, and customer satisfaction.
Complaints.pptx

1. The document outlines the standard operating procedure (SOP) for handling product complaints at a pharmaceutical company. It describes the process for receiving, investigating, taking corrective actions for complaints, and generating monthly reports.
2. Key aspects of the SOP include categorizing complaints, timelines for investigation, using a product complaint data sheet to document details, conducting documentation-based and laboratory investigations, and providing feedback and corrective actions to customers.
3. Monthly reports analyze complaint trends to assess quantities and types of complaints received. This allows the company to improve product quality and maintain good customer relationships.
ICH introduction.pptx

The International Conference on Harmonization (ICH) aims to harmonize technical requirements for pharmaceutical registration across regions to increase consistency, make more efficient use of resources, and help develop and make more medicines available while maintaining quality and efficacy. ICH seeks to standardize guidelines and requirements for drug registration to reduce costs and better meet public health needs. Its objectives include achieving greater consistency in technical guidelines, more economical use of human and material resources, and facilitating more drug development and availability with safeguards for quality and effectiveness.
Training.pptx

Training is essential for personnel at a pharma company. The manufacturer must provide training for all employees who work in production or quality control to ensure they can properly operate equipment and perform duties that affect product quality. Maintaining training records for every employee can improve quality performance by helping to ensure personnel have the necessary skills.
Responsibilities In Pharma Company

The document outlines the responsibilities of individuals in a pharma company's quality control and production departments. The head of quality control is responsible for approving or rejecting materials and finished products, validating analytical procedures and equipment calibration. The head of production is responsible for maintaining appropriate documentation to improve production instructions, and ensuring proper maintenance of departments, premises, and equipment.
Hygiene In Pharma Company

Personal hygiene is extremely important for those working in a pharmaceutical company. Workers must be healthy and have good hygiene habits like proper hand sanitization. They should avoid eating, drinking, smoking or storing food in the manufacturing unit. Anyone infected should not be permitted in the unit to prevent the spread of infection.
Elements and objectives of QBD.pptx

The document discusses the elements, objectives, benefits, and advantages of Quality by Design (QBD). QBD aims to ensure robust manufacturing processes, demonstrate understanding to regulators, and encourage sufficient product understanding. Benefits include reduced validation costs, improved product design, fewer manufacturing problems, enabling continuous improvement, and lower costs. The objectives are to have robust processes, demonstrate enhanced understanding to agencies, and develop sufficient product knowledge.
Total quality management 

Total quality management (TQM) is defined as an integrated organizational effort to improve quality at every level based on key principles: focusing on customers, continuous improvement, teamwork and mutual respect, and producing quality work the first time. TQM is characterized by process improvements, measuring processes, flexible production, and an open organization. Benefits of TQM include improved teamwork, quality, productivity, and profitability. Seven gurus contributed to the development of TQM, including Deming who proposed the PDCA cycle and Juran who developed the concept of quality trilogy.
Recommended
Good Warehousing Practice.pptx-1.pdf

This document discusses good warehousing practices, including warehouse layout and design, safety and security measures, quality control and inspection, and environmental considerations. Proper inventory management, efficient warehouse layout, safety protocols, quality checks, and sustainability efforts are important for maintaining product quality, delivering goods efficiently, and reducing costs and environmental impact. Implementing good warehousing practices provides benefits like increased productivity, profitability, reputation, and customer satisfaction.
Complaints.pptx

1. The document outlines the standard operating procedure (SOP) for handling product complaints at a pharmaceutical company. It describes the process for receiving, investigating, taking corrective actions for complaints, and generating monthly reports.
2. Key aspects of the SOP include categorizing complaints, timelines for investigation, using a product complaint data sheet to document details, conducting documentation-based and laboratory investigations, and providing feedback and corrective actions to customers.
3. Monthly reports analyze complaint trends to assess quantities and types of complaints received. This allows the company to improve product quality and maintain good customer relationships.
ICH introduction.pptx

The International Conference on Harmonization (ICH) aims to harmonize technical requirements for pharmaceutical registration across regions to increase consistency, make more efficient use of resources, and help develop and make more medicines available while maintaining quality and efficacy. ICH seeks to standardize guidelines and requirements for drug registration to reduce costs and better meet public health needs. Its objectives include achieving greater consistency in technical guidelines, more economical use of human and material resources, and facilitating more drug development and availability with safeguards for quality and effectiveness.
Training.pptx

