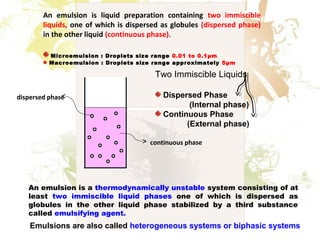
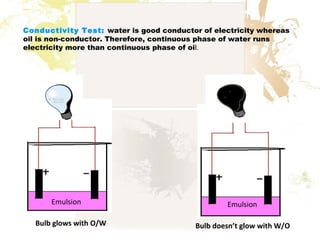
1. An emulsion is a mixture of two immiscible liquids, where one liquid is dispersed as globules in the other liquid.
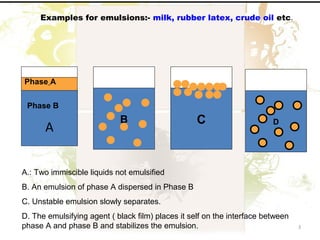
2. Emulsions are thermodynamically unstable and require an emulsifying agent to stabilize the globules and prevent separation.
3. The key types of emulsions are oil-in-water and water-in-oil emulsions, as well as multiple emulsions containing both oil and water phases. Microemulsions are transparent, thermodynamically stable mixtures of oil, water and surfactants.