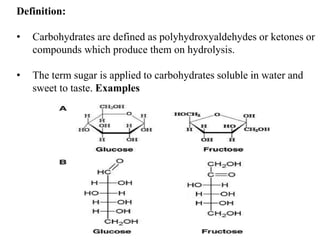
The document provides an overview of carbohydrates, detailing their structure, classification, and functions. It explains the different types of carbohydrates including monosaccharides, oligosaccharides, and polysaccharides, along with their properties, chemical reactions, and significance in biological systems. Additional topics covered include stereoisomerism, optical activity, and the formation of various carbohydrate derivatives.