Training is essential for personnel at a pharma company. The manufacturer must provide training for all employees who work in production or quality control to ensure they can properly operate equipment and perform duties that affect product quality. Maintaining training records for every employee can improve quality performance by helping to ensure personnel have the necessary skills.
Responsibilities In Pharma Company

The document outlines the responsibilities of individuals in a pharma company's quality control and production departments. The head of quality control is responsible for approving or rejecting materials and finished products, validating analytical procedures and equipment calibration. The head of production is responsible for maintaining appropriate documentation to improve production instructions, and ensuring proper maintenance of departments, premises, and equipment.
Hygiene In Pharma Company

Personal hygiene is extremely important for those working in a pharmaceutical company. Workers must be healthy and have good hygiene habits like proper hand sanitization. They should avoid eating, drinking, smoking or storing food in the manufacturing unit. Anyone infected should not be permitted in the unit to prevent the spread of infection.
Elements and objectives of QBD.pptx

The document discusses the elements, objectives, benefits, and advantages of Quality by Design (QBD). QBD aims to ensure robust manufacturing processes, demonstrate understanding to regulators, and encourage sufficient product understanding. Benefits include reduced validation costs, improved product design, fewer manufacturing problems, enabling continuous improvement, and lower costs. The objectives are to have robust processes, demonstrate enhanced understanding to agencies, and develop sufficient product knowledge.
Total quality management 

Total quality management (TQM) is defined as an integrated organizational effort to improve quality at every level based on key principles: focusing on customers, continuous improvement, teamwork and mutual respect, and producing quality work the first time. TQM is characterized by process improvements, measuring processes, flexible production, and an open organization. Benefits of TQM include improved teamwork, quality, productivity, and profitability. Seven gurus contributed to the development of TQM, including Deming who proposed the PDCA cycle and Juran who developed the concept of quality trilogy.
TQM Benefits and Gurus.pptx

Total quality management (TQM) aims to improve quality at all levels through an integrated organizational effort. The main TQM principles are focusing on customers, continuous improvement, teamwork, and producing quality work. TQM is characterized by process improvements, measuring processes, flexible production, and an open organization. Benefits of TQM include improved teamwork, quality, productivity, and profitability. The seven gurus of TQM who developed key concepts include Walter Shewhart, W. Edwards Deming, Joseph Juran, Armand Feigenbaum, Philip Crosby, Genichi Taguchi, and Kaoru Ishikawa.
TQM Advantages & disadvantages.pptx

Total quality management (TQM) aims to improve quality at all levels through an integrated organizational effort focused on customer satisfaction. Key elements of TQM include focusing on the customer, employee involvement, and continuous improvement. Some advantages of TQM are improved quality, employee participation, teamwork, and better working relationships, while disadvantages can include initial introduction costs and worker resistance to change. TQM is important in the pharmaceutical industry for ensuring quality in areas like handling, storage, packaging, facilities, equipment, and sterile areas.
ICH goals and process.pptx

The document discusses the goals and process of the International Conference on Harmonization (ICH). The ICH aims to harmonize technical requirements for pharmaceutical registration across regions to increase efficiency and access to new medicines while maintaining quality and safety. The ICH process involves developing guidelines through formal procedures, answering questions, revising existing guidelines, and maintaining established standards.
Introduction to ICH.pptx

The document introduces the International Conference on Harmonization (ICH), which aims to harmonize technical requirements for pharmaceutical registration across regions to increase consistency, make more efficient use of resources, and help develop new medicines while maintaining safety and effectiveness. ICH seeks to standardize guidelines and registration processes for drugs to reduce costs and better meet public health needs.
Mixing.pptx

The document discusses mixing in the context of pharmaceutical production. It outlines that the main objective of mixing is to produce a bulk mixture that, when divided into doses, ensures each dose contains the correct proportions. It notes mixing is a critical intermediate step for tablets and capsules. The document also lists factors that affect mixing and different mechanisms for solid, liquid, and semisolid mixing. Specifically, it explains solid mixing involves convective, shear, and diffusive mechanisms while liquid mixing uses bulk transport, turbulent flow, laminar flow, and molecular diffusion.
Freeze dryer.pptx

