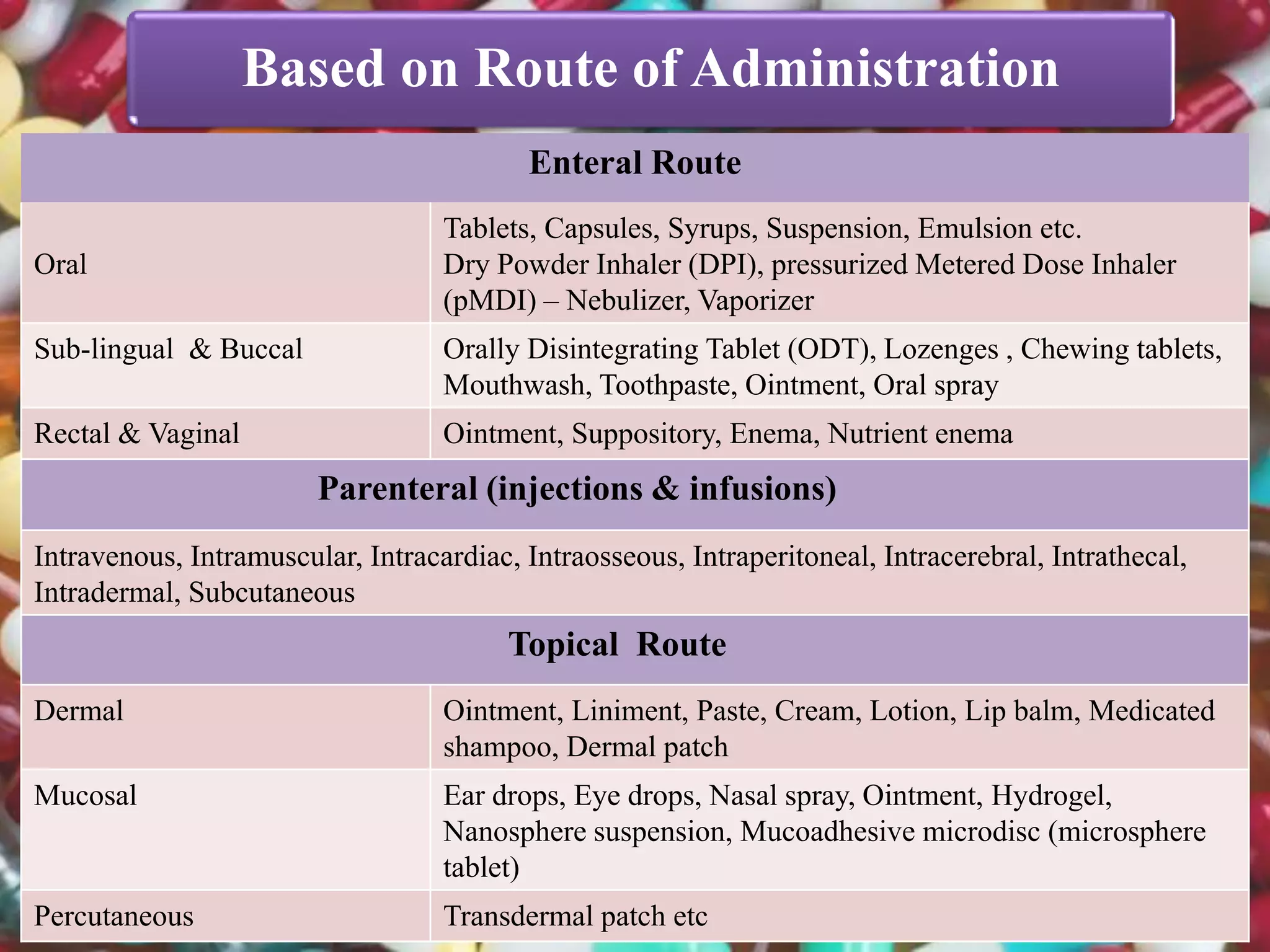
