Exam 2 Naming for Oxyanion Series, Ionic/Covalent Compounds, Binary acids/Oxyacids
•Download as DOCX, PDF•
1 like•1,295 views
This packet goes in order with PP2. Fill out all of the examples and then go back to the front and do the worksheet to see if you retained any of the information.
Report
Share
Report
Share
Recommended
Reactions Of Metals And Metal Compounds

The document discusses chemical reactions involving metals and their compounds. It explains that metals react with acids to produce salts and hydrogen gas. It also discusses how metals react with oxygen and carbon dioxide to form metal oxides and metal carbonates, which can then further react with acids. The document provides examples of word equations for common metal-acid reactions and identifies the products and reactants in various chemical changes involving metals and their compounds.
Chapter 4 carbon and its compounds

crstrareti et4sttugdfdsrsytuffwesythjrsrtrughdsutyufesugdrer7itujgfs45e76rtiugjhfgdre5r67tiujhgfxsr4e57586iykhjghgfsr4e578ihjggfxdse5r6t7uykhjcfdert56u7yhjgfdrtyhgvbcfdrtyghvbcfdrthjbnvcfdtyhjbvcxdftghbvcxdfghvbcxdfrtghvcxdfrtghvcxdrtghvfdrtghvcdsasedfgthyujikaertyuioasdfghjklasdfghjklzxcvbnm,asdfghjkqwertyuiopasdfghjkzxcvbnmasdfghjklqwertyuiop
Empirical and molecular formulas

The document discusses empirical and molecular formulas. It defines molecular formulas as showing the exact number of each type of atom in a compound, while empirical formulas show the simplest whole number ratio of elements in a compound. It provides examples of molecular and empirical formulas being the same or different. The document also provides steps for calculating empirical formulas from elemental composition percentages and determining the molecular formula from the empirical formula and molar mass.
Types of chemical formula

Chemical formulas represent compounds by showing the types and numbers of atoms present. There are two main types of chemical formulas: empirical formulas and molecular formulas. Empirical formulas show the simplest whole number ratio of atoms in a compound, such as CH2O for glucose. Molecular formulas show the actual number and types of atoms in a molecule, such as C6H12O6 for glucose. The molecular formula can be a multiple of the empirical formula, such as how C6H12O6 is a multiple of CH2O for glucose. Some compounds, like HCl and H2O, have the same empirical and molecular formulas.
Insoluble salts

This document discusses insoluble salts. It defines insoluble salts as salts that cannot dissolve in water at room temperature. It explains that insoluble salts can be prepared through precipitation reactions or double decomposition reactions by mixing aqueous solutions containing the cations and anions that will form the insoluble salt. This causes the ions to interchange and form an insoluble precipitate. Examples of insoluble salts formed and their ionic equations are also provided.
Valence Electrons Worksheet

This document is a worksheet about valence electrons that contains questions asking students to:
1) Identify the number of electrons in various elements.
2) Draw Lewis structures and identify the number of outer shell electrons for different elements.
3) Identify the number of valence electrons elements have by filling in their electron configurations.
Chemical Formula

The document discusses chemical formulas and how they are derived. It explains that chemical formulas show the number and type of atoms in a molecule using element symbols. It then provides examples of how to determine formulas using valency, including transition metals that can have multiple valencies indicated by Roman numerals. Prefixes in compound names can also indicate the number of atoms present.
Solubility and mixtures.pptx

This document provides an overview of solutions and related concepts in chemistry. It defines key terms like mixtures, solutions, solutes, solvents, concentration, solubility, and factors that affect solubility. It also discusses homogeneous and heterogeneous mixtures, concentration in terms of molarity and percent by mass, and how to perform calculations involving dilution of solutions and solution stoichiometry. The document uses examples and diagrams to illustrate these concepts.
Recommended
Reactions Of Metals And Metal Compounds

The document discusses chemical reactions involving metals and their compounds. It explains that metals react with acids to produce salts and hydrogen gas. It also discusses how metals react with oxygen and carbon dioxide to form metal oxides and metal carbonates, which can then further react with acids. The document provides examples of word equations for common metal-acid reactions and identifies the products and reactants in various chemical changes involving metals and their compounds.
Chapter 4 carbon and its compounds

crstrareti et4sttugdfdsrsytuffwesythjrsrtrughdsutyufesugdrer7itujgfs45e76rtiugjhfgdre5r67tiujhgfxsr4e57586iykhjghgfsr4e578ihjggfxdse5r6t7uykhjcfdert56u7yhjgfdrtyhgvbcfdrtyghvbcfdrthjbnvcfdtyhjbvcxdftghbvcxdfghvbcxdfrtghvcxdfrtghvcxdrtghvfdrtghvcdsasedfgthyujikaertyuioasdfghjklasdfghjklzxcvbnm,asdfghjkqwertyuiopasdfghjkzxcvbnmasdfghjklqwertyuiop
Empirical and molecular formulas

The document discusses empirical and molecular formulas. It defines molecular formulas as showing the exact number of each type of atom in a compound, while empirical formulas show the simplest whole number ratio of elements in a compound. It provides examples of molecular and empirical formulas being the same or different. The document also provides steps for calculating empirical formulas from elemental composition percentages and determining the molecular formula from the empirical formula and molar mass.
Types of chemical formula

Chemical formulas represent compounds by showing the types and numbers of atoms present. There are two main types of chemical formulas: empirical formulas and molecular formulas. Empirical formulas show the simplest whole number ratio of atoms in a compound, such as CH2O for glucose. Molecular formulas show the actual number and types of atoms in a molecule, such as C6H12O6 for glucose. The molecular formula can be a multiple of the empirical formula, such as how C6H12O6 is a multiple of CH2O for glucose. Some compounds, like HCl and H2O, have the same empirical and molecular formulas.
Insoluble salts

