Haworth Structures of Monosaccharides
•Download as PPTX, PDF•
10 likes•10,875 views
The Haworth structures of pentose and hexose sugars are five- or six-membered rings. These cyclic structures are produced through an intramolecular reaction between a carbonyl group and hydroxyl group on the same monosaccharide molecule. Common examples are the α- and β-anomers of D-glucose. Monosaccharides can be oxidized to form sugar acids by oxidizing the aldehyde group. They can also be reduced to form sugar alcohols by reducing the carbonyl group. Fructose can rearrange and oxidize to form glucose. Sugar testing uses Benedict's reagent to detect glucose in the urine, indicating diabetes.
Report
Share
Report
Share
Recommended
General Introduction and classification of Carbohydrates 

This presentation is made for F.Y.Bsc. Students.
The presentation includes the General Properties of Carbohydrate and the classification of carbohydrates.
Disaccharides

Disaccharides are double sugars that yield two simple sugars called monosaccharides upon hydrolysis. The three main disaccharides are sucrose, maltose, and lactose. They differ in their solubility, with sucrose being very soluble, maltose fairly soluble, and lactose only slightly soluble. Disaccharides are formed through a dehydration synthesis reaction combining two monosaccharides. Their structures depend on the type of glycosidic linkage between the monosaccharides. This determines their properties such as whether they are reducing sugars or able to undergo fermentation.
Monosaccharides

This document discusses carbohydrates and monosaccharides. It defines carbohydrates as compounds composed of carbon, hydrogen, and oxygen. Monosaccharides are the simplest form of carbohydrates and include trioses, tetroses, pentoses, and hexoses. The document discusses various properties of monosaccharides including isomerism, anomerism, mutarotation, and common chemical reactions like oxidation, reduction, and reactions with acids and bases. It also summarizes important derivatives of monosaccharides such as amino sugars, deoxy sugars, sugar acids, sugar alcohols, esters, and glycosides.
Fischer projections of monosaccharides

These slides would help to understand the chiral carbon atoms, L-and D- configurations in monosaccharides
DISACCHARIDES

A BRIEF PRESENTATION ON DISACCHARIDES.
NOTE- FONTS MAY APPEAR WEIRD BCOZ THE FONTS I USED DO NOT APPEAR IN THIS PPT.
Monosaccharides

Monosaccharides are simple sugars that cannot be further broken down. They are categorized by the number of carbons they contain and whether they have an aldehyde or ketone functional group. Monosaccharides can exist as different isomers depending on the spatial arrangement of their atoms. Some types of isomerism in monosaccharides include stereoisomers, enantiomers, epimers, anomers, and pyranose-furanose isomers.
Disaccharides

Disaccharides are composed of two monosaccharides joined by an O-glycosidic linkage. The main disaccharides discussed are:
1) Sucrose (table sugar), which hydrolyzes into glucose and fructose. Inversion of sucrose produces invert sugar, which is sweeter.
2) Maltose, formed from two glucose molecules and is a reducing sugar. It is found in germinating seeds.
3) Lactose is the sugar in milk, formed from glucose and galactose. It is hydrolyzed by lactase in the small intestine.
Sugar derivatives and reactions of monosaccharides

Reactions of monosaccharides, osazone formation, reduction, oxidation, reaction with acids and alkalies, ester formation and formation of amino sugars, amino sugar acids and deoxy sugars.
Recommended
General Introduction and classification of Carbohydrates 

This presentation is made for F.Y.Bsc. Students.
The presentation includes the General Properties of Carbohydrate and the classification of carbohydrates.
Disaccharides

Disaccharides are double sugars that yield two simple sugars called monosaccharides upon hydrolysis. The three main disaccharides are sucrose, maltose, and lactose. They differ in their solubility, with sucrose being very soluble, maltose fairly soluble, and lactose only slightly soluble. Disaccharides are formed through a dehydration synthesis reaction combining two monosaccharides. Their structures depend on the type of glycosidic linkage between the monosaccharides. This determines their properties such as whether they are reducing sugars or able to undergo fermentation.
Monosaccharides

This document discusses carbohydrates and monosaccharides. It defines carbohydrates as compounds composed of carbon, hydrogen, and oxygen. Monosaccharides are the simplest form of carbohydrates and include trioses, tetroses, pentoses, and hexoses. The document discusses various properties of monosaccharides including isomerism, anomerism, mutarotation, and common chemical reactions like oxidation, reduction, and reactions with acids and bases. It also summarizes important derivatives of monosaccharides such as amino sugars, deoxy sugars, sugar acids, sugar alcohols, esters, and glycosides.
Fischer projections of monosaccharides

These slides would help to understand the chiral carbon atoms, L-and D- configurations in monosaccharides
DISACCHARIDES

A BRIEF PRESENTATION ON DISACCHARIDES.
NOTE- FONTS MAY APPEAR WEIRD BCOZ THE FONTS I USED DO NOT APPEAR IN THIS PPT.
Monosaccharides

Monosaccharides are simple sugars that cannot be further broken down. They are categorized by the number of carbons they contain and whether they have an aldehyde or ketone functional group. Monosaccharides can exist as different isomers depending on the spatial arrangement of their atoms. Some types of isomerism in monosaccharides include stereoisomers, enantiomers, epimers, anomers, and pyranose-furanose isomers.
Disaccharides

Disaccharides are composed of two monosaccharides joined by an O-glycosidic linkage. The main disaccharides discussed are:
1) Sucrose (table sugar), which hydrolyzes into glucose and fructose. Inversion of sucrose produces invert sugar, which is sweeter.
2) Maltose, formed from two glucose molecules and is a reducing sugar. It is found in germinating seeds.
3) Lactose is the sugar in milk, formed from glucose and galactose. It is hydrolyzed by lactase in the small intestine.
Sugar derivatives and reactions of monosaccharides

Reactions of monosaccharides, osazone formation, reduction, oxidation, reaction with acids and alkalies, ester formation and formation of amino sugars, amino sugar acids and deoxy sugars.
Carbohydrates 

