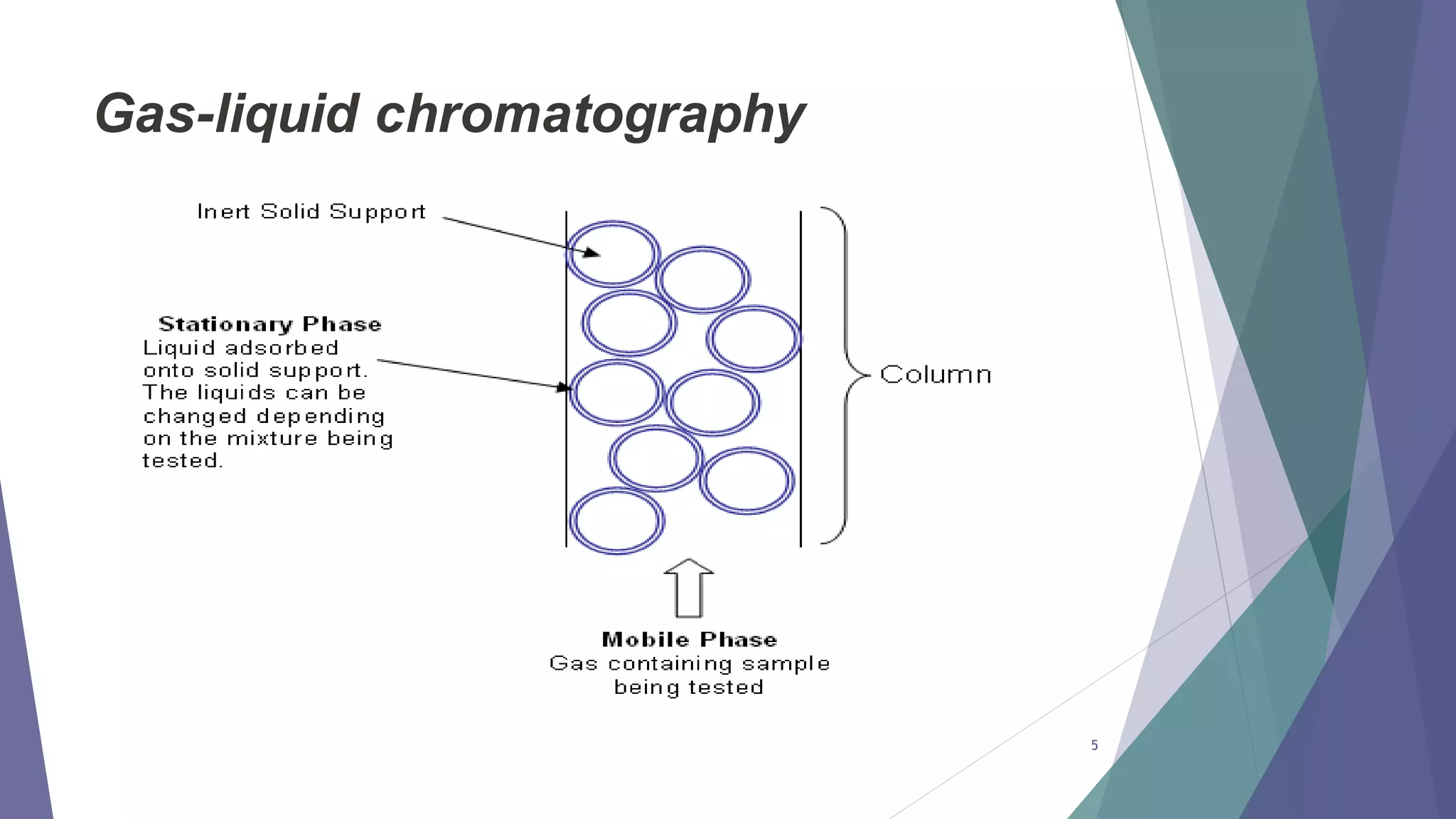
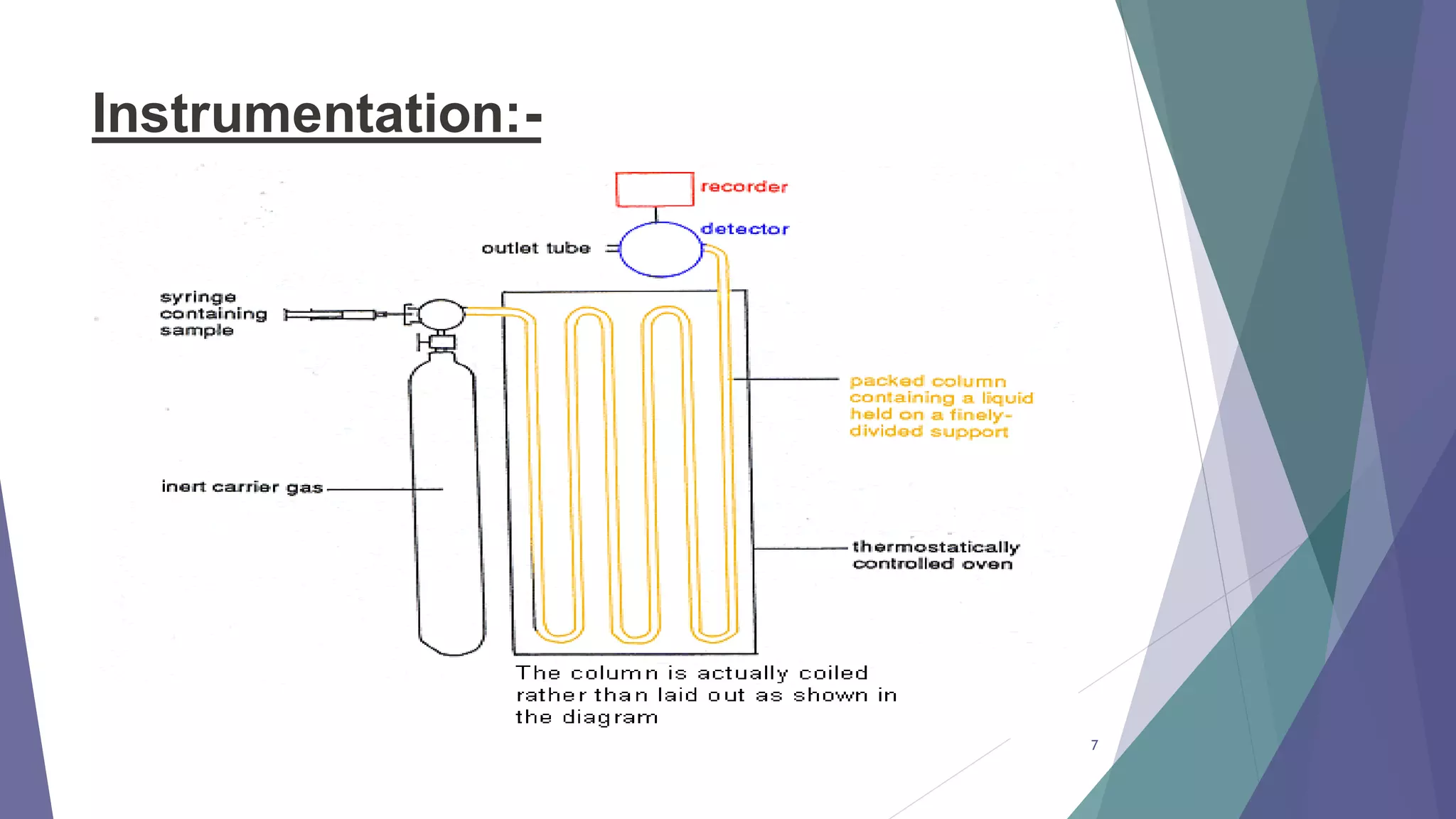
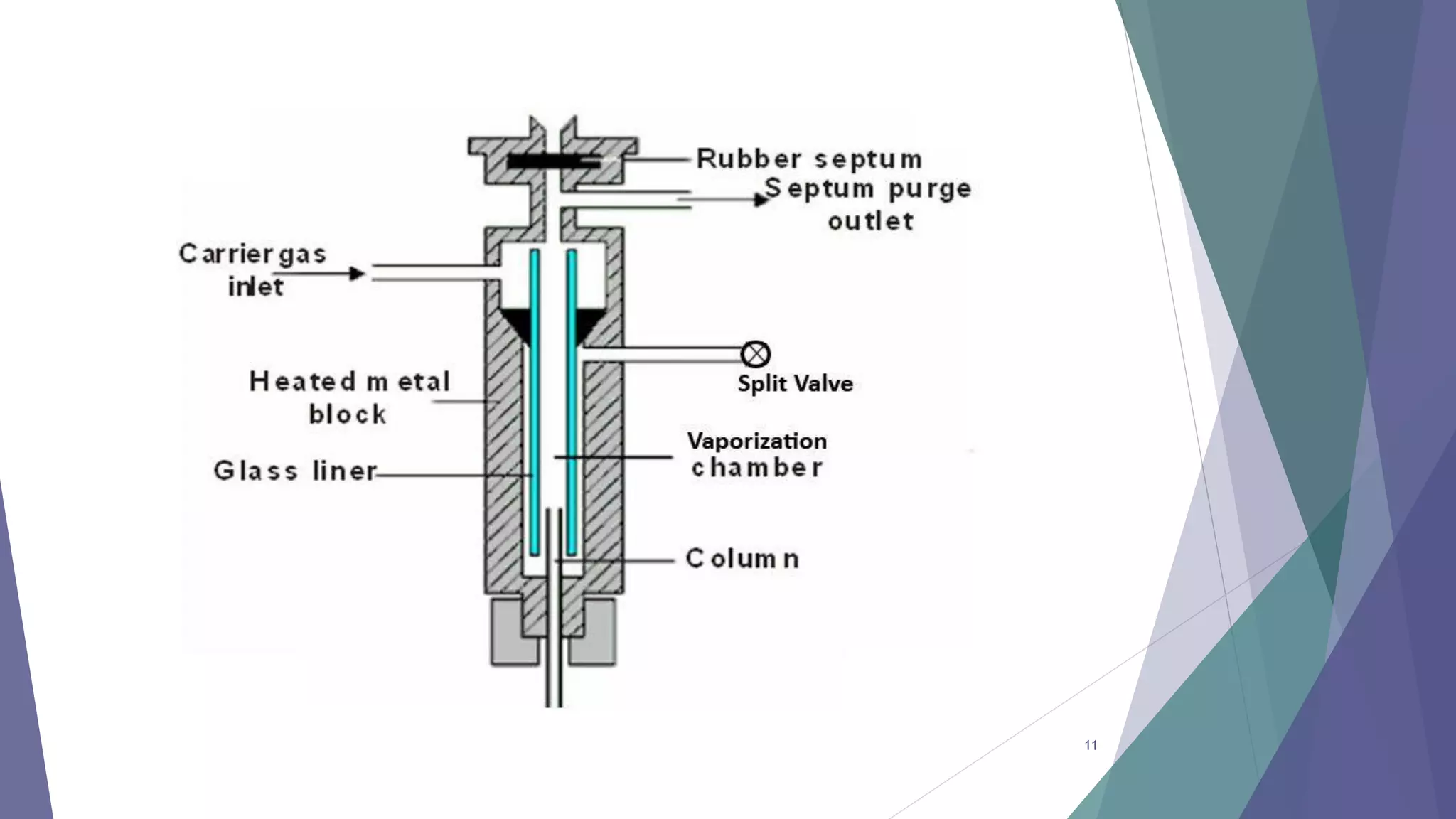
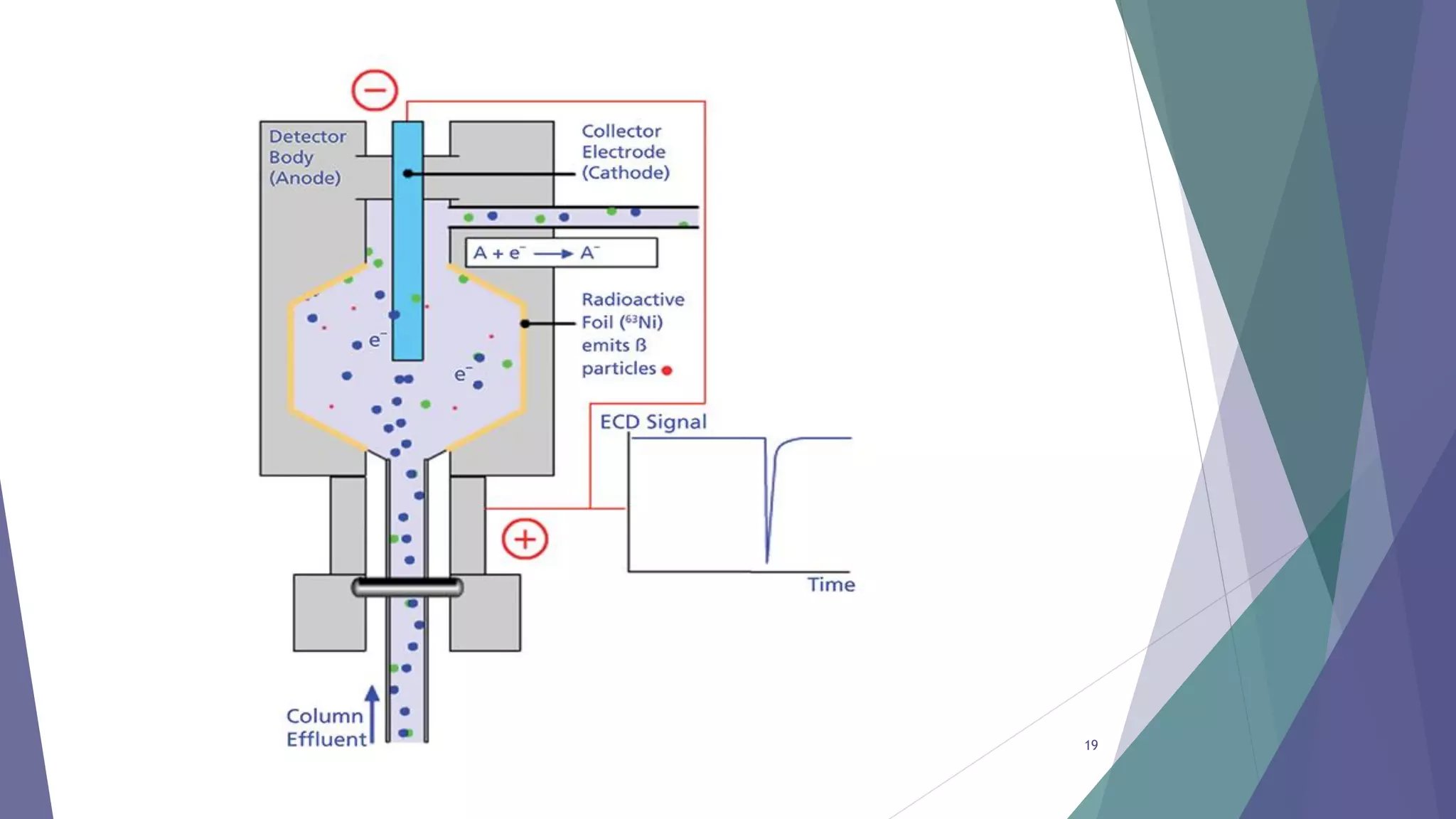