This document discusses insoluble salts. It defines insoluble salts as salts that cannot dissolve in water at room temperature. It explains that insoluble salts can be prepared through precipitation reactions or double decomposition reactions by mixing aqueous solutions containing the cations and anions that will form the insoluble salt. This causes the ions to interchange and form an insoluble precipitate. Examples of insoluble salts formed and their ionic equations are also provided.
Valence Electrons Worksheet

This document is a worksheet about valence electrons that contains questions asking students to:
1) Identify the number of electrons in various elements.
2) Draw Lewis structures and identify the number of outer shell electrons for different elements.
3) Identify the number of valence electrons elements have by filling in their electron configurations.
Chemical Formula

The document discusses chemical formulas and how they are derived. It explains that chemical formulas show the number and type of atoms in a molecule using element symbols. It then provides examples of how to determine formulas using valency, including transition metals that can have multiple valencies indicated by Roman numerals. Prefixes in compound names can also indicate the number of atoms present.
Solubility and mixtures.pptx

This document provides an overview of solutions and related concepts in chemistry. It defines key terms like mixtures, solutions, solutes, solvents, concentration, solubility, and factors that affect solubility. It also discusses homogeneous and heterogeneous mixtures, concentration in terms of molarity and percent by mass, and how to perform calculations involving dilution of solutions and solution stoichiometry. The document uses examples and diagrams to illustrate these concepts.
Chpt 7 part 1 - chemical nomenclature 042403

The document provides an overview of chemical nomenclature and naming conventions for compounds. It discusses the need for a systematic naming system to 1) prevent confusion between compounds, 2) avoid complex formulas, and 3) group similar compounds into families. It then covers naming conventions for binary compounds containing two nonmetals, binary compounds containing a metal and nonmetal, and compounds named using the "ic/ous" method involving metal oxidation states. The goal is to establish a logical and consistent system for naming chemical compounds based on their formulas and elemental components.
Ch. 3 stoichiometry

This document provides an overview of stoichiometry concepts in chemistry. It defines stoichiometry as calculations with chemical formulas and equations. Key concepts covered include the law of conservation of mass, types of chemical reactions, anatomy of chemical equations, formula weights and molecular weights, moles, mole ratios, empirical formulas, combustion analysis, stoichiometric calculations, limiting reactants, theoretical yield, and percent yield.
Chemical properties

Chemical properties describe how a substance reacts or changes during a chemical reaction. Some key chemical properties include flammability, reactivity, and the ability to undergo chemical changes that alter the composition of the substance. Common signs that a chemical change has occurred include a change in color, production of a gas, or formation of a precipitate. However, physical changes can sometimes exhibit similar signs, so the only definitive way to identify a chemical change is if the composition of the substance changes to form new substances.
2.6.1 oxidation numbers

This document discusses oxidation and reduction reactions and provides rules for determining oxidation numbers:
1) Oxidation involves loss of electrons and increases oxidation number, while reduction involves gain of electrons and decreases oxidation number.
2) The sum of oxidation numbers in a molecule or ion must equal the overall charge.
3) Transition metals can have variable oxidation numbers depending on their compounds. Their oxidation states are included in names.
4) Some anomalies may occur where oxidation numbers are fractional or elements appear to gain/lose no electrons.
CALCULATIONS WITH CHEMICAL FORMULAS AND EQUATION

Calculate the empirical formula of vitamin C from the
given data.
MOLE; Avogadro's Number; Percentage Composition

The document defines key chemistry concepts related to moles, including:
- A mole refers to Avogadro's number (6.02x10^23) of particles like atoms, molecules, ions or formula units.
- 1 mole of an element contains 6.02x10^23 atoms, 1 mole of a molecular compound contains 6.02x10^23 molecules, and 1 mole of an ionic compound contains 6.02x10^23 formula units.
- Gram atomic mass refers to the mass of one mole of an element in grams, and gram formula mass refers to the sum of atomic weights that make up one mole of a compound.
- Chemical formulas represent the
Naming compounds

The document provides an overview of naming chemical compounds. It discusses the rules for naming binary ionic compounds that contain metals and nonmetals (types I and II), binary covalent compounds between two nonmetals (type III), compounds containing polyatomic ions, and acids. Examples of applying the naming conventions are provided throughout. The overall strategies are to consider whether the compounds are ionic or covalent, identify cations and anions or elemental names, and apply prefixes or suffixes accordingly when arriving at the systematic name.
Simple techniques of separating mixtures

This document provides information on techniques for separating mixtures. It discusses several methods including sedimentation and decantation, filtration, evaporation, distillation, crystallization, sublimation, chromatography, centrifugation and solvent extraction. The document is intended for a 9th grade science class and aims to teach students how to select the appropriate separation method based on the components of a mixture. It includes links to videos demonstrating each technique.
Writing and Naming formula

This document discusses writing and naming chemical formulas and ions. It begins by explaining that cations are positively charged ions that migrate to the cathode, while anions are negatively charged ions that migrate to the anode. Examples of common cations and anions are provided. The document then discusses rules for writing formulas for ionic compounds without transition metals, ionic compounds with transition metals, and covalent compounds. It also covers naming conventions for polyatomic ions and provides examples of writing formulas from names and names from formulas.
Molarity Molality Dilutions

This document discusses concentration of solutions in chemistry. It defines molarity as moles of solute per liter of solution. It provides an example problem calculating the molarity of a solution made by dissolving 5 grams of nickel chloride hexahydrate in 250 mL of water. It also discusses molality, defined as moles of solute per kilogram of solvent, and provides an example problem calculating the molality of a sodium chloride solution. Finally, it discusses the process of diluting solutions through calculations that conserve the number of moles while changing the volume.
The mole concept

