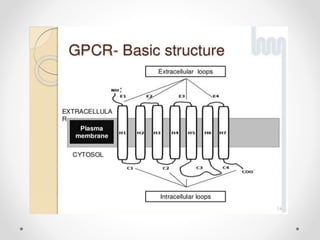
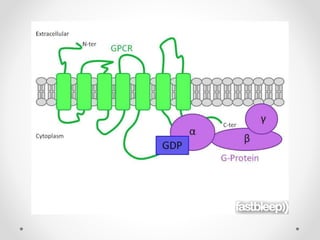
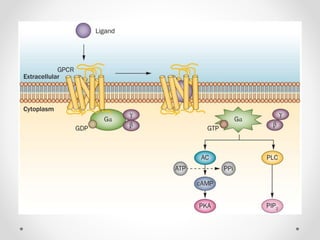
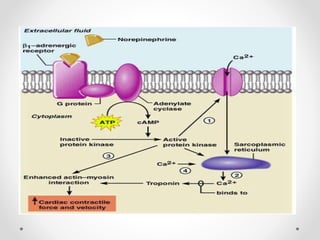
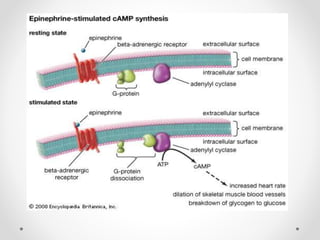
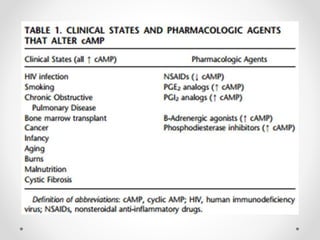
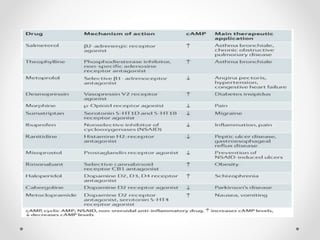
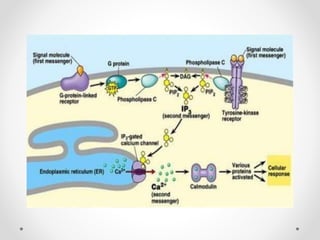
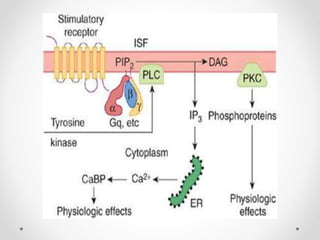
G protein-coupled receptors (GPCRs) are a large family of transmembrane receptors that sense molecules outside the cell and activate intracellular signal transduction pathways. They have seven transmembrane domains and transmit signals by coupling to heterotrimeric G proteins on the inner cell surface. When an agonist binds to a GPCR, it causes a conformational change that activates the G protein, starting intracellular signaling cascades through second messengers like cAMP or IP3. Approximately half of all drugs target GPCRs, making them an important drug discovery area.