The document discusses India's Food Safety and Standards (Labelling and Display) Regulations, 2020. Some key points:
- The new regulations aim to make national labelling policies more robust and effective by separating packaging, labelling and advertising requirements into different regulations.
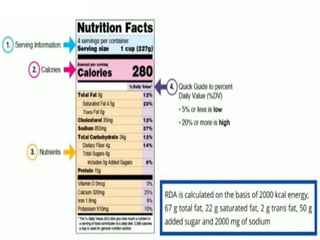
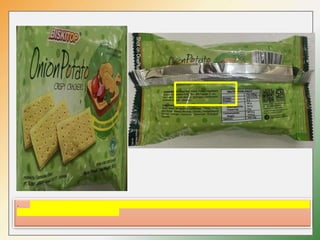
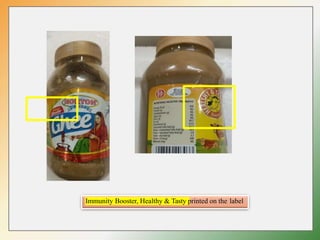
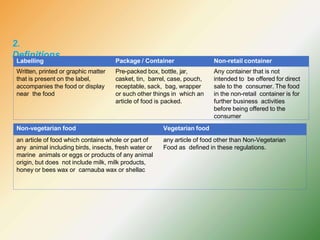
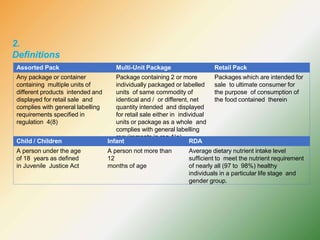
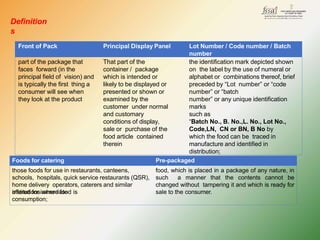
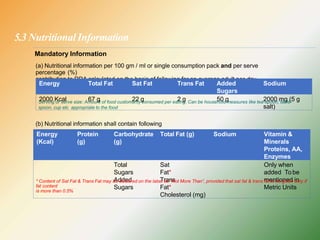
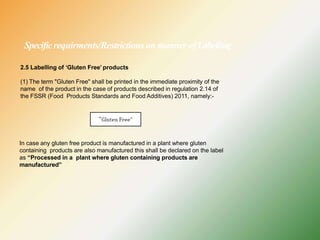
- Key labelling requirements include declaring ingredients, nutritional information, allergens, expiry dates, manufacturer details, and logos indicating if a product is vegetarian or non-vegetarian.
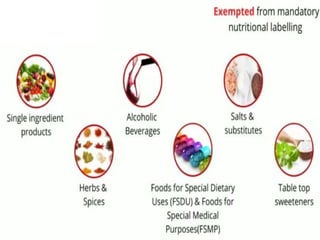
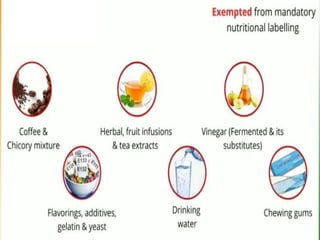
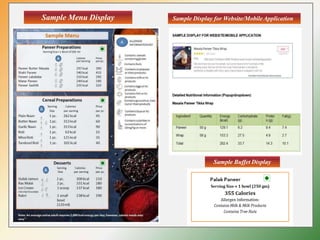
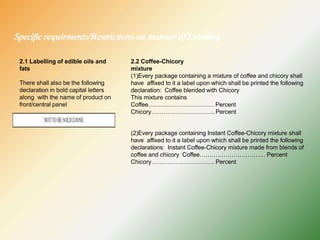
- Specific labelling is required for products containing additives like colors, sweeteners, MSG, etc. Menu labelling in restaurants is also addressed.
- The regulations seek to provide consumers with more information to make informed choices about food products