This document discusses moles, molar mass, and Avogadro's number. It explains that a mole is the amount of a substance that contains 6.022x1023 particles, known as Avogadro's number. It also defines molar mass as the mass in grams of one mole of a substance. The document provides examples of calculating molar mass from atomic masses and using molar mass to determine the number of moles or particles in a given mass of a substance.
Empirical and molecular formula class 11

The document discusses empirical and molecular formulas. An empirical formula shows the simplest whole number ratio of atoms in a compound, while a molecular formula shows the exact number of each atom. To calculate molecular formula from empirical formula: 1) make a table with elements, percentages, atomic masses, moles, and simplest ratios; 2) the empirical formula mass is the sum of the atoms' masses; 3) divide the molar mass by the empirical formula mass to get the common factor for the molecular formula. Two examples are given to calculate empirical and molecular formulas from percentage compositions and molar masses.
Air and water

The document provides information about various chemistry concepts related to air and water:
- It describes chemical tests to identify water and the purification of water supplies through filtration and chlorination.
- The composition of clean air is described as 78% nitrogen, 21% oxygen and small quantities of other gases. Common air pollutants like carbon monoxide and their sources are stated.
- Fractional distillation is outlined as the process used to separate oxygen and nitrogen from liquid air based on their different boiling points.
- Rusting is described as a reaction between iron, air and water that can be prevented by methods like painting and galvanizing to exclude oxygen.
Nitric acid

The document provides information about nitric acid, including its physical and chemical properties, manufacturing processes, uses, and production in Pakistan compared to worldwide. It describes nitric acid as a colorless liquid used to make fertilizers, dyes, and explosives. Three common industrial methods are discussed for manufacturing nitric acid: the Chile saltpeter method using sodium nitrate, Birkland-Eyde's method using air, and Ostwald's ammonia oxidation process. Nitric acid production in Pakistan is compared to global production levels, and major Pakistani nitric acid producers are listed.
Valence Electrons

Valence electrons are the outermost shell electrons of an atom that are involved in bonding. Elements in the same group on the periodic table have the same number of valence electrons because they exhibit similar chemical properties based on their valence electron configuration. Atoms seek to attain a full outer shell of 8 electrons to achieve stability through gaining, losing or sharing valence electrons in chemical bonds.
Chemical Names and Formulas

This document discusses classifying and naming ionic and covalent compounds, as well as writing their formulas. It provides rules for:
- Classifying compounds as ionic or covalent based on their formula
- Naming ionic compounds using stock systems and identifying polyatomic ions
- Naming covalent compounds using prefixes to indicate the number of atoms
- Writing formulas for ionic compounds by balancing charges and for covalent compounds using prefixes
It also discusses acids, bases, and how to name and write formulas for acids based on their anion name endings.
Stoichiometry & The Mole

Class presentation for our Mole and Stoichiometry units as part of the Grade 9 Introduction to Chemistry course.
Chemical Compounds

This document discusses different types of chemical formulas used to represent compounds:
- A chemical compound is made of two or more elements that are combined in fixed ratios.
- The chemical formula uses symbols of the elements and subscripts to show the relative proportions.
- The empirical formula shows the lowest whole number ratio of atoms in a compound, while the molecular formula gives the exact number of each atom.
- The structural formula depicts the specific bonding arrangement of the atoms in a compound.
Percent composition

1. The document discusses calculating percent composition and determining empirical and molecular formulas.
2. To calculate percent composition, the formula mass is determined and the mass of each element is calculated as a percentage of the total mass.
3. An empirical formula shows the lowest whole number ratio of atoms in a compound, while a molecular formula shows the actual number of atoms. Molecular formulas for ionic compounds are always empirical, while molecular formulas for molecular compounds may or may not be empirical.
Chemistry - Chp 9 - Chemical Names and Formulas - Notes

1) This document provides information on naming and writing formulas for ionic and molecular compounds. It discusses monatomic and polyatomic ions, and how to determine the charges on cations and anions.
2) Rules are provided for writing formulas and naming ionic compounds, including those containing transition metals. Prefixes are introduced for naming molecular compounds based on the number of atoms present.
3) Examples are worked through for writing formulas from names and names from formulas for different types of compounds. Practice problems are also included for students to try.
Nomenclature

This document provides information on naming and writing formulas for various types of compounds including:
- Ionic compounds formed from cations and anions
- Molecular compounds formed from nonmetals
- Acids
It discusses common names versus IUPAC names, determining charges on ions, writing formulas to balance charges, and using prefixes to indicate the number of atoms in molecular compounds. Examples are provided to illustrate each type of nomenclature.
More Related Content
What's hot
Chpt 7 part 1 - chemical nomenclature 042403

The document provides an overview of chemical nomenclature and naming conventions for compounds. It discusses the need for a systematic naming system to 1) prevent confusion between compounds, 2) avoid complex formulas, and 3) group similar compounds into families. It then covers naming conventions for binary compounds containing two nonmetals, binary compounds containing a metal and nonmetal, and compounds named using the "ic/ous" method involving metal oxidation states. The goal is to establish a logical and consistent system for naming chemical compounds based on their formulas and elemental components.
Ch. 3 stoichiometry

This document provides an overview of stoichiometry concepts in chemistry. It defines stoichiometry as calculations with chemical formulas and equations. Key concepts covered include the law of conservation of mass, types of chemical reactions, anatomy of chemical equations, formula weights and molecular weights, moles, mole ratios, empirical formulas, combustion analysis, stoichiometric calculations, limiting reactants, theoretical yield, and percent yield.
Chemical properties

Chemical properties describe how a substance reacts or changes during a chemical reaction. Some key chemical properties include flammability, reactivity, and the ability to undergo chemical changes that alter the composition of the substance. Common signs that a chemical change has occurred include a change in color, production of a gas, or formation of a precipitate. However, physical changes can sometimes exhibit similar signs, so the only definitive way to identify a chemical change is if the composition of the substance changes to form new substances.
2.6.1 oxidation numbers