Carbohydrates are sugars that provide energy when consumed. Our bodies break down carbohydrates into glucose, which is the primary energy source. Carbohydrates are classified based on their structure from simple to complex: monosaccharides like glucose and fructose are the simplest; disaccharides like sucrose and lactose contain two monosaccharide units; and polysaccharides like starch contain many monosaccharide units joined by glycosidic bonds. Common tests are used to identify and characterize different carbohydrates.
Polysaccharide

Polysaccharide introduction, example, structure, starch, cellulose, chitin those structure and important functions and their presence in plants and animals, polysaccharide types based on functions and their composition , functions of polysaccharides , important images for relevant polysaccharides types, polysaccharide role in plants and animal cells. Starch - structure and functions, cellulose structure and functions, chitin - structure and functions
Physical properties of Carbohydrates

The document discusses the physical properties of carbohydrates including monosaccharides, disaccharides, and polysaccharides. Monosaccharides are the simplest sugars and include glucose, fructose, and galactose. Disaccharides are formed when two monosaccharides join together, such as sucrose, lactose, and maltose. Polysaccharides have long chains of monosaccharide units and include starch, glycogen, cellulose, and chitin. The properties of carbohydrates differ based on their size, from monosaccharides being the smallest and most soluble to polysaccharides having more complex structures and being less soluble.
Oligosaccharides

This document provides information about various carbohydrates including monosaccharides, oligosaccharides, and polysaccharides. It discusses the structures and properties of common disaccharides like maltose, lactose, and sucrose. Larger carbohydrates covered include maltodextrins, dextrans, inulin, chitin, cellulose, starch, and glycogen. For each carbohydrate, details are given about its source, structure, properties when tested, and digestive breakdown. The document aims to describe the chemistry of many important carbohydrates.
Carbohydrate structure

This document discusses carbohydrate chemistry. It defines carbohydrates as organic molecules found in nature that are made up of carbon, hydrogen, and oxygen. Carbohydrates are classified based on their structure into monosaccharides, disaccharides, oligosaccharides, and polysaccharides. Monosaccharides include important hexoses like glucose and fructose. Carbohydrates exhibit properties like optical activity and stereoisomerism due to asymmetric carbon atoms. Key topics covered include glycosidic linkages, reducing sugars, ring structure forms (pyranose and furanose), and epimers. Physiologically important monosaccharides and derived sugars are also mentioned.
Carbohydrates

The present topic focus on carbohydrates which will be beneficial for pharma as well as science graduate students
Biochemistry of carbohydrates_prepared_by_Drx_Raju_Yadav_2021

Carbohydrates, or carbs, are sugar molecules. Along with proteins and fats, carbohydrates are one of three main nutrients found in foods and drinks. Your body breaks down carbohydrates into glucose. Glucose, or blood sugar, is the main source of energy for your body's cells, tissues, and organs
Polysaccharides types and Structural Features

1. The document discusses types and structural features of polysaccharides. It describes homopolysaccharides like starch, dextrin, inulin, glycogen, and cellulose.
2. Starch is composed of amylose and amylopectin subunits linked by alpha-glucosidic bonds. Dextrin is formed from starch hydrolysis and has a similar structure to amylopectin.
3. Inulin is a polymer of fructose typically with a terminal glucose. Glycogen stores glucose in animals and has highly branched alpha-linked subunits. Cellulose, the main component of plant cell walls, is composed of beta-glucose units.
Polysaccharides

Polysaccharides are complex monosaccharide polymers that serve a wide variety of functions. They can be classified as homopolymers containing a single monosaccharide unit or heteropolymers containing different sugar units. Starch is a major plant polysaccharide composed of amylose and amylopectin. It is used as food, in pharmaceuticals, and to produce dextrins and soluble starch. Dextrins are prepared from starch by partial hydrolysis and are used as substitutes for gums. Cyclodextrins are obtained from starch and have a hydrophobic central cavity, making them useful for enclosing drugs.
Biochemistry lecture notes carbohydrates

This document provides information about carbohydrates. It begins by defining carbohydrates and describing their main biological functions. It then discusses the three main classes of carbohydrates: monosaccharides, disaccharides, and polysaccharides. For each class, key examples are provided and their structures and properties are explained. The document also covers topics like stereochemistry of carbohydrates, glycosaminoglycans, and important monosaccharides and polysaccharides like starch, cellulose, and glycogen. In summary, it serves as a comprehensive overview of carbohydrate structure, classification, and functions in biological systems.
Cholesterol Biosynthesis

This document summarizes the biosynthesis of cholesterol in 5 steps:
1) Mevalonate is formed from acetyl-CoA in the cytosol. 2) Isoprenoid units are formed from mevalonate. 3) Six isoprenoid units condense to form squalene. 4) Squalene is cyclized to form lanosterol. 5) Lanosterol is modified through a series of changes to ultimately form cholesterol in the endoplasmic reticulum. Cholesterol biosynthesis is a major regulatory point for cholesterol levels and is the target of statin drugs.
Complex Lipids (Phosholipids, Glycolipids and Lipoproteins) (Chemistry of Lip...

This document discusses different types of phospholipids and their structures and functions. It notes that phospholipids are made up of fatty acids, glycerol, phosphoric acid, and a nitrogenous base. They are major constituents of cell membranes and are amphipathic in nature. The document describes different classes of phospholipids, including phosphatidylcholines, phosphatidylethanolamines, phosphatidylserines, cardiolipins, sphingomyelins, and phosphatidylinositols. It also discusses glycolipids and lipoproteins, providing details on their compositions and roles in cells and tissues.
Fructose 

This document provides information about various sugars and carbohydrates. It discusses fructose, a simple sugar found in plants that is absorbed directly into the blood. Sucrose, or table sugar, is introduced as a disaccharide made of glucose and fructose. Starch is described as a glucan made of glucose molecules linked together in plants. The document also examines the structures of fructose, sucrose, starch, and cellulose, including their cyclic formations and glycosidic linkages.
Lipids properties, classification, function

