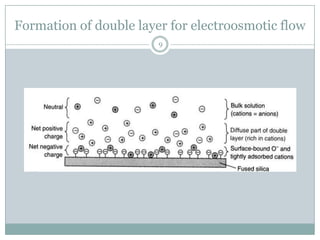
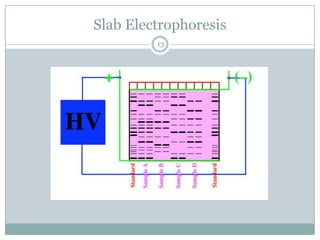
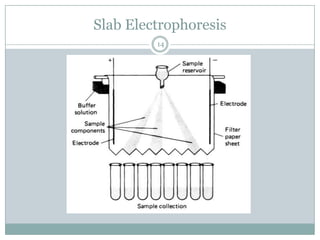
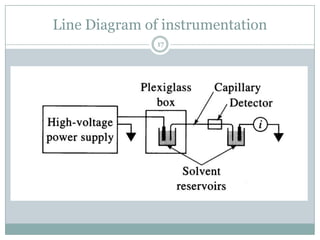
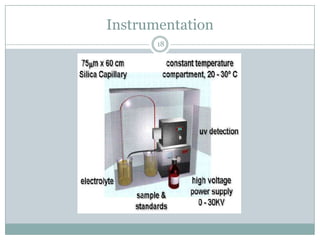
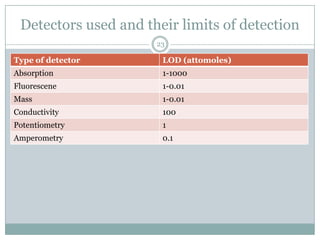
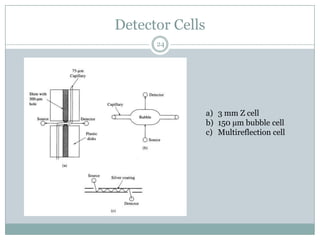
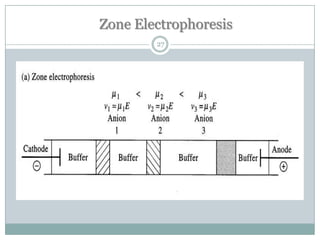
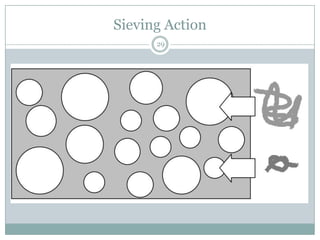
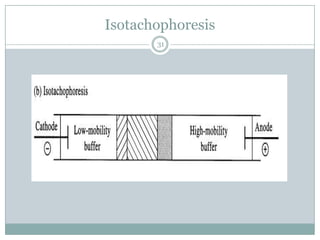
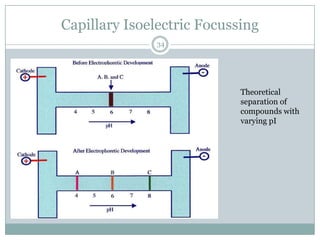
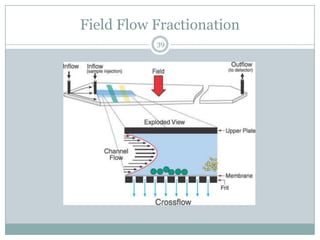
This document provides information about electrophoresis. It discusses different types of electrophoretic techniques including slab electrophoresis, capillary electrophoresis, capillary zone electrophoresis, capillary gel electrophoresis, capillary isotachophoresis, and micellar electrokinetic chromatography. It also covers principles, instrumentation, applications in areas like DNA analysis and vaccine analysis.