This document discusses oxidation and reduction reactions and provides rules for determining oxidation numbers:
1) Oxidation involves loss of electrons and increases oxidation number, while reduction involves gain of electrons and decreases oxidation number.
2) The sum of oxidation numbers in a molecule or ion must equal the overall charge.
3) Transition metals can have variable oxidation numbers depending on their compounds. Their oxidation states are included in names.
4) Some anomalies may occur where oxidation numbers are fractional or elements appear to gain/lose no electrons.
CALCULATIONS WITH CHEMICAL FORMULAS AND EQUATION

Calculate the empirical formula of vitamin C from the
given data.
MOLE; Avogadro's Number; Percentage Composition

The document defines key chemistry concepts related to moles, including:
- A mole refers to Avogadro's number (6.02x10^23) of particles like atoms, molecules, ions or formula units.
- 1 mole of an element contains 6.02x10^23 atoms, 1 mole of a molecular compound contains 6.02x10^23 molecules, and 1 mole of an ionic compound contains 6.02x10^23 formula units.
- Gram atomic mass refers to the mass of one mole of an element in grams, and gram formula mass refers to the sum of atomic weights that make up one mole of a compound.
- Chemical formulas represent the
Naming compounds

The document provides an overview of naming chemical compounds. It discusses the rules for naming binary ionic compounds that contain metals and nonmetals (types I and II), binary covalent compounds between two nonmetals (type III), compounds containing polyatomic ions, and acids. Examples of applying the naming conventions are provided throughout. The overall strategies are to consider whether the compounds are ionic or covalent, identify cations and anions or elemental names, and apply prefixes or suffixes accordingly when arriving at the systematic name.
Simple techniques of separating mixtures

This document provides information on techniques for separating mixtures. It discusses several methods including sedimentation and decantation, filtration, evaporation, distillation, crystallization, sublimation, chromatography, centrifugation and solvent extraction. The document is intended for a 9th grade science class and aims to teach students how to select the appropriate separation method based on the components of a mixture. It includes links to videos demonstrating each technique.
Writing and Naming formula

This document discusses writing and naming chemical formulas and ions. It begins by explaining that cations are positively charged ions that migrate to the cathode, while anions are negatively charged ions that migrate to the anode. Examples of common cations and anions are provided. The document then discusses rules for writing formulas for ionic compounds without transition metals, ionic compounds with transition metals, and covalent compounds. It also covers naming conventions for polyatomic ions and provides examples of writing formulas from names and names from formulas.
Molarity Molality Dilutions

This document discusses concentration of solutions in chemistry. It defines molarity as moles of solute per liter of solution. It provides an example problem calculating the molarity of a solution made by dissolving 5 grams of nickel chloride hexahydrate in 250 mL of water. It also discusses molality, defined as moles of solute per kilogram of solvent, and provides an example problem calculating the molality of a sodium chloride solution. Finally, it discusses the process of diluting solutions through calculations that conserve the number of moles while changing the volume.
The mole concept

This document discusses moles, molar mass, and Avogadro's number. It explains that a mole is the amount of a substance that contains 6.022x1023 particles, known as Avogadro's number. It also defines molar mass as the mass in grams of one mole of a substance. The document provides examples of calculating molar mass from atomic masses and using molar mass to determine the number of moles or particles in a given mass of a substance.
Empirical and molecular formula class 11

The document discusses empirical and molecular formulas. An empirical formula shows the simplest whole number ratio of atoms in a compound, while a molecular formula shows the exact number of each atom. To calculate molecular formula from empirical formula: 1) make a table with elements, percentages, atomic masses, moles, and simplest ratios; 2) the empirical formula mass is the sum of the atoms' masses; 3) divide the molar mass by the empirical formula mass to get the common factor for the molecular formula. Two examples are given to calculate empirical and molecular formulas from percentage compositions and molar masses.
Air and water

The document provides information about various chemistry concepts related to air and water:
- It describes chemical tests to identify water and the purification of water supplies through filtration and chlorination.
- The composition of clean air is described as 78% nitrogen, 21% oxygen and small quantities of other gases. Common air pollutants like carbon monoxide and their sources are stated.
- Fractional distillation is outlined as the process used to separate oxygen and nitrogen from liquid air based on their different boiling points.
- Rusting is described as a reaction between iron, air and water that can be prevented by methods like painting and galvanizing to exclude oxygen.
Nitric acid

The document provides information about nitric acid, including its physical and chemical properties, manufacturing processes, uses, and production in Pakistan compared to worldwide. It describes nitric acid as a colorless liquid used to make fertilizers, dyes, and explosives. Three common industrial methods are discussed for manufacturing nitric acid: the Chile saltpeter method using sodium nitrate, Birkland-Eyde's method using air, and Ostwald's ammonia oxidation process. Nitric acid production in Pakistan is compared to global production levels, and major Pakistani nitric acid producers are listed.
Valence Electrons

Valence electrons are the outermost shell electrons of an atom that are involved in bonding. Elements in the same group on the periodic table have the same number of valence electrons because they exhibit similar chemical properties based on their valence electron configuration. Atoms seek to attain a full outer shell of 8 electrons to achieve stability through gaining, losing or sharing valence electrons in chemical bonds.
Chemical Names and Formulas

This document discusses classifying and naming ionic and covalent compounds, as well as writing their formulas. It provides rules for:
- Classifying compounds as ionic or covalent based on their formula
- Naming ionic compounds using stock systems and identifying polyatomic ions
- Naming covalent compounds using prefixes to indicate the number of atoms
- Writing formulas for ionic compounds by balancing charges and for covalent compounds using prefixes
It also discusses acids, bases, and how to name and write formulas for acids based on their anion name endings.
Stoichiometry & The Mole

Class presentation for our Mole and Stoichiometry units as part of the Grade 9 Introduction to Chemistry course.
Chemical Compounds