This document defines and describes various types of lipids. It begins by explaining that lipids are a heterogeneous group of compounds related to fatty acids, fats, oils, waxes and other substances. It then discusses the basic components of lipids like fatty acids, glycerol and their esters known as triglycerides. The document further classifies lipids into simple lipids, compound lipids and derived lipids. Various types of phospholipids, glycolipids, sterols and terpenoids are also explained. Physical and chemical properties of lipids are outlined along with their important functions in living organisms.
CARBOHYDRATE CHEMISTRY

- Plants convert 100 metric tons of CO2 into carbohydrates each year through photosynthesis.
- Carbohydrates are the most abundant organic molecules and serve important functions like energy storage, structure, and encoding biologic information through oligosaccharide chains.
- Monosaccharides can exist as cyclic or linear structures and take on different configurations that impact their chemical and physical properties. Common techniques like mutarotation, osazone formation, and oxidation reactions are used to characterize carbohydrates.
Monosaccharides(2)

Monosaccharides are simple sugars that serve as building blocks for other carbohydrates. They include pentoses with 5 carbons like ribose and hexoses with 6 carbons like glucose and fructose. Glucose and fructose are isomers with the same formula but different structures. Monosaccharides can exist as linear molecules or in ring forms like furanoses. They undergo various reactions including oxidation, reduction, and fermentation.
Glycolipid ppt

Glycolipids Presented by students of BS Microbiology Batch 1st Indus International Institute D.G.Khan (Punjab) Pakistan.
Biosynthesis of glycerides, phospholipids and cholesterol

This document summarizes the biosynthesis of glycerides, phospholipids, and cholesterol. It states that glycerides are the major lipids in the body and are esters of glycerol and fatty acids. Phospholipids are the main constituents of biological membranes and contain a phosphate group. Cholesterol is needed by the body but in moderation, as unhealthy levels can lead to high blood cholesterol. The document then goes on to describe the enzymatic biosynthesis processes of each compound in more detail through multiple steps.
corbohydrates chemistry structure of isomerism

• The compounds possessing identical
molecular formula but different structures are
called isomers.
Various types of isomerism
1. Structural isomerism
2. Stereoisomerism
Stereoisomerism
• Same molecular formula and same structure
but they differ in configuration.
• That is arrangement of their atoms in space.
• Presence of asymmetric carbon atoms allow
the formation of stereoisomerism
Carbohydrates

Carbohydrates are polyhydroxy aldehydes or ketones or compounds derived from their hydrolysis.
includes- Definition, classification, examples, enantiomers, epimers, anomers, D and L isomers, ozasone testing, reducing and non reducing sugars, chemical tests and disease.
Carbohydrates - the carbohydrates are optically active polyhydroxy aldehydes/...

the carbohydrates are optically active polyhydroxy aldehydes/ ketones or the compounds which produce such units on hydrolysis.
Chem 1123 unit 7a

Carbohydrates are a major source of energy and are composed of carbon, hydrogen, and oxygen. There are three main types of carbohydrates: monosaccharides, disaccharides, and polysaccharides. Monosaccharides include glucose and fructose, which can form cyclic structures in solution. Disaccharides are formed from two monosaccharides and include maltose, lactose, and sucrose. Polysaccharides are polymers of monosaccharides and starch is a polymer of glucose that provides long-term energy storage.
More Related Content
What's hot
Carbohydrates 

Carbohydrates are sugars that provide energy when consumed. Our bodies break down carbohydrates into glucose, which is the primary energy source. Carbohydrates are classified based on their structure from simple to complex: monosaccharides like glucose and fructose are the simplest; disaccharides like sucrose and lactose contain two monosaccharide units; and polysaccharides like starch contain many monosaccharide units joined by glycosidic bonds. Common tests are used to identify and characterize different carbohydrates.
Polysaccharide

Polysaccharide introduction, example, structure, starch, cellulose, chitin those structure and important functions and their presence in plants and animals, polysaccharide types based on functions and their composition , functions of polysaccharides , important images for relevant polysaccharides types, polysaccharide role in plants and animal cells. Starch - structure and functions, cellulose structure and functions, chitin - structure and functions
Physical properties of Carbohydrates

The document discusses the physical properties of carbohydrates including monosaccharides, disaccharides, and polysaccharides. Monosaccharides are the simplest sugars and include glucose, fructose, and galactose. Disaccharides are formed when two monosaccharides join together, such as sucrose, lactose, and maltose. Polysaccharides have long chains of monosaccharide units and include starch, glycogen, cellulose, and chitin. The properties of carbohydrates differ based on their size, from monosaccharides being the smallest and most soluble to polysaccharides having more complex structures and being less soluble.
Oligosaccharides

This document provides information about various carbohydrates including monosaccharides, oligosaccharides, and polysaccharides. It discusses the structures and properties of common disaccharides like maltose, lactose, and sucrose. Larger carbohydrates covered include maltodextrins, dextrans, inulin, chitin, cellulose, starch, and glycogen. For each carbohydrate, details are given about its source, structure, properties when tested, and digestive breakdown. The document aims to describe the chemistry of many important carbohydrates.
Carbohydrate structure

This document discusses carbohydrate chemistry. It defines carbohydrates as organic molecules found in nature that are made up of carbon, hydrogen, and oxygen. Carbohydrates are classified based on their structure into monosaccharides, disaccharides, oligosaccharides, and polysaccharides. Monosaccharides include important hexoses like glucose and fructose. Carbohydrates exhibit properties like optical activity and stereoisomerism due to asymmetric carbon atoms. Key topics covered include glycosidic linkages, reducing sugars, ring structure forms (pyranose and furanose), and epimers. Physiologically important monosaccharides and derived sugars are also mentioned.
Carbohydrates

The present topic focus on carbohydrates which will be beneficial for pharma as well as science graduate students
Biochemistry of carbohydrates_prepared_by_Drx_Raju_Yadav_2021

Carbohydrates, or carbs, are sugar molecules. Along with proteins and fats, carbohydrates are one of three main nutrients found in foods and drinks. Your body breaks down carbohydrates into glucose. Glucose, or blood sugar, is the main source of energy for your body's cells, tissues, and organs
Polysaccharides types and Structural Features