Freeze drying, or lyophilisation, is a process that converts solutions or suspensions into solids for storage and distribution. It works by sublimating a material, changing it directly from a solid to a gas, below the triple point. Key components of a freeze dryer include a drying chamber, heating elements, a vapor condensing system, and a vacuum pump. Freeze drying has advantages like preserving heat-sensitive materials and proteins without denaturation. However, it is an expensive and slow process limited to specific valuable products.
Vacuum dryer 

The document describes a vacuum dryer used to dry heat sensitive and hygroscopic materials. It works by reducing chamber pressure below water vapor pressure through a vacuum pump, allowing water to boil and evaporate faster. Trays inside the hollow oven increase surface area for heat conduction. Materials are dried at 25-30°C as water vapor is evaporated under vacuum and condenses before being collected.
FLUIDIZED BED DRYER

The document summarizes a fluidized bed dryer. It works by fluidizing wet solid particles using hot air forced through a perforated bed from the bottom. The dryer is made of stainless steel or plastic with a detachable bowl for charging and discharging materials. Hot air is circulated through the bed to dry particles completely before exiting through bag filters. It allows for continuous, rapid drying at a constant rate with high efficiency and capacity compared to other dryers. Pharmaceutical uses include drying granules for tablet production and granule coating.
SPRAY DRYER

Spray drying is an industrial process that transforms a liquid feed containing dissolved or dispersed solids into a spray of small droplets that are exposed to hot air to be dried. It allows for effective control of product quality and properties during continuous high-tonnage production. While spray dryers are bulky and expensive, they permit thermolabile pharmaceuticals to be dried at low temperature and are used to dry products like penicillin, enzymes, and vaccines.
Drum Dryer

The document describes a drum dryer, which consists of one or two horizontally mounted hollow cylinders or drums that are heated internally by steam and rotated. Liquid or slurry is spread as a film onto the heated, rotating drum surface where it dries rapidly via conduction and steam heating. The dried material is then scraped off and collected while the drum continues rotating. Drum drying allows for rapid drying of thermosensitive materials in a compact system, though it requires careful control of processing parameters and has high maintenance costs.
tray drayer.pptx

The tray dryer uses forced hot air convection to remove moisture from solids placed on stainless steel trays. It has double-walled insulation and doors with gaskets. Hot air is blown through the chamber at 2-5 meters per second to evaporate water from the surface of the solids on each tray, which are loaded and unloaded batch-wise. Tray dryers are efficient, require little labor, and are available with capacities from 6 to 192 trays.
Mechanism f drying

The document discusses the mechanisms and principles of drying, including that drying involves the removal of moisture through evaporation and transport using heat, and describes key terms used in drying systems like bound water, free moisture content, equilibrium moisture content, and critical moisture content. It also provides a classification of different types of dryers based on their mode of operation and heating method.
Drying.pptx

Drying involves the removal of water or other solvents from solids, semi-solids, or liquids through the application of heat to produce a liquid-free solid product. The main objectives of drying are to preserve foods by reducing water content and activity to increase shelf life without refrigeration, reduce storage and transport costs, and improve stability of pharmaceutical products by removing moisture. Drying techniques include thermal methods using heat as well as non-thermal methods like squeezing, adsorption, or extraction. Water is the solvent most commonly removed through drying of bioproducts like foods and pharmaceuticals like vaccines.
DRYING.pptx

This document discusses drying and its objectives and applications in pharmaceutical technology. It begins by defining drying as the removal of water or other solvents from a solid, semi-solid, or liquid material through the application of heat. The main objectives of drying are to preserve foods and drugs by reducing water content and avoiding the need for refrigeration during storage and transport. In pharmaceuticals specifically, drying is used to avoid moisture that can degrade drugs, improve material properties, reduce volume for transportation and handling, and serve as the final step in various processes like evaporation and crystallization. The document provides examples of how drying is used in preparing bulk drugs and preserving drug products and materials.
Heat transfer Mechanism.pptx

Heat transfer is a form of energy associated with the motion of atoms or molecules that is transferred from higher temperature objects to lower temperature objects. Precise temperature control through heat transfer is important for pharmaceutical manufacturing processes like purification where applying heat causes crystallization. Various industries like oil and gas processing utilize heat transfer mechanisms like conduction, convection, and radiation using heat transfer fluids to control temperatures in heating and cooling phases. Heat transfer can be classified based on the process of heat flowing from higher to lower temperature objects through conduction, convection, or radiation.
evaporation.pptx