This document discusses different types of chemical formulas used to represent compounds:
- A chemical compound is made of two or more elements that are combined in fixed ratios.
- The chemical formula uses symbols of the elements and subscripts to show the relative proportions.
- The empirical formula shows the lowest whole number ratio of atoms in a compound, while the molecular formula gives the exact number of each atom.
- The structural formula depicts the specific bonding arrangement of the atoms in a compound.
Percent composition

1. The document discusses calculating percent composition and determining empirical and molecular formulas.
2. To calculate percent composition, the formula mass is determined and the mass of each element is calculated as a percentage of the total mass.
3. An empirical formula shows the lowest whole number ratio of atoms in a compound, while a molecular formula shows the actual number of atoms. Molecular formulas for ionic compounds are always empirical, while molecular formulas for molecular compounds may or may not be empirical.
What's hot (20)
Similar to Exam 2 Naming for Oxyanion Series, Ionic/Covalent Compounds, Binary acids/Oxyacids
Chemistry - Chp 9 - Chemical Names and Formulas - Notes

1) This document provides information on naming and writing formulas for ionic and molecular compounds. It discusses monatomic and polyatomic ions, and how to determine the charges on cations and anions.
2) Rules are provided for writing formulas and naming ionic compounds, including those containing transition metals. Prefixes are introduced for naming molecular compounds based on the number of atoms present.
3) Examples are worked through for writing formulas from names and names from formulas for different types of compounds. Practice problems are also included for students to try.
Nomenclature

This document provides information on naming and writing formulas for various types of compounds including:
- Ionic compounds formed from cations and anions
- Molecular compounds formed from nonmetals
- Acids
It discusses common names versus IUPAC names, determining charges on ions, writing formulas to balance charges, and using prefixes to indicate the number of atoms in molecular compounds. Examples are provided to illustrate each type of nomenclature.
7. chemical formulas and naming compnds [Autosaved].pptx![7. chemical formulas and naming compnds [Autosaved].pptx](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![7. chemical formulas and naming compnds [Autosaved].pptx](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
This document provides information on naming and writing formulas for different types of compounds. It discusses binary ionic compounds and how their names and formulas are determined based on the ions present. It also covers binary covalent compounds and how prefixes are used in their names to indicate the ratio of elements. Finally, it mentions polyatomic ions and how their names are used in ionic compound names and formulas.
Naming ii smart

This document discusses how to determine the chemical formula from the name for several different types of compounds, including:
1. Binary covalent compounds by identifying the nonmetals and using prefixes to determine the number of atoms of each element.
2. Acids by identifying if they are binary or ternary, then using oxidation states and the criss-cross method to determine the formula.
3. Ionic compounds by identifying the cation and anion, writing their oxidation states, and using criss-crossing to determine the formula.
4. Compounds with polyatomic ions by identifying cations and anions which may be polyatomic, writing oxidation states, and using criss-cross
Short quiz 1 Chemistry

The document contains a short chemistry quiz asking the student to:
1) Write the formula for common ions.
2) Write the chemical formulas for calcium chloride, aluminum oxide, and sodium hydroxide.
3) Write the chemical equation for the reaction of sodium with oxygen producing a metallic oxide.
Ionic Naming

The document provides information on naming ionic compounds, cations, anions, and polyatomic ions. It discusses how to name monatomic and transition metal cations by adding "-ion" or the charge in Roman numerals after the element name. Anions are named by adding "-ide" after the element root. Ionic compounds are formed by writing the cation first followed by the anion. Polyatomic ions are also included.
Tema 4 the chemical compounds

This document provides information about chemical compounds and their nomenclature. It defines a chemical compound as a pure substance formed by two or more elements combined in fixed proportions. Examples of common compounds and their chemical formulas are provided. The document then discusses different systems for naming compounds, including classical nomenclature, nomenclature using Roman numerals to indicate elemental valence, and nomenclature using Greek prefixes to indicate the number of atoms present. Specific rules and examples are provided for naming oxides, hydroxides, and peroxides. A series of exercises are included to have the reader practice writing formulas and names according to the various nomenclature systems.
Chapter 9

This document provides information on naming and writing formulas for different types of chemical compounds including:
1. Ionic compounds consisting of monatomic and polyatomic ions are named by writing the cation first followed by the anion. Transition metal ions have stock and classical naming systems.
2. Molecular compounds consisting of two nonmetals are named using prefixes to indicate the number of atoms of each element followed by the "-ide" suffix.
3. Acids are named based on the anion present, using prefixes like "hydro-", "-ous", or "-ic" depending on the anion suffix. Their formulas are written with hydrogen and the anion.
4. Bases are named by writing the cation
Naming 2 SMART

This document provides instructions for determining the chemical formula from the name of different types of compounds, including:
- Binary covalent compounds, where the formula is determined by identifying the nonmetals and using prefixes to indicate the number of atoms.
- Acids, where binary acids use oxidation states to determine the formula of hydrogen and the nonmetal, and ternary acids identify polyatomic ions.
- Ionic compounds, where the metal and nonmetal are identified and oxidation states are used with the crisscross method to determine the formula.
- Compounds with polyatomic ions, which also use oxidation states and crisscrossing to determine the formula when a polyatomic ion is present.
Naming Compounds (2).ppt

This document provides information on naming ionic and covalent compounds. It discusses how to name ionic compounds based on their cation and anion. It also discusses naming transition metal ions based on their charge. For covalent compounds, it describes naming binary molecular compounds and compounds containing hydrogen or carbon. It provides examples of naming ionic compounds, transition metal compounds, and molecular compounds systematically. It also discusses writing chemical formulas, identifying common polyatomic ions, and recognizing ionic versus covalent character.
Wks reviewing concepts