1. The document discusses types and structural features of polysaccharides. It describes homopolysaccharides like starch, dextrin, inulin, glycogen, and cellulose.
2. Starch is composed of amylose and amylopectin subunits linked by alpha-glucosidic bonds. Dextrin is formed from starch hydrolysis and has a similar structure to amylopectin.
3. Inulin is a polymer of fructose typically with a terminal glucose. Glycogen stores glucose in animals and has highly branched alpha-linked subunits. Cellulose, the main component of plant cell walls, is composed of beta-glucose units.
Polysaccharides

Polysaccharides are complex monosaccharide polymers that serve a wide variety of functions. They can be classified as homopolymers containing a single monosaccharide unit or heteropolymers containing different sugar units. Starch is a major plant polysaccharide composed of amylose and amylopectin. It is used as food, in pharmaceuticals, and to produce dextrins and soluble starch. Dextrins are prepared from starch by partial hydrolysis and are used as substitutes for gums. Cyclodextrins are obtained from starch and have a hydrophobic central cavity, making them useful for enclosing drugs.
Biochemistry lecture notes carbohydrates

This document provides information about carbohydrates. It begins by defining carbohydrates and describing their main biological functions. It then discusses the three main classes of carbohydrates: monosaccharides, disaccharides, and polysaccharides. For each class, key examples are provided and their structures and properties are explained. The document also covers topics like stereochemistry of carbohydrates, glycosaminoglycans, and important monosaccharides and polysaccharides like starch, cellulose, and glycogen. In summary, it serves as a comprehensive overview of carbohydrate structure, classification, and functions in biological systems.
Cholesterol Biosynthesis

This document summarizes the biosynthesis of cholesterol in 5 steps:
1) Mevalonate is formed from acetyl-CoA in the cytosol. 2) Isoprenoid units are formed from mevalonate. 3) Six isoprenoid units condense to form squalene. 4) Squalene is cyclized to form lanosterol. 5) Lanosterol is modified through a series of changes to ultimately form cholesterol in the endoplasmic reticulum. Cholesterol biosynthesis is a major regulatory point for cholesterol levels and is the target of statin drugs.
Complex Lipids (Phosholipids, Glycolipids and Lipoproteins) (Chemistry of Lip...

This document discusses different types of phospholipids and their structures and functions. It notes that phospholipids are made up of fatty acids, glycerol, phosphoric acid, and a nitrogenous base. They are major constituents of cell membranes and are amphipathic in nature. The document describes different classes of phospholipids, including phosphatidylcholines, phosphatidylethanolamines, phosphatidylserines, cardiolipins, sphingomyelins, and phosphatidylinositols. It also discusses glycolipids and lipoproteins, providing details on their compositions and roles in cells and tissues.
Fructose 

This document provides information about various sugars and carbohydrates. It discusses fructose, a simple sugar found in plants that is absorbed directly into the blood. Sucrose, or table sugar, is introduced as a disaccharide made of glucose and fructose. Starch is described as a glucan made of glucose molecules linked together in plants. The document also examines the structures of fructose, sucrose, starch, and cellulose, including their cyclic formations and glycosidic linkages.
Lipids properties, classification, function

This document defines and describes various types of lipids. It begins by explaining that lipids are a heterogeneous group of compounds related to fatty acids, fats, oils, waxes and other substances. It then discusses the basic components of lipids like fatty acids, glycerol and their esters known as triglycerides. The document further classifies lipids into simple lipids, compound lipids and derived lipids. Various types of phospholipids, glycolipids, sterols and terpenoids are also explained. Physical and chemical properties of lipids are outlined along with their important functions in living organisms.
CARBOHYDRATE CHEMISTRY

- Plants convert 100 metric tons of CO2 into carbohydrates each year through photosynthesis.
- Carbohydrates are the most abundant organic molecules and serve important functions like energy storage, structure, and encoding biologic information through oligosaccharide chains.
- Monosaccharides can exist as cyclic or linear structures and take on different configurations that impact their chemical and physical properties. Common techniques like mutarotation, osazone formation, and oxidation reactions are used to characterize carbohydrates.
Monosaccharides(2)

Monosaccharides are simple sugars that serve as building blocks for other carbohydrates. They include pentoses with 5 carbons like ribose and hexoses with 6 carbons like glucose and fructose. Glucose and fructose are isomers with the same formula but different structures. Monosaccharides can exist as linear molecules or in ring forms like furanoses. They undergo various reactions including oxidation, reduction, and fermentation.
Glycolipid ppt

Glycolipids Presented by students of BS Microbiology Batch 1st Indus International Institute D.G.Khan (Punjab) Pakistan.
Biosynthesis of glycerides, phospholipids and cholesterol

This document summarizes the biosynthesis of glycerides, phospholipids, and cholesterol. It states that glycerides are the major lipids in the body and are esters of glycerol and fatty acids. Phospholipids are the main constituents of biological membranes and contain a phosphate group. Cholesterol is needed by the body but in moderation, as unhealthy levels can lead to high blood cholesterol. The document then goes on to describe the enzymatic biosynthesis processes of each compound in more detail through multiple steps.
corbohydrates chemistry structure of isomerism

• The compounds possessing identical
molecular formula but different structures are
called isomers.
Various types of isomerism
1. Structural isomerism
2. Stereoisomerism
Stereoisomerism
• Same molecular formula and same structure
but they differ in configuration.
• That is arrangement of their atoms in space.
• Presence of asymmetric carbon atoms allow
the formation of stereoisomerism
Carbohydrates

Carbohydrates are polyhydroxy aldehydes or ketones or compounds derived from their hydrolysis.
includes- Definition, classification, examples, enantiomers, epimers, anomers, D and L isomers, ozasone testing, reducing and non reducing sugars, chemical tests and disease.
What's hot (20)
Biochemistry of carbohydrates_prepared_by_Drx_Raju_Yadav_2021

Biochemistry of carbohydrates_prepared_by_Drx_Raju_Yadav_2021
Complex Lipids (Phosholipids, Glycolipids and Lipoproteins) (Chemistry of Lip...