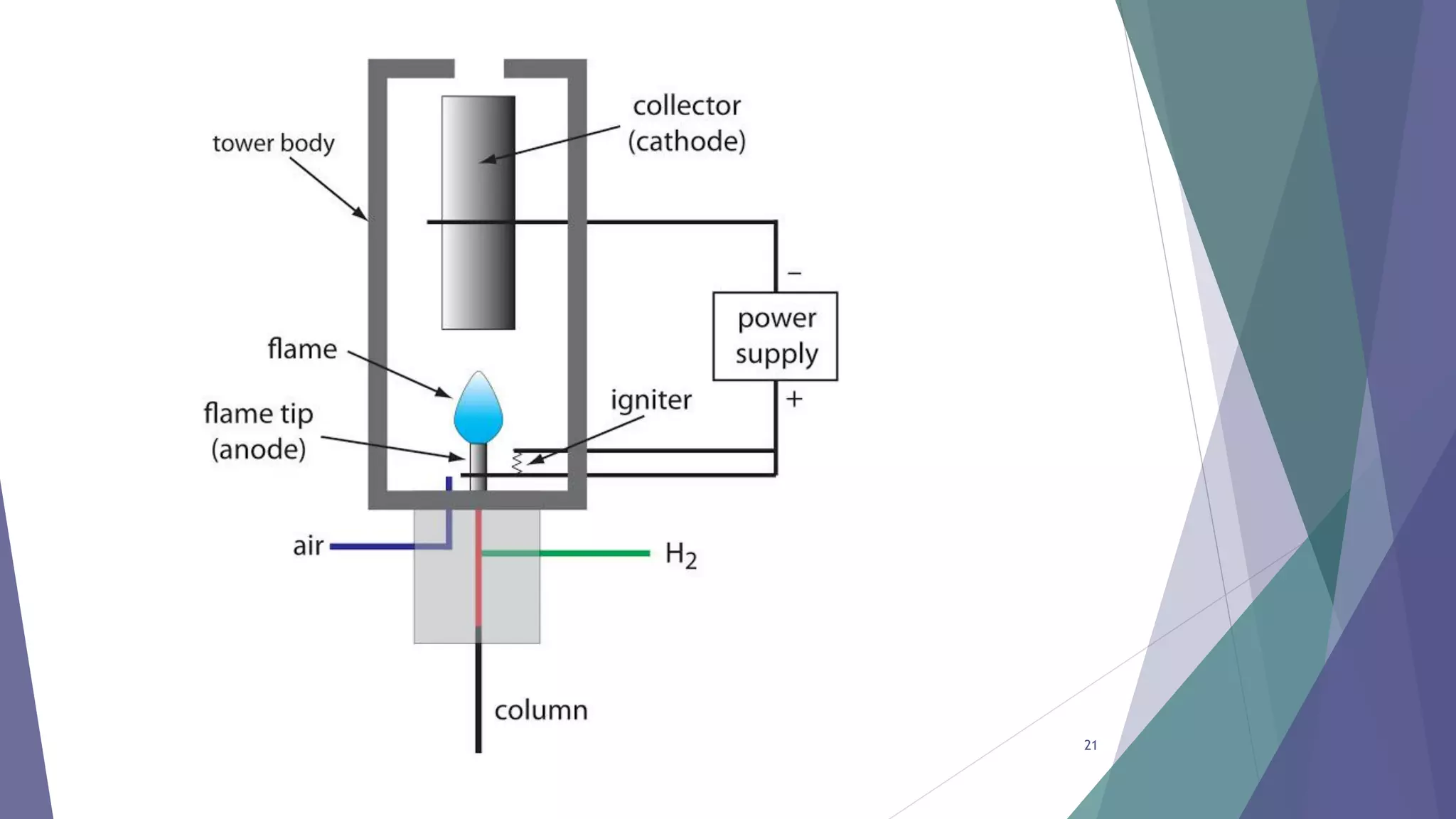
This document discusses gas chromatography, including its principles, instrumentation, and applications. Gas chromatography is a process that separates components in a mixture using an inert gas as the mobile phase. There are two main types - gas-solid chromatography uses a gas mobile phase and solid stationary phase, while gas-liquid chromatography uses a gas mobile phase and liquid stationary phase coated on an inert solid support. Key components of a gas chromatography instrument include the carrier gas, sample injection port, columns, and detectors such as the flame ionization detector. Gas chromatography has applications in quality control and analysis of drugs and metabolites.