The document contains a review of concepts related to acids and bases including:
1) Acids have a sour taste and react with metals and carbonates to produce gases. They release H+ ions in water and turn litmus paper red.
2) Bases taste bitter and feel slippery. They conduct electricity in solution, react with fats to produce soap, and turn litmus paper blue.
3) The document asks questions about strong vs weak acids/bases, gives examples of each, and discusses ionization in solutions and chemical equations involving acids and bases.
Formula-Writing-and-Naming-Compounds.pdf

This document provides information on naming ions and compounds. It discusses rules for naming positive and negative ions, as well as binary ionic compounds, binary molecular compounds, exceptions, binary acids, ternary compounds, and ternary acids. Examples are given for each type of compound. The objectives are for learners to be able to name compounds from formulas and write formulas from names. Review questions and an assignment are included at the end.
_orcututgk_Naming Compounds-Chemistry.ppt

1. The document discusses naming ionic and covalent compounds. It provides steps for naming ionic compounds, which involve writing the name of the metal ion and nonmetal ion. Covalent compounds are named by writing the names of the nonmetals and adding prefixes to indicate ratios.
2. Binary covalent compounds are between two nonmetals and are named using prefixes like mono, di, and tri to show ratios of elements. Polyatomic ions are also named and used in ionic compounds.
3. Determining if a compound is ionic or covalent depends on whether it contains a metal and nonmetal (ionic) or only nonmetals (covalent).
Naming Compounds-Chemistry.ppt

This document provides instructions for naming ionic and binary covalent compounds. It explains that ionic compounds are formed from metals and nonmetals, while covalent compounds contain only nonmetals. The naming process for ionic compounds involves writing the name of the metal ion followed by the nonmetal ion changed to -ide. For covalent compounds, prefixes are used to indicate the number of each nonmetal present. Practice problems are provided to name compounds and write formulas.
Naming and Writing Formula of Compounds.pptx

This document provides instructions for naming ionic and binary covalent compounds. It explains that ionic compounds are formed from metals and nonmetals, while covalent compounds contain only nonmetals. The naming process for ionic compounds involves writing the name of the metal ion followed by the nonmetal ion changed to -ide. For covalent compounds, prefixes are used to indicate the number of each nonmetal present.
Chemical Reactions

Here are the steps to write the formula for calcium bicarbonate:
1. Write the ions involved - Ca2+ and HCO3-
2. Work out the ratio of ions needed for charge balance. Ca2+ : HCO3- = 1 : 2
3. Write the formula without charges - CaHCO3
4. Add subscripts to reflect the ratio - Ca(HCO3)2
The formula for calcium bicarbonate is Ca(HCO3)2.
C04 elements and compounds

- The document discusses elements and compounds. It defines an element as a substance that cannot be broken down further by chemical means, while a compound is made up of two or more elements chemically bonded together.
- Formulas are used to represent the composition of compounds, showing the symbols and ratios of elements present. Writing balanced chemical equations requires determining the correct ratios of reactants and products to satisfy the law of conservation of mass.
C04 elements and compounds

The document is a chapter about elements and compounds from a chemistry textbook. It contains the following key points in 3 sentences:
The chapter defines elements as substances that cannot be broken down further, while compounds are substances made of two or more elements chemically bonded together. It explains that elements are represented by chemical symbols and compounds by chemical formulas showing the ratios of atoms present. The chapter also discusses writing and balancing chemical equations to represent chemical reactions in terms of reactants and products.
Ch 05 nomenclature packet ch099 a

The document provides information on naming and identifying different types of chemical compounds including:
- Ionic compounds can be identified by their metal and nonmetal components and named using stock nomenclature.
- Molecular compounds contain two nonmetals and include acids, hydrocarbons, and binary compounds.
- Practice problems are provided to test the ability to identify, name, and write formulas for various ionic and molecular compounds.
Naming compounds chemistry

The document provides instructions for naming ionic and binary covalent compounds. Ionic compounds are formed from metals and nonmetals, with the metal ions taking positive charges and the nonmetal ions taking negative charges. Binary covalent compounds are formed from two nonmetals sharing electrons in ratios indicated by prefixes like di-, tri-, and tetra-. The naming process for ionic compounds involves writing the name of the metal ion followed by the name of the nonmetal ion. For binary covalent compounds, the process involves writing the names of the two nonmetals, changing the second to end in "-ide", and adding prefixes to indicate ratios.
Similar to Exam 2 Naming for Oxyanion Series, Ionic/Covalent Compounds, Binary acids/Oxyacids (20)
Chemistry - Chp 9 - Chemical Names and Formulas - Notes

Chemistry - Chp 9 - Chemical Names and Formulas - Notes
7. chemical formulas and naming compnds [Autosaved].pptx![7. chemical formulas and naming compnds [Autosaved].pptx](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![7. chemical formulas and naming compnds [Autosaved].pptx](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
7. chemical formulas and naming compnds [Autosaved].pptx
Recently uploaded
How to Setup Warehouse & Location in Odoo 17 Inventory

In this slide, we'll explore how to set up warehouses and locations in Odoo 17 Inventory. This will help us manage our stock effectively, track inventory levels, and streamline warehouse operations.
BÀI TẬP BỔ TRỢ TIẾNG ANH 8 CẢ NĂM - GLOBAL SUCCESS - NĂM HỌC 2023-2024 (CÓ FI...

BÀI TẬP BỔ TRỢ TIẾNG ANH 8 CẢ NĂM - GLOBAL SUCCESS - NĂM HỌC 2023-2024 (CÓ FI...Nguyen Thanh Tu Collection
https://app.box.com/s/y977uz6bpd3af4qsebv7r9b7s21935vdStack Memory Organization of 8086 Microprocessor

The stack memory organization of 8086 microprocessor.
BBR 2024 Summer Sessions Interview Training

Qualitative research interview training by Professor Katrina Pritchard and Dr Helen Williams
Leveraging Generative AI to Drive Nonprofit Innovation

In this webinar, participants learned how to utilize Generative AI to streamline operations and elevate member engagement. Amazon Web Service experts provided a customer specific use cases and dived into low/no-code tools that are quick and easy to deploy through Amazon Web Service (AWS.)
HYPERTENSION - SLIDE SHARE PRESENTATION.