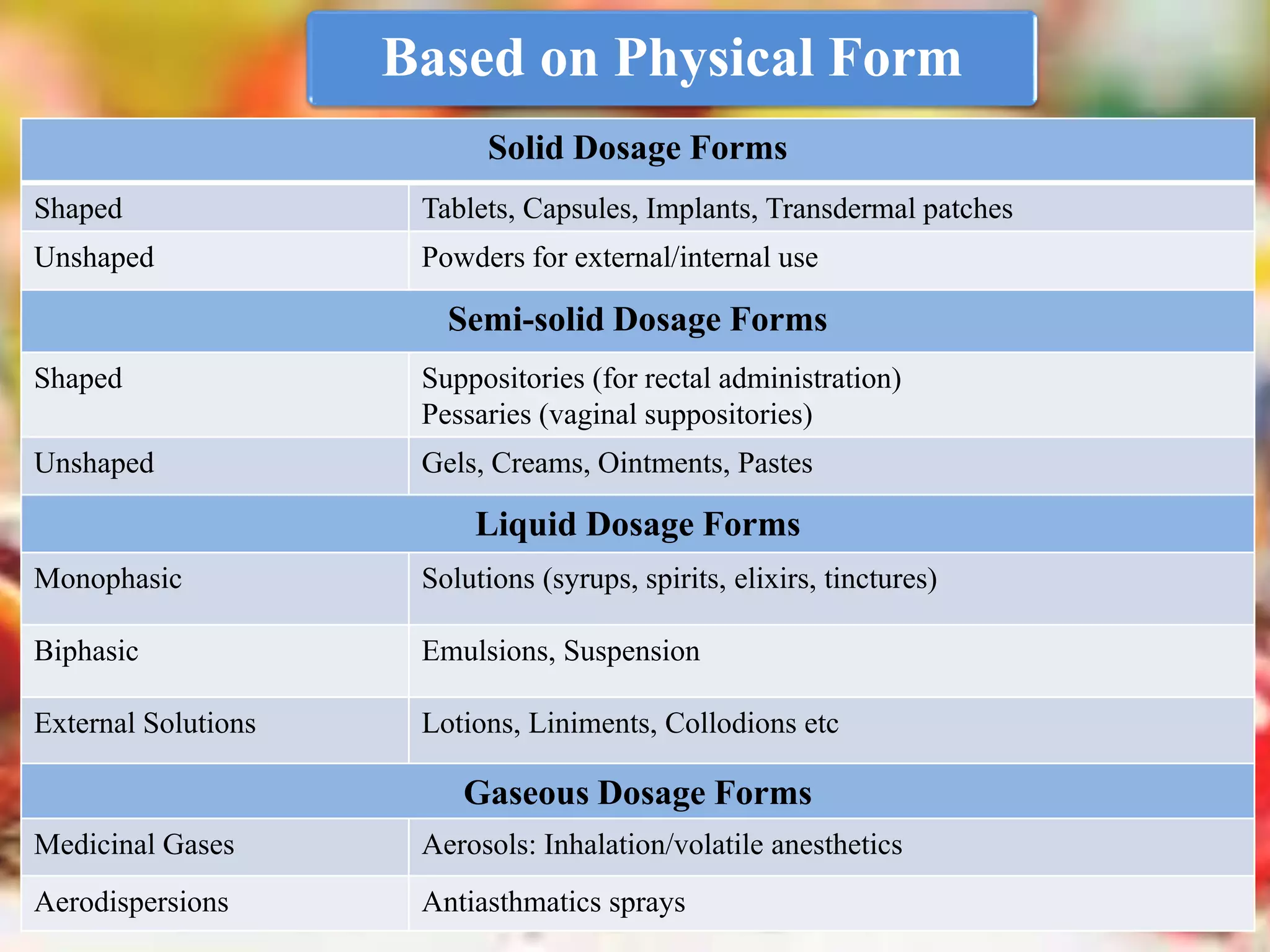