Evaporation is the process by which a liquid is vaporized from a solution, leaving behind a more concentrated residue. It is commonly used to prepare liquid, soft, and dry extracts from solutions by removing the solvent through heating. Evaporation is also employed to concentrate blood plasma, serum, enzymes, hormones, antibiotics, and other agents. The rate of evaporation is influenced by factors like temperature, time, moisture content, the type of desired product, and concentration of the solution.
VACCUM DISTILLATION.pptx

Vacuum distillation is a distillation technique where the pressure above the liquid mixture is reduced below the vapor pressure of the mixture. This allows distillation to occur at lower temperatures than normal distillation, making it suitable for heat-sensitive materials. Vacuum distillation works by causing boiling when the vapor pressure exceeds the reduced ambient pressure. The document provides an introduction to distillation and vacuum distillation, explaining that distillation separates mixtures based on differences in boiling points under reduced pressure.
STEAM DISTILLATION.pptx

Steam distillation is a process that uses steam to separate high boiling substances from non-volatile impurities. It works by introducing steam into a mixture, which causes volatile components to evaporate more readily than they otherwise would. This allows for the separation of volatile oils and other compounds from immiscible liquids and water. Steam distillation is useful for extracting volatile oils from plants like clove and eucalyptus because it can separate these oils at lower temperatures than simple distillation, avoiding potential degradation of heat-sensitive compounds.
Simple distillation.pptx

Distillation is a process that separates mixtures into their component parts based on differences in boiling points. Simple distillation involves a single stage of vaporization and condensation to separate components with large boiling point differences of 25-35°C, such as separating volatile liquids from nonvolatile liquids. It is used to purify seawater but only provides an impure product. The document discusses the basic principles and six types of distillation processes including simple, flash, fractional, reduced pressure, steam, and molecular distillation.
MOLECULAR Distillation.pptx

Molecular distillation is a separation technique that works on a molecular level under vacuum conditions. It takes advantage of the differences in vapor pressure and boiling points between mixture components to allow individual molecules to travel freely in the vapor phase without colliding before condensing in a purified state. The document introduces molecular distillation and conventional distillation, noting that molecular distillation is useful for purifying chemicals, fixed oils, and separating components of fish liver oil as it can achieve separation at the single molecule level under vacuum.
How to Make a Field Mandatory in Odoo 17

In Odoo, making a field required can be done through both Python code and XML views. When you set the required attribute to True in Python code, it makes the field required across all views where it's used. Conversely, when you set the required attribute in XML views, it makes the field required only in the context of that particular view.
Gender and Mental Health - Counselling and Family Therapy Applications and In...

A proprietary approach developed by bringing together the best of learning theories from Psychology, design principles from the world of visualization, and pedagogical methods from over a decade of training experience, that enables you to: Learn better, faster!
More Related Content
More from KashishWilson1
TQM Benefits and Gurus.pptx

Total quality management (TQM) aims to improve quality at all levels through an integrated organizational effort. The main TQM principles are focusing on customers, continuous improvement, teamwork, and producing quality work. TQM is characterized by process improvements, measuring processes, flexible production, and an open organization. Benefits of TQM include improved teamwork, quality, productivity, and profitability. The seven gurus of TQM who developed key concepts include Walter Shewhart, W. Edwards Deming, Joseph Juran, Armand Feigenbaum, Philip Crosby, Genichi Taguchi, and Kaoru Ishikawa.
TQM Advantages & disadvantages.pptx

Total quality management (TQM) aims to improve quality at all levels through an integrated organizational effort focused on customer satisfaction. Key elements of TQM include focusing on the customer, employee involvement, and continuous improvement. Some advantages of TQM are improved quality, employee participation, teamwork, and better working relationships, while disadvantages can include initial introduction costs and worker resistance to change. TQM is important in the pharmaceutical industry for ensuring quality in areas like handling, storage, packaging, facilities, equipment, and sterile areas.
ICH goals and process.pptx