IT WILL BE HELPFULL FOR THE NUSING STUDENTS
IT FOCUSED ON MEDICAL MANAGEMENT AND NURSING MANAGEMENT.
HIGHLIGHTS ON HEALTH EDUCATION.
Temple of Asclepius in Thrace. Excavation results

The temple and the sanctuary around were dedicated to Asklepios Zmidrenus. This name has been known since 1875 when an inscription dedicated to him was discovered in Rome. The inscription is dated in 227 AD and was left by soldiers originating from the city of Philippopolis (modern Plovdiv).
Philippine Edukasyong Pantahanan at Pangkabuhayan (EPP) Curriculum

(𝐓𝐋𝐄 𝟏𝟎𝟎) (𝐋𝐞𝐬𝐬𝐨𝐧 𝟏)-𝐏𝐫𝐞𝐥𝐢𝐦𝐬
𝐃𝐢𝐬𝐜𝐮𝐬𝐬 𝐭𝐡𝐞 𝐄𝐏𝐏 𝐂𝐮𝐫𝐫𝐢𝐜𝐮𝐥𝐮𝐦 𝐢𝐧 𝐭𝐡𝐞 𝐏𝐡𝐢𝐥𝐢𝐩𝐩𝐢𝐧𝐞𝐬:
- Understand the goals and objectives of the Edukasyong Pantahanan at Pangkabuhayan (EPP) curriculum, recognizing its importance in fostering practical life skills and values among students. Students will also be able to identify the key components and subjects covered, such as agriculture, home economics, industrial arts, and information and communication technology.
𝐄𝐱𝐩𝐥𝐚𝐢𝐧 𝐭𝐡𝐞 𝐍𝐚𝐭𝐮𝐫𝐞 𝐚𝐧𝐝 𝐒𝐜𝐨𝐩𝐞 𝐨𝐟 𝐚𝐧 𝐄𝐧𝐭𝐫𝐞𝐩𝐫𝐞𝐧𝐞𝐮𝐫:
-Define entrepreneurship, distinguishing it from general business activities by emphasizing its focus on innovation, risk-taking, and value creation. Students will describe the characteristics and traits of successful entrepreneurs, including their roles and responsibilities, and discuss the broader economic and social impacts of entrepreneurial activities on both local and global scales.
A Visual Guide to 1 Samuel | A Tale of Two Hearts

These slides walk through the story of 1 Samuel. Samuel is the last judge of Israel. The people reject God and want a king. Saul is anointed as the first king, but he is not a good king. David, the shepherd boy is anointed and Saul is envious of him. David shows honor while Saul continues to self destruct.
LAND USE LAND COVER AND NDVI OF MIRZAPUR DISTRICT, UP

This Dissertation explores the particular circumstances of Mirzapur, a region located in the
core of India. Mirzapur, with its varied terrains and abundant biodiversity, offers an optimal
environment for investigating the changes in vegetation cover dynamics. Our study utilizes
advanced technologies such as GIS (Geographic Information Systems) and Remote sensing to
analyze the transformations that have taken place over the course of a decade.
The complex relationship between human activities and the environment has been the focus
of extensive research and worry. As the global community grapples with swift urbanization,
population expansion, and economic progress, the effects on natural ecosystems are becoming
more evident. A crucial element of this impact is the alteration of vegetation cover, which plays a
significant role in maintaining the ecological equilibrium of our planet.Land serves as the foundation for all human activities and provides the necessary materials for
these activities. As the most crucial natural resource, its utilization by humans results in different
'Land uses,' which are determined by both human activities and the physical characteristics of the
land.
The utilization of land is impacted by human needs and environmental factors. In countries
like India, rapid population growth and the emphasis on extensive resource exploitation can lead
to significant land degradation, adversely affecting the region's land cover.
Therefore, human intervention has significantly influenced land use patterns over many
centuries, evolving its structure over time and space. In the present era, these changes have
accelerated due to factors such as agriculture and urbanization. Information regarding land use and
cover is essential for various planning and management tasks related to the Earth's surface,
providing crucial environmental data for scientific, resource management, policy purposes, and
diverse human activities.
Accurate understanding of land use and cover is imperative for the development planning
of any area. Consequently, a wide range of professionals, including earth system scientists, land
and water managers, and urban planners, are interested in obtaining data on land use and cover
changes, conversion trends, and other related patterns. The spatial dimensions of land use and
cover support policymakers and scientists in making well-informed decisions, as alterations in
these patterns indicate shifts in economic and social conditions. Monitoring such changes with the
help of Advanced technologies like Remote Sensing and Geographic Information Systems is
crucial for coordinated efforts across different administrative levels. Advanced technologies like
Remote Sensing and Geographic Information Systems
9
Changes in vegetation cover refer to variations in the distribution, composition, and overall
structure of plant communities across different temporal and spatial scales. These changes can
occur natural.
Lifelines of National Economy chapter for Class 10 STUDY MATERIAL PDF

The chapter Lifelines of National Economy in Class 10 Geography focuses on the various modes of transportation and communication that play a vital role in the economic development of a country. These lifelines are crucial for the movement of goods, services, and people, thereby connecting different regions and promoting economic activities.
How to Make a Field Mandatory in Odoo 17

In Odoo, making a field required can be done through both Python code and XML views. When you set the required attribute to True in Python code, it makes the field required across all views where it's used. Conversely, when you set the required attribute in XML views, it makes the field required only in the context of that particular view.
Recently uploaded (20)
How to Setup Warehouse & Location in Odoo 17 Inventory

How to Setup Warehouse & Location in Odoo 17 Inventory
BÀI TẬP BỔ TRỢ TIẾNG ANH 8 CẢ NĂM - GLOBAL SUCCESS - NĂM HỌC 2023-2024 (CÓ FI...