Complex Lipids (Phosholipids, Glycolipids and Lipoproteins) (Chemistry of Lip...
Biosynthesis of glycerides, phospholipids and cholesterol

Biosynthesis of glycerides, phospholipids and cholesterol
Similar to Haworth Structures of Monosaccharides
Carbohydrates - the carbohydrates are optically active polyhydroxy aldehydes/...

the carbohydrates are optically active polyhydroxy aldehydes/ ketones or the compounds which produce such units on hydrolysis.
Chem 1123 unit 7a

Carbohydrates are a major source of energy and are composed of carbon, hydrogen, and oxygen. There are three main types of carbohydrates: monosaccharides, disaccharides, and polysaccharides. Monosaccharides include glucose and fructose, which can form cyclic structures in solution. Disaccharides are formed from two monosaccharides and include maltose, lactose, and sucrose. Polysaccharides are polymers of monosaccharides and starch is a polymer of glucose that provides long-term energy storage.
chapter 2 carbohydrates.ppt

This document provides an outline and overview of carbohydrate chemistry. It begins by acknowledging various universities that contribute to carbohydrate research. The learning objectives are to describe carbohydrate properties, explain digestion and metabolism of carbohydrates. The document then covers the classes of carbohydrates including monosaccharides, oligosaccharides, and polysaccharides. Key aspects of carbohydrate chemistry such as isomerism, reactions, and tests are discussed. Carbohydrate structure, properties, and functions are examined in detail.
Biomolecules.pptx

Carbohydrates are classified based on their structure and behavior during hydrolysis. Monosaccharides like glucose and fructose can further cyclize to form rings. Glucose forms a 6-membered pyranose ring while fructose forms a 5-membered furanose ring. Carbohydrates can also be classified as reducing or non-reducing based on whether their functional groups are free to participate in reduction reactions. Glucose and fructose both exist as alpha and beta cyclic isomers differentiated by the orientation of their hydroxyl group. Characteristic reactions and inability to explain properties with an open chain form indicate carbohydrates exist predominantly in ring structures.
Biomolecules.pptx

Carbohydrates are classified based on their structure and behavior during hydrolysis. Monosaccharides like glucose and fructose can further cyclize to form rings. Glucose forms a 6-membered pyranose ring while fructose forms a 5-membered furanose ring. Carbohydrates can also be classified as reducing or non-reducing based on whether their functional groups are free to participate in reduction reactions. Glucose and fructose both exist as cyclic structures with α and β anomers formed by the addition of the hydroxyl group to the carbonyl carbon. Their open chain forms do not fully explain properties like crystallization behavior and lack of reactivity of functional groups.
Introduction to carbohydrates

- Carbohydrates provide energy and are composed of carbon, hydrogen, and oxygen. Glucose is a primary carbohydrate that our bodies use for energy.
- Carbohydrates exist as monosaccharides, disaccharides, and polysaccharides. Monosaccharides like glucose cannot be broken down further. Disaccharides contain two monosaccharide units joined by a glycosidic bond. Polysaccharides contain long chains of monosaccharide units.
- Examples of monosaccharides are glucose, fructose, and galactose. Disaccharides include sucrose, lactose, and maltose. Starch, glycogen, and cellulose are examples of polysaccharides that provide energy storage or structural support
CHO-CNP .ppt

Carbohydrates can be classified as sugars, oligosaccharides, or polysaccharides based on their number of sugar units. Monosaccharides exist as both open-chain and cyclic forms when in solution. Cyclization leads to the formation of hemiacetals and hemiketals, with monosaccharides having five or more carbons existing primarily as five- or six-membered rings. The cyclic forms can interconvert between alpha and beta anomers. Monosaccharides undergo various reactions including reduction, oxidation, and formation of derivatives like glycosides.
Biochemistry of Carbohydrates

This document provides information about carbohydrates. It discusses that carbohydrates are the most abundant organic molecules in nature and an important source of energy for cells. Carbohydrates can also act as structural components and be involved in cell membranes, surface antigens, and extracellular substances. The document further describes different types of carbohydrates including monosaccharides, disaccharides, and polysaccharides. It provides examples and characteristics of important carbohydrates such as glucose, fructose, sucrose, lactose, and glycogen. Reaction and derivatives of monosaccharides are also summarized.
Carbohydrates - Monosaccharides and its qualitative tests - Part 1

Discusses about monosaccharides definition, classification, structure and reactions of glucose, galactose, and fructose. Qualitative tests for carbohydrates with reaction scheme. Terminologies in carbohydrates such as epimeris, anomers and mutarotation.
Carbohydrates

Carbohydrates are the most abundant organic compounds in plants. They act as storehouses of chemical energy and components of supportive structures. There are four main types of carbohydrates: monosaccharides, disaccharides, oligosaccharides, and polysaccharides. Monosaccharides include glucose, fructose, and galactose. Disaccharides such as sucrose, lactose, and maltose are formed from two monosaccharide units. Polysaccharides including starch, glycogen, and cellulose are long chains of monosaccharide units and serve as energy storage. Carbohydrates undergo various reactions including formation of glycosides, esters, and reduction to alcohols.
Carbohydrates

Carbohydrates are the most abundant organic compounds in plants. They act as storehouses of chemical energy and components of supportive structures. There are four main types of carbohydrates: monosaccharides, disaccharides, oligosaccharides, and polysaccharides. Monosaccharides include glucose, fructose, and galactose. Disaccharides such as sucrose, lactose, and maltose are formed from two monosaccharide units. Polysaccharides including starch, cellulose, and glycogen are long chains of monosaccharide units that provide structural support and energy storage. Carbohydrates undergo various reactions including formation of glycosides, esters, and reductions to alcohols.
The nature of carbohydrates 

Carbohydrates are organic compounds made of carbon, hydrogen, and oxygen. They serve as an important energy source and structural component. There are three main types of carbohydrates: monosaccharides (simple sugars), disaccharides, and polysaccharides. Glucose is a common monosaccharide that exists as both an open chain and ring structure. Carbohydrates undergo chemical reactions like oxidation, reduction, and esterification. They also exhibit mutarotation when dissolved in water.
Lecture notes on Chemistry of carbohydrates

