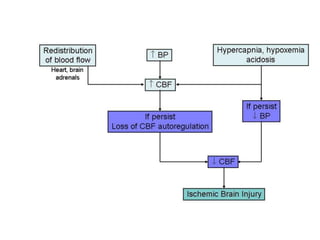
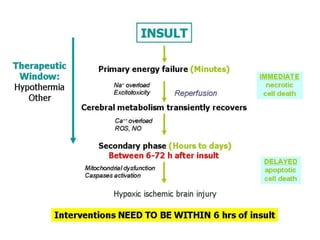
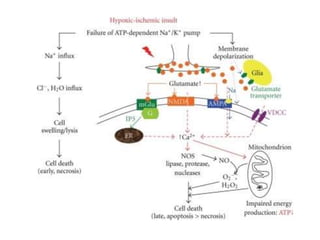
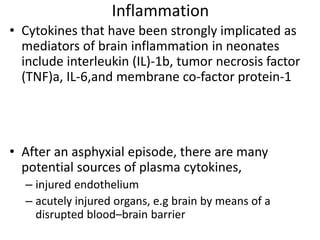
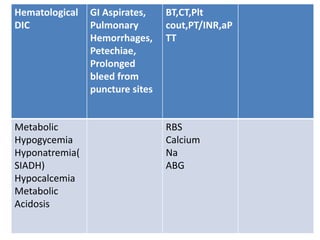
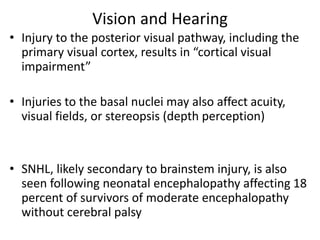
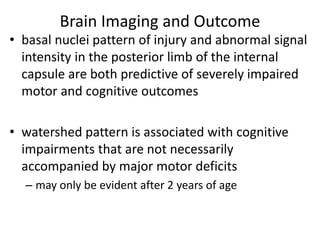
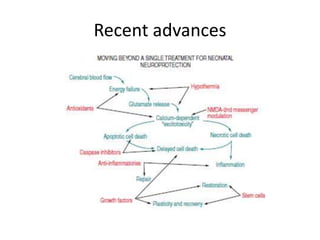
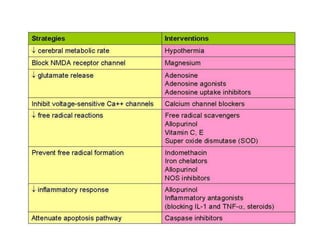
This document discusses hypoxic ischemic encephalopathy (HIE), focusing on recent advances. It provides data on the scope of the problem, defining key terms like HIE, perinatal asphyxia, and neonatal encephalopathy. It discusses the etiology and risk factors for HIE, as well as the pathophysiology involving disrupted cerebral blood flow, energy metabolism, excitotoxicity, oxidative stress, inflammation, and apoptosis. Indian data on HIE is presented based on Apgar scores and neurological manifestations.











![• Hypoxic-ischemic encephalopathy (HIE) describes
encephalopathy as defined before, with objective data to
support a hypoxic-ischemic mechanism as the underlying
cause for the encephalopathy.
• Hypoxic-ischemic (HI) brain injury refers to
neuropathology attributable to hypoxia and/or ischemia
as evidenced by
– biochemical (such as serum creatine k inase brain bound [CK-
BB]),
– electrophysiologic (EEG),
– neuroimaging (head ultrasonography [HUS], magnetic resonance
imaging [MRI], computed tomog-raphy [CT]),
– pathologic (postmortem) abnormalities.](https://image.slidesharecdn.com/hypoxicischemicencephalopathywithafocusonrecentadvances-150324124444-conversion-gate01/85/Hypoxic-ischemic-encephalopathy-with-a-focus-on-recent-advances-12-320.jpg)





























































































![• Conflicting data exist regarding the effectiveness
of MgSO4 as a neuroprotective agent.
• Prenatal administration of MgSO4 to mothers at
risk for preterm delivery is associated with
reduced incidence of cerebral palsy at 3 years and
improved neurodevelopmental outcomes
• When MgSO4 was administered postnatally to
term infants with HIE, there was no improvement
in their amplitude-integrated EEG, and when
administered in large doses, MgSO4 can cause
profound hypotension.[26]](https://image.slidesharecdn.com/hypoxicischemicencephalopathywithafocusonrecentadvances-150324124444-conversion-gate01/85/Hypoxic-ischemic-encephalopathy-with-a-focus-on-recent-advances-123-320.jpg)









![• .
• Seizures
• HIE is often reported to be the most frequent cause of neonatal seizures. They usually occur 12-24 hours after birth and are difficult to control with
anticonvulsants. Large, unilateral infarcts occur with neonatal seizures in as many as 80% of patients. Seizures are often partial (focal) and
contralateral to the cortical lesion. About two thirds of newborns with cerebral venous infarcts have seizures. Those with multiple or diffuse lesions
and cerebral venous infarcts often have multifocal or migratory seizures. Seizures are observed during physical examination and may confirm the
diagnosis. Observation often reveals clonic rhythmic contractions. When holding the limb affected by clonic seizures, the examiner's hand shakes or
feels limb movement. Limb flexion or extension does not suppress the clonic activity, as it does in jitteriness and clonus. Newborn infants cannot
have generalized seizures due to immaturity of the neuronal pathways connecting the 2 halves of the brain.
• Tonic, unilateral, or focal seizures consistently have an EEG signature. In the seizures, unilateral arm and leg posturing is often accompanied by
ipsilateral trunk flexion. Generalized tonic posturing (eg, extension of the upper and lower extremities or extension of the legs and flexion of the
arms) is related to an EEG seizure in 15% of affected neonates.
• Tonic seizures can be seen in neonates with local anesthetic intoxication. Although generalized tonic posturing is infrequently associated with
electrical seizures, it is not a benign sign. Of neonates with tonic posturing and an abnormal EEG background, 13% have normal development.
• Subtle seizures may be a part of the HIE-NE picture. Subtle manifestations of neonatal seizures are confirmed on EEG and include apnea; tonic eye
deviation; sustained eye opening; slow, rhythmic, tongue thrusting; and boxing, bicycling, and swimming movements. Most still accept that some
subtle seizures may be correlated with EEG results. However, publications since the late 1980s have shown that seizures are not as frequent as
previously thought and that they are unusual in patients close to term. Several other patterns of subtle neonatal seizures are described without EEG
confirmation. The lack of an EEG signature does not exclude CNS pathology because neonates with HIE often have motor automatisms without EEG
seizures. Management is controversial, but treatment is not usually beneficial unless more overt seizure activity is noted.[31]
• Seizures may be difficult to clinically diagnose in the premature neonate. Subtle seizures associated with ictal EEG changes are not rare in premature
infants. The subtle patterns of neonatal seizures in the premature infant include sustained eye opening, oral-buccal-lingual movements (smacking,
drooling, chewing), pedaling movements, grimacing, and autonomic manifestations.[#sarnat]](https://image.slidesharecdn.com/hypoxicischemicencephalopathywithafocusonrecentadvances-150324124444-conversion-gate01/85/Hypoxic-ischemic-encephalopathy-with-a-focus-on-recent-advances-133-320.jpg)
