The document discusses a plant health management standard and audits. It provides information on the plant health management standard (PHMS) which outlines requirements for businesses to operate responsibly to improve plant health and biosecurity. It discusses the plant healthy website and plant health assurance scheme (PHAS) which are related initiatives. The document also gives an overview of what an audit of the standard would involve, examining the paperwork, site assessment, pest risk analysis, supply chain management, and other areas to check compliance with the PHMS requirements.













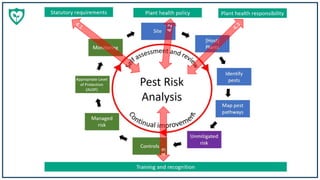
![Stages adapted from the
International Plant Protection
Convention’s framework for
pest risk analysis. For the
purposes of applying the
principles at the site level.
Pest Risk
Analysis
Unmitigated
risk The level of risk to the business /
organisation and environment if
appropriate controls are NOT in place –
see - UK Plant Health Risk Register
(2) Pest Risk Assessment
Site
[Host]
Plants
Identify
pests
Map pest
pathways
List plants handled by a business (run
through this process with specifications of
plant and new species handled)
Pests that can infest plants
handled by business – see
Plant Health Risk Register
How the pests can arrive,
proliferate and be transferred
from the site (e.g. through
trade, visitors or natural
processes).
Define site boundaries (nursery, garden
centre, landscapers yard, garden)(1) Initiation
Controls
Managed
risk
Appropriate Level
of Protection
(ALOP)
The husbandry and housekeeping
measures available to control pests
The level of risk to the business /
organisation and environment if
suitable controls and management
systems are in place.
Define ALOP for site and
demonstrate that the
level of risk is acceptable
Monitoring
linked to the pest risk
assessment (unmitigated risk)
for specific pests –
demonstrates effectiveness of
controls and management
systems
(3) Pest Risk Management
Note – the term ‘Pests’ includes diseases
and ‘infest’ includes ‘infect’](https://image.slidesharecdn.com/helenandamandaplanthealthy-200114095824/85/Helen-Bentley-Fox-Amanda-Calvert-13-Nov-19-15-320.jpg)