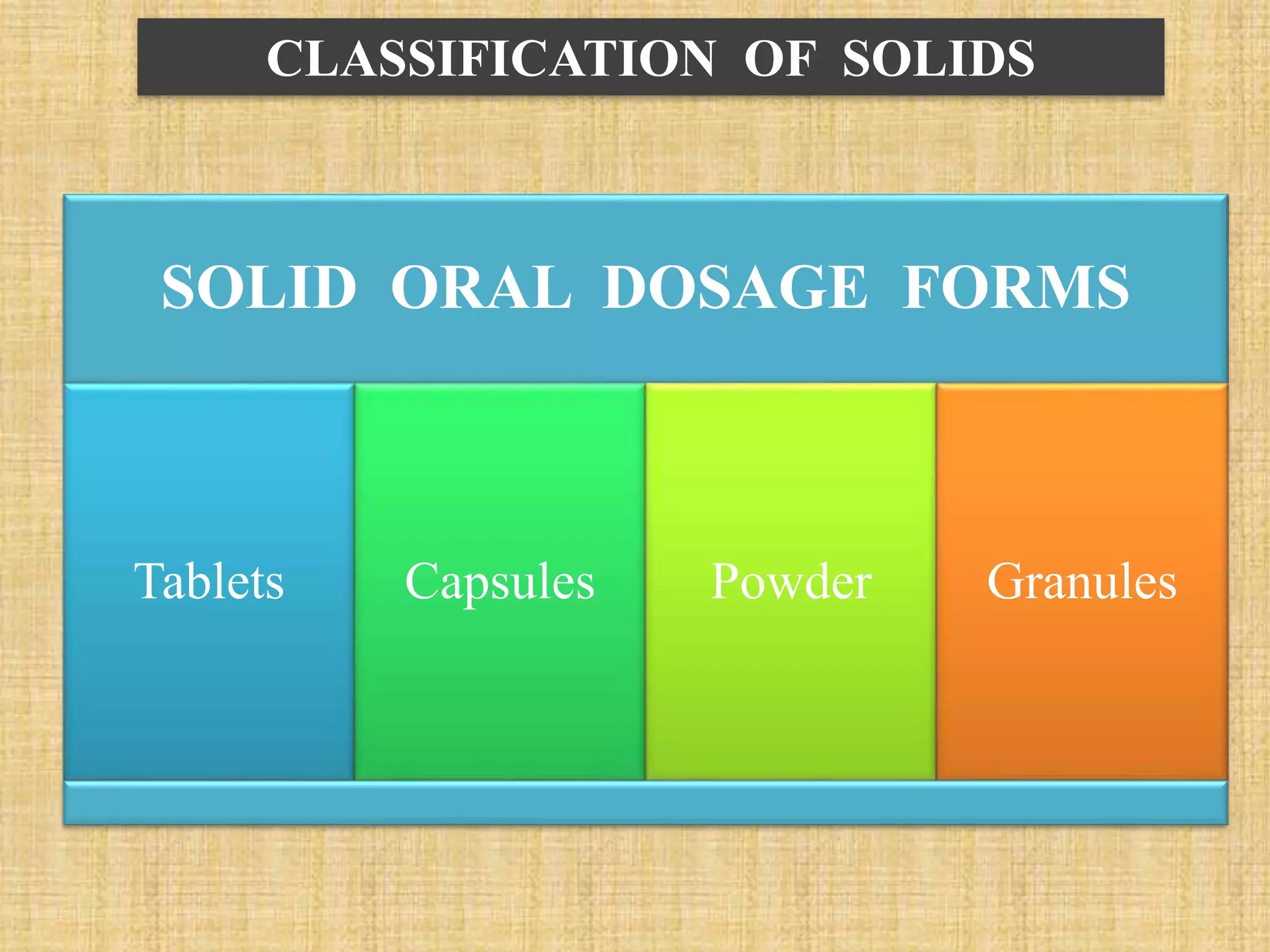