The document discusses the goals and process of the International Conference on Harmonization (ICH). The ICH aims to harmonize technical requirements for pharmaceutical registration across regions to increase efficiency and access to new medicines while maintaining quality and safety. The ICH process involves developing guidelines through formal procedures, answering questions, revising existing guidelines, and maintaining established standards.
Introduction to ICH.pptx

The document introduces the International Conference on Harmonization (ICH), which aims to harmonize technical requirements for pharmaceutical registration across regions to increase consistency, make more efficient use of resources, and help develop new medicines while maintaining safety and effectiveness. ICH seeks to standardize guidelines and registration processes for drugs to reduce costs and better meet public health needs.
Mixing.pptx

The document discusses mixing in the context of pharmaceutical production. It outlines that the main objective of mixing is to produce a bulk mixture that, when divided into doses, ensures each dose contains the correct proportions. It notes mixing is a critical intermediate step for tablets and capsules. The document also lists factors that affect mixing and different mechanisms for solid, liquid, and semisolid mixing. Specifically, it explains solid mixing involves convective, shear, and diffusive mechanisms while liquid mixing uses bulk transport, turbulent flow, laminar flow, and molecular diffusion.
Freeze dryer.pptx

Freeze drying, or lyophilisation, is a process that converts solutions or suspensions into solids for storage and distribution. It works by sublimating a material, changing it directly from a solid to a gas, below the triple point. Key components of a freeze dryer include a drying chamber, heating elements, a vapor condensing system, and a vacuum pump. Freeze drying has advantages like preserving heat-sensitive materials and proteins without denaturation. However, it is an expensive and slow process limited to specific valuable products.
Vacuum dryer 

The document describes a vacuum dryer used to dry heat sensitive and hygroscopic materials. It works by reducing chamber pressure below water vapor pressure through a vacuum pump, allowing water to boil and evaporate faster. Trays inside the hollow oven increase surface area for heat conduction. Materials are dried at 25-30°C as water vapor is evaporated under vacuum and condenses before being collected.
FLUIDIZED BED DRYER

The document summarizes a fluidized bed dryer. It works by fluidizing wet solid particles using hot air forced through a perforated bed from the bottom. The dryer is made of stainless steel or plastic with a detachable bowl for charging and discharging materials. Hot air is circulated through the bed to dry particles completely before exiting through bag filters. It allows for continuous, rapid drying at a constant rate with high efficiency and capacity compared to other dryers. Pharmaceutical uses include drying granules for tablet production and granule coating.
SPRAY DRYER

Spray drying is an industrial process that transforms a liquid feed containing dissolved or dispersed solids into a spray of small droplets that are exposed to hot air to be dried. It allows for effective control of product quality and properties during continuous high-tonnage production. While spray dryers are bulky and expensive, they permit thermolabile pharmaceuticals to be dried at low temperature and are used to dry products like penicillin, enzymes, and vaccines.
Drum Dryer

The document describes a drum dryer, which consists of one or two horizontally mounted hollow cylinders or drums that are heated internally by steam and rotated. Liquid or slurry is spread as a film onto the heated, rotating drum surface where it dries rapidly via conduction and steam heating. The dried material is then scraped off and collected while the drum continues rotating. Drum drying allows for rapid drying of thermosensitive materials in a compact system, though it requires careful control of processing parameters and has high maintenance costs.
tray drayer.pptx

The tray dryer uses forced hot air convection to remove moisture from solids placed on stainless steel trays. It has double-walled insulation and doors with gaskets. Hot air is blown through the chamber at 2-5 meters per second to evaporate water from the surface of the solids on each tray, which are loaded and unloaded batch-wise. Tray dryers are efficient, require little labor, and are available with capacities from 6 to 192 trays.
Mechanism f drying

The document discusses the mechanisms and principles of drying, including that drying involves the removal of moisture through evaporation and transport using heat, and describes key terms used in drying systems like bound water, free moisture content, equilibrium moisture content, and critical moisture content. It also provides a classification of different types of dryers based on their mode of operation and heating method.
Drying.pptx

Drying involves the removal of water or other solvents from solids, semi-solids, or liquids through the application of heat to produce a liquid-free solid product. The main objectives of drying are to preserve foods by reducing water content and activity to increase shelf life without refrigeration, reduce storage and transport costs, and improve stability of pharmaceutical products by removing moisture. Drying techniques include thermal methods using heat as well as non-thermal methods like squeezing, adsorption, or extraction. Water is the solvent most commonly removed through drying of bioproducts like foods and pharmaceuticals like vaccines.
DRYING.pptx