BÀI TẬP BỔ TRỢ TIẾNG ANH 8 CẢ NĂM - GLOBAL SUCCESS - NĂM HỌC 2023-2024 (CÓ FI...
Leveraging Generative AI to Drive Nonprofit Innovation

Leveraging Generative AI to Drive Nonprofit Innovation
Philippine Edukasyong Pantahanan at Pangkabuhayan (EPP) Curriculum

Philippine Edukasyong Pantahanan at Pangkabuhayan (EPP) Curriculum
SWOT analysis in the project Keeping the Memory @live.pptx

SWOT analysis in the project Keeping the Memory @live.pptx
LAND USE LAND COVER AND NDVI OF MIRZAPUR DISTRICT, UP

LAND USE LAND COVER AND NDVI OF MIRZAPUR DISTRICT, UP
Lifelines of National Economy chapter for Class 10 STUDY MATERIAL PDF

Lifelines of National Economy chapter for Class 10 STUDY MATERIAL PDF
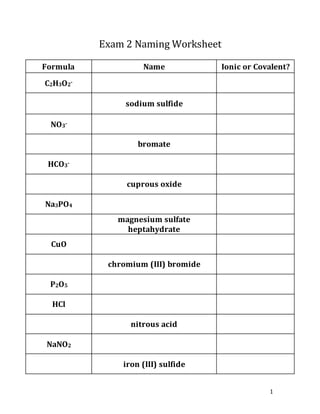
Exam 2 Naming for Oxyanion Series, Ionic/Covalent Compounds, Binary acids/Oxyacids
- 1. 1 Exam 2 Naming Worksheet Formula Name Ionic or Covalent? C2H3O2- sodium sulfide NO3- bromate HCO3- cuprous oxide Na3PO4 magnesium sulfate heptahydrate CuO chromium (III) bromide P2O5 HCl nitrous acid NaNO2 iron (III) sulfide
- 2. 2 Monoatomic Ions Metals: Nonmetals: Polyatomic Ions: covalently bonded compounds made of two or more atoms that are considered as a single unit. Ammonia: ____________ Ammonium: __________ Acetate: _______________ Cyanide:_______________ Hydroxide:____________ Peroxide:______________ Oxyanions: type of polyatomic ions with added oxygen atom BO33- borate CO32- carbonate Two Ions in Series Four Ions in Series Compound more oxygen atoms ends with ________ Compound with less oxygen atoms ends with ________ NO3- ____________ NO2- ____________ SO42- ____________ SO3-2 ____________ SeO42-___________ SeO32- ____________ SO42-____________ SO3-2_____________ TeO42-___________ TeO32-____________ AsO43-___________ AsO33-___________ PO43-____________ PO33-____________ Oxyanion with more oxygen atoms than the “-ate”, now also begins with _______ Oxyanion with less oxygen atoms than the “-ite”, now also begins with ________ ClO4- ___________ IO4-____________ ClO3- ___________ IO3- ____________ ClO2- ___________ IO2-____________ ClO- ____________ IO- ____________ BrO4-____________ BrO3- ____________ BrO2- ____________ BrO- ___________
- 3. 3 Oxyanions with H+ added Current Version: (-prefix hydrogen OR dihyrogen)+(anion) PO43- phosphate ion = HPO42- hydrogen phosphate ion = H2PO4- dihydrogen phosphate ion Older Version: (-prefix bi)+(anion) HCO3- _______________________ HPO42- _______________________ Binary Ionic Compounds Metals forming a single ion: ________________________________________ KCl ____________________ Na2S ____________________ Metals forming multiple ions Current: ______________________________________________ CuO _________________ Cu2O ___________________ ________ iron (III) sulfate ________ copper (II) chloride Older Version: ____________________________________________ FeS ___________________ Fe2S3 _____________________ ________ copper (I) chloride _______ mercury (II) oxide
- 4. 4 Ionic Compounds containing polyatomic Ions Since we should have the polyatomic ions charge memorized, we do not need to indicate how many of each ion there will be. MgSO4 _______________________ __________ aluminum chromate Na3PO4 _______________________ Hydrates: Ionic compounds with water molecules added We use prefixes to indicate the number of water molecules 1=mono 2=di 3=tri 4=tetra 5=penta 6=hexa 7=hepta 8=octa 9=nona 10=deca MgSO4 7H2O magnesium sulfate ________hydrate CoCl2 6H2O cobalt (II) chloride ________hydrate (Barely Ever seen on Exams) Covalent Compounds Formula:(prefix)+(1st non-metal)+(prefix)+(2nd non-metal) (same prefixes as the hydrates) 1=mono 2=di 3=tri 4=tetra 5=penta 6=hexa 7=hepta 8=octa 9=nona 10=deca NCl3 nitrogen trichloride P2O5 diphosphorous pentoxide ________ disulfur dichloride Cl2O7 __________________________
- 5. 5 Binary Acids: Hydrogen + non-metal Formula: (hydro)+(nonmetal+ic)+(acid) HBr hydrobromic acid _______ hydrochloric acid H2S ____________________________ HF ____________________________ Oxyacids: Hydrogen + polyatomic ion If polyatomic ion ended in –ate, it changes to –ic acid If polyatomic ion ended in –ite, it changes to –ous acid EX: chloratechloric acid chlorite chlorous acid HNO4 pernitric acid HNO3 __________________ HNO2 __________________ ________ hyponitrous acid H2CO4 _______________________ H2CO3 ______________________ __________ carbonous acid H2CO________________________
