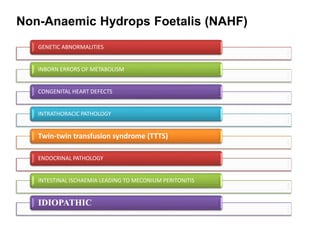
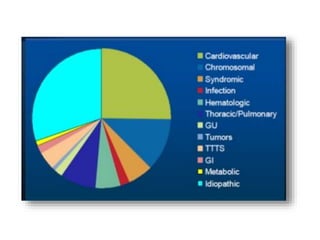
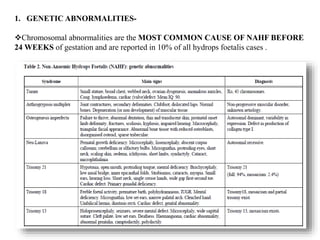
Hydrops fetalis, or fetal hydrops, is an abnormal accumulation of fluid in fetal compartments and body cavities. It is classified as immune or non-immune. Immune hydrops is caused by maternal-fetal blood group incompatibility while non-immune hydrops has various potential causes including genetic abnormalities, infections, heart defects, and tumors. Diagnosis involves ultrasound to detect fluid accumulation while treatment depends on the underlying etiology but may include fetal transfusions, medications, or early delivery. Prognosis varies significantly depending on the cause and gestational age at presentation and treatment.