This document discusses drying and its objectives and applications in pharmaceutical technology. It begins by defining drying as the removal of water or other solvents from a solid, semi-solid, or liquid material through the application of heat. The main objectives of drying are to preserve foods and drugs by reducing water content and avoiding the need for refrigeration during storage and transport. In pharmaceuticals specifically, drying is used to avoid moisture that can degrade drugs, improve material properties, reduce volume for transportation and handling, and serve as the final step in various processes like evaporation and crystallization. The document provides examples of how drying is used in preparing bulk drugs and preserving drug products and materials.
Heat transfer Mechanism.pptx

Heat transfer is a form of energy associated with the motion of atoms or molecules that is transferred from higher temperature objects to lower temperature objects. Precise temperature control through heat transfer is important for pharmaceutical manufacturing processes like purification where applying heat causes crystallization. Various industries like oil and gas processing utilize heat transfer mechanisms like conduction, convection, and radiation using heat transfer fluids to control temperatures in heating and cooling phases. Heat transfer can be classified based on the process of heat flowing from higher to lower temperature objects through conduction, convection, or radiation.
evaporation.pptx

Evaporation is the process by which a liquid is vaporized from a solution, leaving behind a more concentrated residue. It is commonly used to prepare liquid, soft, and dry extracts from solutions by removing the solvent through heating. Evaporation is also employed to concentrate blood plasma, serum, enzymes, hormones, antibiotics, and other agents. The rate of evaporation is influenced by factors like temperature, time, moisture content, the type of desired product, and concentration of the solution.
VACCUM DISTILLATION.pptx

Vacuum distillation is a distillation technique where the pressure above the liquid mixture is reduced below the vapor pressure of the mixture. This allows distillation to occur at lower temperatures than normal distillation, making it suitable for heat-sensitive materials. Vacuum distillation works by causing boiling when the vapor pressure exceeds the reduced ambient pressure. The document provides an introduction to distillation and vacuum distillation, explaining that distillation separates mixtures based on differences in boiling points under reduced pressure.
STEAM DISTILLATION.pptx

Steam distillation is a process that uses steam to separate high boiling substances from non-volatile impurities. It works by introducing steam into a mixture, which causes volatile components to evaporate more readily than they otherwise would. This allows for the separation of volatile oils and other compounds from immiscible liquids and water. Steam distillation is useful for extracting volatile oils from plants like clove and eucalyptus because it can separate these oils at lower temperatures than simple distillation, avoiding potential degradation of heat-sensitive compounds.
Simple distillation.pptx

Distillation is a process that separates mixtures into their component parts based on differences in boiling points. Simple distillation involves a single stage of vaporization and condensation to separate components with large boiling point differences of 25-35°C, such as separating volatile liquids from nonvolatile liquids. It is used to purify seawater but only provides an impure product. The document discusses the basic principles and six types of distillation processes including simple, flash, fractional, reduced pressure, steam, and molecular distillation.
MOLECULAR Distillation.pptx

Molecular distillation is a separation technique that works on a molecular level under vacuum conditions. It takes advantage of the differences in vapor pressure and boiling points between mixture components to allow individual molecules to travel freely in the vapor phase without colliding before condensing in a purified state. The document introduces molecular distillation and conventional distillation, noting that molecular distillation is useful for purifying chemicals, fixed oils, and separating components of fish liver oil as it can achieve separation at the single molecule level under vacuum.
More from KashishWilson1 (20)
Recently uploaded
How to Make a Field Mandatory in Odoo 17

In Odoo, making a field required can be done through both Python code and XML views. When you set the required attribute to True in Python code, it makes the field required across all views where it's used. Conversely, when you set the required attribute in XML views, it makes the field required only in the context of that particular view.
Gender and Mental Health - Counselling and Family Therapy Applications and In...