Carbohydrates are an important class of biological molecules that serve as energy sources and structural components. They are classified based on their structure as monosaccharides, disaccharides, oligosaccharides, or polysaccharides. Monosaccharides like glucose are the simplest units and cannot be further broken down. Disaccharides form when two monosaccharides join, such as sucrose from glucose and fructose or maltose from two glucose units. Polysaccharides are long chains of monosaccharide units and provide structure and energy storage. Carbohydrates play key roles in biology through their various forms and reactions.
Chemistry of carbohydrates and isomerism

carbohydrates with their brief introduction and their classification. then structural features of carbohydrates
Lecture7-Carbohydrates101.pptx

Carbohydrates are organic compounds made of carbon, hydrogen, and oxygen. They include sugars (monosaccharides and disaccharides) and starch (polysaccharides). Monosaccharides are single sugars that cannot be broken down further. Common monosaccharides include glucose, fructose, and galactose. Disaccharides are formed when two monosaccharides bond together, such as sucrose, maltose, and lactose. Polysaccharides are long chains of monosaccharides and include starch, cellulose, and glycogen. Plants and animals use carbohydrates for energy storage and structural support.
PBS-FCH-322._L1._Carbohydrates_Chemistry.pdf

Synthesis of carbohydrates
Classification of carbohydrates
Classification of monosaccharides
Structural formulas
Chemistry of carbohydrates

This document provides information on carbohydrates including their definition, classification, structures, and biological importance. It begins by defining carbohydrates and discussing their classification into monosaccharides, oligosaccharides, and polysaccharides based on the number of sugar units. Important monosaccharides like glucose, fructose, and galactose are described along with their reactions and structural aspects. Disaccharides such as sucrose, lactose, and maltose are also discussed. The document then covers polysaccharides including starch, glycogen, and mucopolysaccharides; and concludes by emphasizing the key roles and functions of carbohydrates in biological systems.
Food chemistry Carbohydrates presentation.ppt

This document provides an overview of carbohydrate classification and structure. It begins by classifying carbohydrates as monosaccharides, disaccharides, or polysaccharides based on whether they hydrolyze into single sugars, two sugars, or many sugars. Key monosaccharides like glucose, fructose, and galactose are described. Cyclic ring structures of monosaccharides are explained. Important polysaccharides such as starch, glycogen, and cellulose composed of glucose monomers linked by glycosidic bonds are also summarized.
carbohydrates ppt-1.pptx

Carbohydrates are polyhydroxy aldehydes or ketones that yield these derivatives upon hydrolysis. They are classified as monosaccharides, disaccharides, oligosaccharides, or polysaccharides depending on the number of monosaccharide units. Monosaccharides include glucose, fructose and galactose. Disaccharides are formed from two monosaccharide units and include sucrose, lactose and maltose. Polysaccharides like starch, glycogen and cellulose are made of many glucose units and act as energy stores. Carbohydrates provide energy, aid digestion, and support brain function.
Similar to Haworth Structures of Monosaccharides (20)
Carbohydrates - the carbohydrates are optically active polyhydroxy aldehydes/...

Carbohydrates - the carbohydrates are optically active polyhydroxy aldehydes/...
3, 4. isomerism, Chemical properties of monosaccharides.pptx

3, 4. isomerism, Chemical properties of monosaccharides.pptx
Carbohydrates - Monosaccharides and its qualitative tests - Part 1

Carbohydrates - Monosaccharides and its qualitative tests - Part 1
More from Vsachdev
Conclusion

Carbohydrates are the main source of energy for living things and are made of carbon, hydrogen, and oxygen. They include sugars, starches, and fiber and provide 4 calories of energy per gram. Carbohydrates are found in foods like fruits, vegetables, grains, legumes, and dairy products.
Polysaccharides

Cellulose is composed of glucose units connected by β-1,4-glycosidic bonds. It is a polysaccharide that forms the structural component of plant cell walls and is insoluble in water.
Chemical properties of monosaccharides

Chemical properties would help to understand the products of oxidation or reduction of monosaccharides.
Disaccharides

Disaccharides are carbohydrates formed from two monosaccharides bonded together. The three most common disaccharides are maltose, lactose, and sucrose. Maltose contains two glucose molecules bonded with an alpha-1,4 linkage. Lactose contains glucose and galactose with a beta-1,4 linkage. Sucrose contains glucose and fructose with an alpha-1,2 linkage. These disaccharides differ in their monosaccharide components and bond linkages.
Chiral molecules

This document discusses chiral and achiral molecules. It defines chiral molecules as those with nonsuperimposable mirror images containing a chiral carbon atom bonded to four different groups. Achiral molecules have mirror images that are superimposable. The document provides examples of chiral and achiral carbon atoms and discusses how only one enantiomer of chiral compounds is often biologically active in organisms due to interactions with chiral receptors and enzymes.
Carbohydrates

Paula, a diabetes nurse, teaches Kate to test her blood glucose levels before and after meals. Paula explains that Kate's pre-meal number should be 110 mg/dL or less, and if it increases by more than 50 mg/dL after eating, she needs to lower her carbohydrate intake.
More from Vsachdev (6)
Recently uploaded
The History of Stoke Newington Street Names

Presented at the Stoke Newington Literary Festival on 9th June 2024
www.StokeNewingtonHistory.com
Advanced Java[Extra Concepts, Not Difficult].docx![Advanced Java[Extra Concepts, Not Difficult].docx](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![Advanced Java[Extra Concepts, Not Difficult].docx](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
This is part 2 of my Java Learning Journey. This contains Hashing, ArrayList, LinkedList, Date and Time Classes, Calendar Class and more.
The Diamonds of 2023-2024 in the IGRA collection

A review of the growth of the Israel Genealogy Research Association Database Collection for the last 12 months. Our collection is now passed the 3 million mark and still growing. See which archives have contributed the most. See the different types of records we have, and which years have had records added. You can also see what we have for the future.
South African Journal of Science: Writing with integrity workshop (2024)

