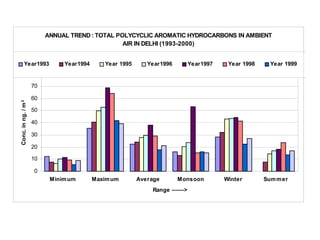
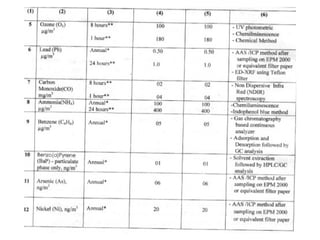
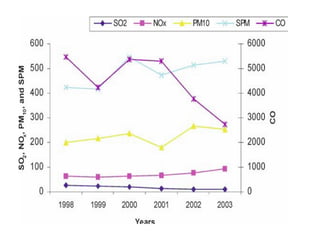
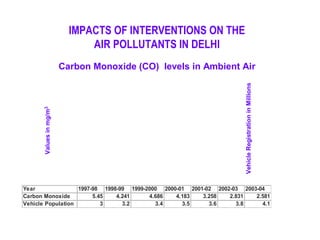
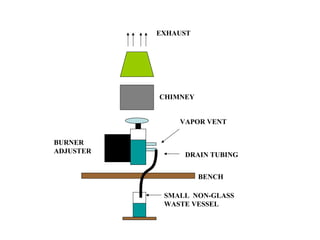
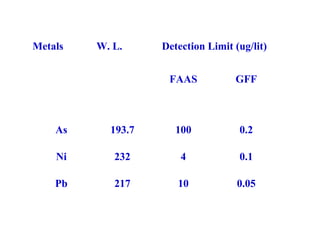
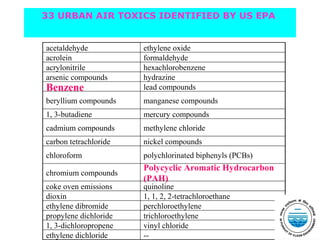
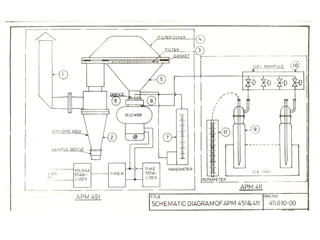
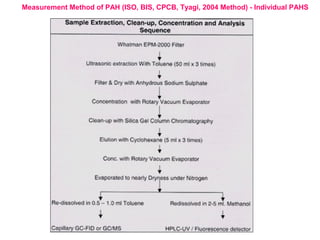
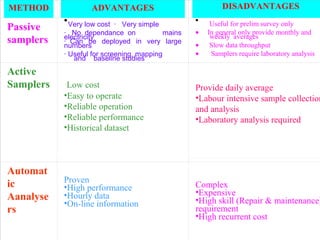
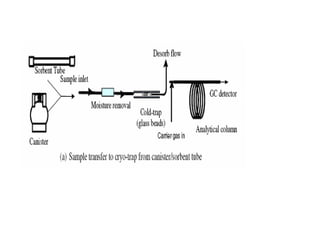
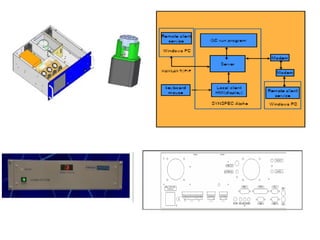
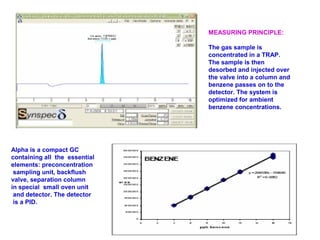
This document discusses techniques for measuring organic pollutants like benzene and benzo(a)pyrene as well as metals in ambient air. It provides an overview of the Central Pollution Control Board of India and their role in monitoring air quality and enforcing standards. Specific analytical techniques are described for sampling and analyzing polycyclic aromatic hydrocarbons and other organic compounds in air particulate matter using gas chromatography.
















































![ppm = (mg/m3) x (24.45/MW)
Expressing
Concentration in ppm
ppm= [(mg/m3)x(24.45/MW)x(760/P)x((T+273)/298)]](https://image.slidesharecdn.com/drtyagilecturepresentnbbitenvirofinal12feb10-151224102531/85/Dr-tyagi-lecture-presentn-bbit-enviro-final-12-feb10-58-320.jpg)