A proprietary approach developed by bringing together the best of learning theories from Psychology, design principles from the world of visualization, and pedagogical methods from over a decade of training experience, that enables you to: Learn better, faster!
Temple of Asclepius in Thrace. Excavation results

The temple and the sanctuary around were dedicated to Asklepios Zmidrenus. This name has been known since 1875 when an inscription dedicated to him was discovered in Rome. The inscription is dated in 227 AD and was left by soldiers originating from the city of Philippopolis (modern Plovdiv).
Your Skill Boost Masterclass: Strategies for Effective Upskilling

Your Skill Boost Masterclass: Strategies for Effective UpskillingExcellence Foundation for South Sudan
Strategies for Effective Upskilling is a presentation by Chinwendu Peace in a Your Skill Boost Masterclass organisation by the Excellence Foundation for South Sudan on 08th and 09th June 2024 from 1 PM to 3 PM on each day.Chapter wise All Notes of First year Basic Civil Engineering.pptx

Chapter wise All Notes of First year Basic Civil Engineering
Syllabus
Chapter-1
Introduction to objective, scope and outcome the subject
Chapter 2
Introduction: Scope and Specialization of Civil Engineering, Role of civil Engineer in Society, Impact of infrastructural development on economy of country.
Chapter 3
Surveying: Object Principles & Types of Surveying; Site Plans, Plans & Maps; Scales & Unit of different Measurements.
Linear Measurements: Instruments used. Linear Measurement by Tape, Ranging out Survey Lines and overcoming Obstructions; Measurements on sloping ground; Tape corrections, conventional symbols. Angular Measurements: Instruments used; Introduction to Compass Surveying, Bearings and Longitude & Latitude of a Line, Introduction to total station.
Levelling: Instrument used Object of levelling, Methods of levelling in brief, and Contour maps.
Chapter 4
Buildings: Selection of site for Buildings, Layout of Building Plan, Types of buildings, Plinth area, carpet area, floor space index, Introduction to building byelaws, concept of sun light & ventilation. Components of Buildings & their functions, Basic concept of R.C.C., Introduction to types of foundation
Chapter 5
Transportation: Introduction to Transportation Engineering; Traffic and Road Safety: Types and Characteristics of Various Modes of Transportation; Various Road Traffic Signs, Causes of Accidents and Road Safety Measures.
Chapter 6
Environmental Engineering: Environmental Pollution, Environmental Acts and Regulations, Functional Concepts of Ecology, Basics of Species, Biodiversity, Ecosystem, Hydrological Cycle; Chemical Cycles: Carbon, Nitrogen & Phosphorus; Energy Flow in Ecosystems.
Water Pollution: Water Quality standards, Introduction to Treatment & Disposal of Waste Water. Reuse and Saving of Water, Rain Water Harvesting. Solid Waste Management: Classification of Solid Waste, Collection, Transportation and Disposal of Solid. Recycling of Solid Waste: Energy Recovery, Sanitary Landfill, On-Site Sanitation. Air & Noise Pollution: Primary and Secondary air pollutants, Harmful effects of Air Pollution, Control of Air Pollution. . Noise Pollution Harmful Effects of noise pollution, control of noise pollution, Global warming & Climate Change, Ozone depletion, Greenhouse effect
Text Books:
1. Palancharmy, Basic Civil Engineering, McGraw Hill publishers.
2. Satheesh Gopi, Basic Civil Engineering, Pearson Publishers.
3. Ketki Rangwala Dalal, Essentials of Civil Engineering, Charotar Publishing House.
4. BCP, Surveying volume 1
Bed Making ( Introduction, Purpose, Types, Articles, Scientific principles, N...

Topic : Bed making
Subject : Nursing Foundation
The History of Stoke Newington Street Names

Presented at the Stoke Newington Literary Festival on 9th June 2024
www.StokeNewingtonHistory.com
How to Create a More Engaging and Human Online Learning Experience 

How to Create a More Engaging and Human Online Learning Experience Wahiba Chair Training & Consulting
Wahiba Chair's Talk at the 2024 Learning Ideas Conference. BÀI TẬP BỔ TRỢ TIẾNG ANH 8 CẢ NĂM - GLOBAL SUCCESS - NĂM HỌC 2023-2024 (CÓ FI...

BÀI TẬP BỔ TRỢ TIẾNG ANH 8 CẢ NĂM - GLOBAL SUCCESS - NĂM HỌC 2023-2024 (CÓ FI...Nguyen Thanh Tu Collection
https://app.box.com/s/y977uz6bpd3af4qsebv7r9b7s21935vdTraditional Musical Instruments of Arunachal Pradesh and Uttar Pradesh - RAYH...