South African Journal of Science: Writing with integrity workshop (2024)Academy of Science of South Africa
A workshop hosted by the South African Journal of Science aimed at postgraduate students and early career researchers with little or no experience in writing and publishing journal articles.How to Make a Field Mandatory in Odoo 17

In Odoo, making a field required can be done through both Python code and XML views. When you set the required attribute to True in Python code, it makes the field required across all views where it's used. Conversely, when you set the required attribute in XML views, it makes the field required only in the context of that particular view.
Hindi varnamala | hindi alphabet PPT.pdf

हिंदी वर्णमाला पीपीटी, hindi alphabet PPT presentation, hindi varnamala PPT, Hindi Varnamala pdf, हिंदी स्वर, हिंदी व्यंजन, sikhiye hindi varnmala, dr. mulla adam ali, hindi language and literature, hindi alphabet with drawing, hindi alphabet pdf, hindi varnamala for childrens, hindi language, hindi varnamala practice for kids, https://www.drmullaadamali.com
Community pharmacy- Social and preventive pharmacy UNIT 5

Covered community pharmacy topic of the subject Social and preventive pharmacy for Diploma and Bachelor of pharmacy
BBR 2024 Summer Sessions Interview Training

Qualitative research interview training by Professor Katrina Pritchard and Dr Helen Williams
What is Digital Literacy? A guest blog from Andy McLaughlin, University of Ab...

What is Digital Literacy? A guest blog from Andy McLaughlin, University of Aberdeen
Walmart Business+ and Spark Good for Nonprofits.pdf

"Learn about all the ways Walmart supports nonprofit organizations.
You will hear from Liz Willett, the Head of Nonprofits, and hear about what Walmart is doing to help nonprofits, including Walmart Business and Spark Good. Walmart Business+ is a new offer for nonprofits that offers discounts and also streamlines nonprofits order and expense tracking, saving time and money.
The webinar may also give some examples on how nonprofits can best leverage Walmart Business+.
The event will cover the following::
Walmart Business + (https://business.walmart.com/plus) is a new shopping experience for nonprofits, schools, and local business customers that connects an exclusive online shopping experience to stores. Benefits include free delivery and shipping, a 'Spend Analytics” feature, special discounts, deals and tax-exempt shopping.
Special TechSoup offer for a free 180 days membership, and up to $150 in discounts on eligible orders.
Spark Good (walmart.com/sparkgood) is a charitable platform that enables nonprofits to receive donations directly from customers and associates.
Answers about how you can do more with Walmart!"
RPMS TEMPLATE FOR SCHOOL YEAR 2023-2024 FOR TEACHER 1 TO TEACHER 3

RPMS Template 2023-2024 by: Irene S. Rueco
ISO/IEC 27001, ISO/IEC 42001, and GDPR: Best Practices for Implementation and...

Denis is a dynamic and results-driven Chief Information Officer (CIO) with a distinguished career spanning information systems analysis and technical project management. With a proven track record of spearheading the design and delivery of cutting-edge Information Management solutions, he has consistently elevated business operations, streamlined reporting functions, and maximized process efficiency.
Certified as an ISO/IEC 27001: Information Security Management Systems (ISMS) Lead Implementer, Data Protection Officer, and Cyber Risks Analyst, Denis brings a heightened focus on data security, privacy, and cyber resilience to every endeavor.
His expertise extends across a diverse spectrum of reporting, database, and web development applications, underpinned by an exceptional grasp of data storage and virtualization technologies. His proficiency in application testing, database administration, and data cleansing ensures seamless execution of complex projects.
What sets Denis apart is his comprehensive understanding of Business and Systems Analysis technologies, honed through involvement in all phases of the Software Development Lifecycle (SDLC). From meticulous requirements gathering to precise analysis, innovative design, rigorous development, thorough testing, and successful implementation, he has consistently delivered exceptional results.
Throughout his career, he has taken on multifaceted roles, from leading technical project management teams to owning solutions that drive operational excellence. His conscientious and proactive approach is unwavering, whether he is working independently or collaboratively within a team. His ability to connect with colleagues on a personal level underscores his commitment to fostering a harmonious and productive workplace environment.
Date: May 29, 2024
Tags: Information Security, ISO/IEC 27001, ISO/IEC 42001, Artificial Intelligence, GDPR
-------------------------------------------------------------------------------
Find out more about ISO training and certification services
Training: ISO/IEC 27001 Information Security Management System - EN | PECB
ISO/IEC 42001 Artificial Intelligence Management System - EN | PECB
General Data Protection Regulation (GDPR) - Training Courses - EN | PECB
Webinars: https://pecb.com/webinars
Article: https://pecb.com/article
-------------------------------------------------------------------------------
For more information about PECB:
Website: https://pecb.com/
LinkedIn: https://www.linkedin.com/company/pecb/
Facebook: https://www.facebook.com/PECBInternational/
Slideshare: http://www.slideshare.net/PECBCERTIFICATION
Your Skill Boost Masterclass: Strategies for Effective Upskilling

Your Skill Boost Masterclass: Strategies for Effective UpskillingExcellence Foundation for South Sudan
Strategies for Effective Upskilling is a presentation by Chinwendu Peace in a Your Skill Boost Masterclass organisation by the Excellence Foundation for South Sudan on 08th and 09th June 2024 from 1 PM to 3 PM on each day.Chapter 4 - Islamic Financial Institutions in Malaysia.pptx

Chapter 4 - Islamic Financial Institutions in Malaysia.pptxMohd Adib Abd Muin, Senior Lecturer at Universiti Utara Malaysia
This slide is special for master students (MIBS & MIFB) in UUM. Also useful for readers who are interested in the topic of contemporary Islamic banking.
Recently uploaded (20)
South African Journal of Science: Writing with integrity workshop (2024)

South African Journal of Science: Writing with integrity workshop (2024)
Pride Month Slides 2024 David Douglas School District

Pride Month Slides 2024 David Douglas School District
Community pharmacy- Social and preventive pharmacy UNIT 5

Community pharmacy- Social and preventive pharmacy UNIT 5
What is Digital Literacy? A guest blog from Andy McLaughlin, University of Ab...