Traditional Musical Instruments of Arunachal Pradesh and Uttar Pradesh
Hindi varnamala | hindi alphabet PPT.pdf

हिंदी वर्णमाला पीपीटी, hindi alphabet PPT presentation, hindi varnamala PPT, Hindi Varnamala pdf, हिंदी स्वर, हिंदी व्यंजन, sikhiye hindi varnmala, dr. mulla adam ali, hindi language and literature, hindi alphabet with drawing, hindi alphabet pdf, hindi varnamala for childrens, hindi language, hindi varnamala practice for kids, https://www.drmullaadamali.com
How to deliver Powerpoint Presentations.pptx

"How to make and deliver dynamic presentations by making it more interactive to captivate your audience attention"
Wound healing PPT

This document provides an overview of wound healing, its functions, stages, mechanisms, factors affecting it, and complications.
A wound is a break in the integrity of the skin or tissues, which may be associated with disruption of the structure and function.
Healing is the body’s response to injury in an attempt to restore normal structure and functions.
Healing can occur in two ways: Regeneration and Repair
There are 4 phases of wound healing: hemostasis, inflammation, proliferation, and remodeling. This document also describes the mechanism of wound healing. Factors that affect healing include infection, uncontrolled diabetes, poor nutrition, age, anemia, the presence of foreign bodies, etc.
Complications of wound healing like infection, hyperpigmentation of scar, contractures, and keloid formation.
Pengantar Penggunaan Flutter - Dart programming language1.pptx

Pengantar Penggunaan Flutter - Dart programming language1.pptx
How to Setup Warehouse & Location in Odoo 17 Inventory

In this slide, we'll explore how to set up warehouses and locations in Odoo 17 Inventory. This will help us manage our stock effectively, track inventory levels, and streamline warehouse operations.
Recently uploaded (20)
Gender and Mental Health - Counselling and Family Therapy Applications and In...

Gender and Mental Health - Counselling and Family Therapy Applications and In...
Film vocab for eal 3 students: Australia the movie

Film vocab for eal 3 students: Australia the movie
Your Skill Boost Masterclass: Strategies for Effective Upskilling

Your Skill Boost Masterclass: Strategies for Effective Upskilling
Chapter wise All Notes of First year Basic Civil Engineering.pptx

Chapter wise All Notes of First year Basic Civil Engineering.pptx
Bed Making ( Introduction, Purpose, Types, Articles, Scientific principles, N...

Bed Making ( Introduction, Purpose, Types, Articles, Scientific principles, N...
How to Create a More Engaging and Human Online Learning Experience 

How to Create a More Engaging and Human Online Learning Experience
BÀI TẬP BỔ TRỢ TIẾNG ANH 8 CẢ NĂM - GLOBAL SUCCESS - NĂM HỌC 2023-2024 (CÓ FI...

BÀI TẬP BỔ TRỢ TIẾNG ANH 8 CẢ NĂM - GLOBAL SUCCESS - NĂM HỌC 2023-2024 (CÓ FI...
Traditional Musical Instruments of Arunachal Pradesh and Uttar Pradesh - RAYH...

Traditional Musical Instruments of Arunachal Pradesh and Uttar Pradesh - RAYH...
NEWSPAPERS - QUESTION 1 - REVISION POWERPOINT.pptx

NEWSPAPERS - QUESTION 1 - REVISION POWERPOINT.pptx
Pengantar Penggunaan Flutter - Dart programming language1.pptx

Pengantar Penggunaan Flutter - Dart programming language1.pptx
How to Setup Warehouse & Location in Odoo 17 Inventory

How to Setup Warehouse & Location in Odoo 17 Inventory
QBD.pptx
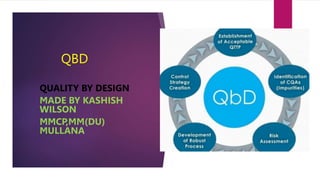
- 1. QBD QUALITY BY DESIGN MADE BY KASHISH WILSON MMCP,MM(DU) MULLANA
- 2. CONTENT OF QBD--- Definition of QBD. Benefits of QBD Elements of QBD
- 3. Elements of QBD –
- 5. Benefits of QBD -- Less validation burden. Better design of product. Fewer problem in manufacturing. Enabling continuous improvement. Low cost.