What is Digital Literacy? A guest blog from Andy McLaughlin, University of Ab...
Walmart Business+ and Spark Good for Nonprofits.pdf

Walmart Business+ and Spark Good for Nonprofits.pdf
RPMS TEMPLATE FOR SCHOOL YEAR 2023-2024 FOR TEACHER 1 TO TEACHER 3

RPMS TEMPLATE FOR SCHOOL YEAR 2023-2024 FOR TEACHER 1 TO TEACHER 3
ISO/IEC 27001, ISO/IEC 42001, and GDPR: Best Practices for Implementation and...

ISO/IEC 27001, ISO/IEC 42001, and GDPR: Best Practices for Implementation and...
Your Skill Boost Masterclass: Strategies for Effective Upskilling

Your Skill Boost Masterclass: Strategies for Effective Upskilling
Chapter 4 - Islamic Financial Institutions in Malaysia.pptx

Chapter 4 - Islamic Financial Institutions in Malaysia.pptx
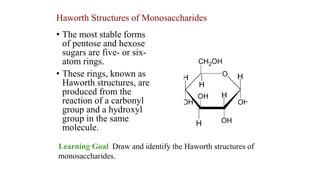
Haworth Structures of Monosaccharides
- 1. • The most stable forms of pentose and hexose sugars are five- or six- atom rings. • These rings, known as Haworth structures, are produced from the reaction of a carbonyl group and a hydroxyl group in the same molecule. Haworth Structures of Monosaccharides Learning Goal Draw and identify the Haworth structures of monosaccharides.
- 2. Guide for Drawing Hayworth Structures
- 3. Cyclic Structure for Glucose STEP 1 Turn the Fischer projection clockwise by 90°.
- 4. STEP 2 Fold clockwise to make a hexagon and bond the O on carbon 5 to carbon 1. • Place the carbon 6 group in the — CH2OH group above carbon 5. • Draw a bond between the oxygen of the — OH group on carbon 5 to the carbonyl carbon.
- 5. Cyclic Structure for Glucose STEP 3 Draw the new — OH group on carbon 1 below the ring to give the α isomer or above the ring to give the β isomer.
- 6. Mutarotation - and β-D-Glucose When placed in solution, • cyclic structures open and close • -D-glucose converts to β-D-glucose and vice versa • at any time, only a small amount of open chain forms
- 7. Haworth Structures of Fructose Fructose • is a ketohexose • forms a five-atom ring structure with carbon 2 at the right corner of the ring • forms when the — OH group on carbon 5 reacts with carbon 2 in the carbonyl group
- 8. Study Check Write the cyclic forms of D-galactose.
- 9. Solution STEP 1 Turn the Fischer projection clockwise by 90°. 1 2 3 4 5 6 6 5 4 3 2 1 C OH H H OH H OH OH H HOH2C O H
- 10. STEP 2 Fold clockwise to make a hexagon and bond the O on carbon 5 to the carbonyl group. • Place the carbon 6 group above the ring. • Write the — OH group on carbon 2 below the ring and the — OH groups on carbon 3 and carbon 4 above the ring. C HO H H OH H OH OH H CH2OH O H
- 11. Solution -D-Galactose -D-Galactose STEP 3 Draw the new — OH group on carbon 1 below the ring to give the α isomer or above the ring to give the isomer.
- 12. 13.5 Chemical Properties of Monosaccharides Sugar alcohols such as D- sorbitol, D-xylitol from D-xylose, and D-mannitol from D-mannose are used as sweeteners in many sugar-free products such as diet drinks and sugarless gum as well as products for people with diabetes. Learning Goal Identify the products of oxidation or reduction of monosaccharides; determine whether a carbohydrate is a reducing sugar.
- 13. Oxidation of Monosaccharides Monosaccharides in solution • have small amounts of the open-chain form present • have an aldehyde group with an adjacent hydroxyl group that can be oxidized to carboxylic acid by an oxidizing agent such as Benedict’s
- 14. Oxidation of Monosaccharides Sugar acids • are produced from the oxidation of the aldehyde form as Cu2+ is reduced to Cu+ • are named by replacing the ose ending of the monosaccharide with onic acid A carbohydrate that reduces another substance (such as the open chain form of D-glucose) is called a reducing sugar.
- 15. Oxidation: Fructose to Glucose Fructose, a ketohexose, • contains a ketone group, which usually can’t be oxidized • can be oxidized in a basic Benedict’s solution when a rearrangement occurs between the ketone group on carbon 2 and the hydroxyl group on carbon 1 • is then converted to glucose, which produces an aldehyde group with an adjacent hydroxyl that can be oxidized
- 16. Oxidation: Fructose to Glucose Fructose, a ketohexose, rearranges to form glucose and is then oxidized in Benedict’s.
- 17. Reduction of Monosaccharides The reduction of the carbonyl group in monosaccharides • produces sugar alcohols, which are also called alditols • converts D-glucose to the sugar alcohol D-sorbitol
- 18. Reducing Sugars The sugar alcohols • are named by replacing the ose ending of the monosaccharide with itol • include D-sorbitol, D-xylitol from D-xylose, and D- mannitol from D-mannose • are used as sweeteners in many sugar-free products such as diet drinks and sugarless gum
- 19. Study Check Write the product of the reduction of D-mannose. D-Mannose
- 20. Solution Write the product of the reduction of D-mannose. D-Mannose D-Mannitol Reduction
- 21. Chemistry Link to Health: Glucose Testing • Normally, blood glucose flows through the kidneys and is reabsorbed into the bloodstream. • When the blood level exceeds about 160 mg of glucose/dL of blood, the kidneys cannot reabsorb all of the glucose, and it spills over into the urine, a condition known as glucosuria.
- 22. Chemistry Link to Health: Glucose Testing • A symptom of diabetes mellitus is a high level of glucose in the urine, which can be identified using Benedict’s test.