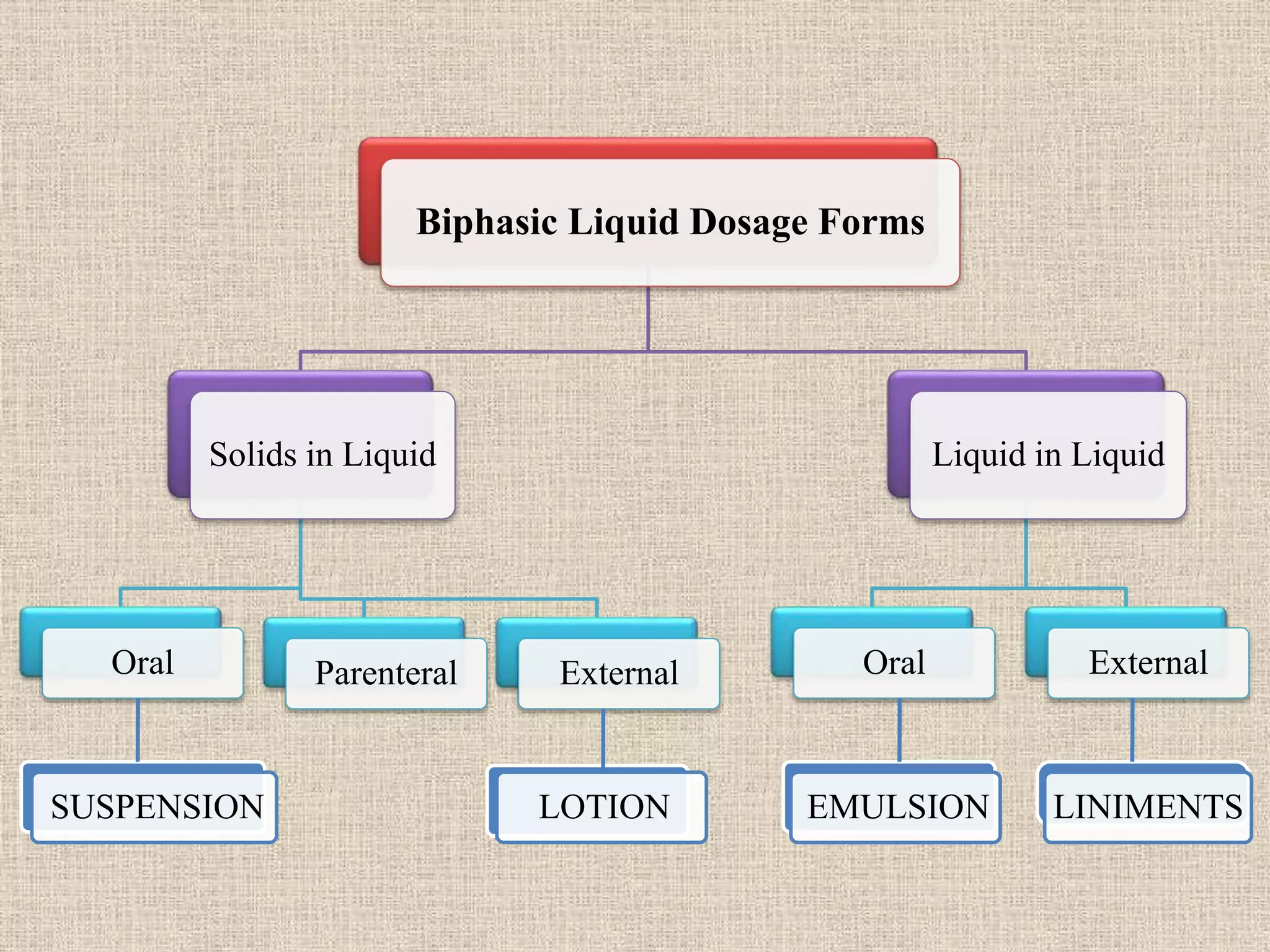
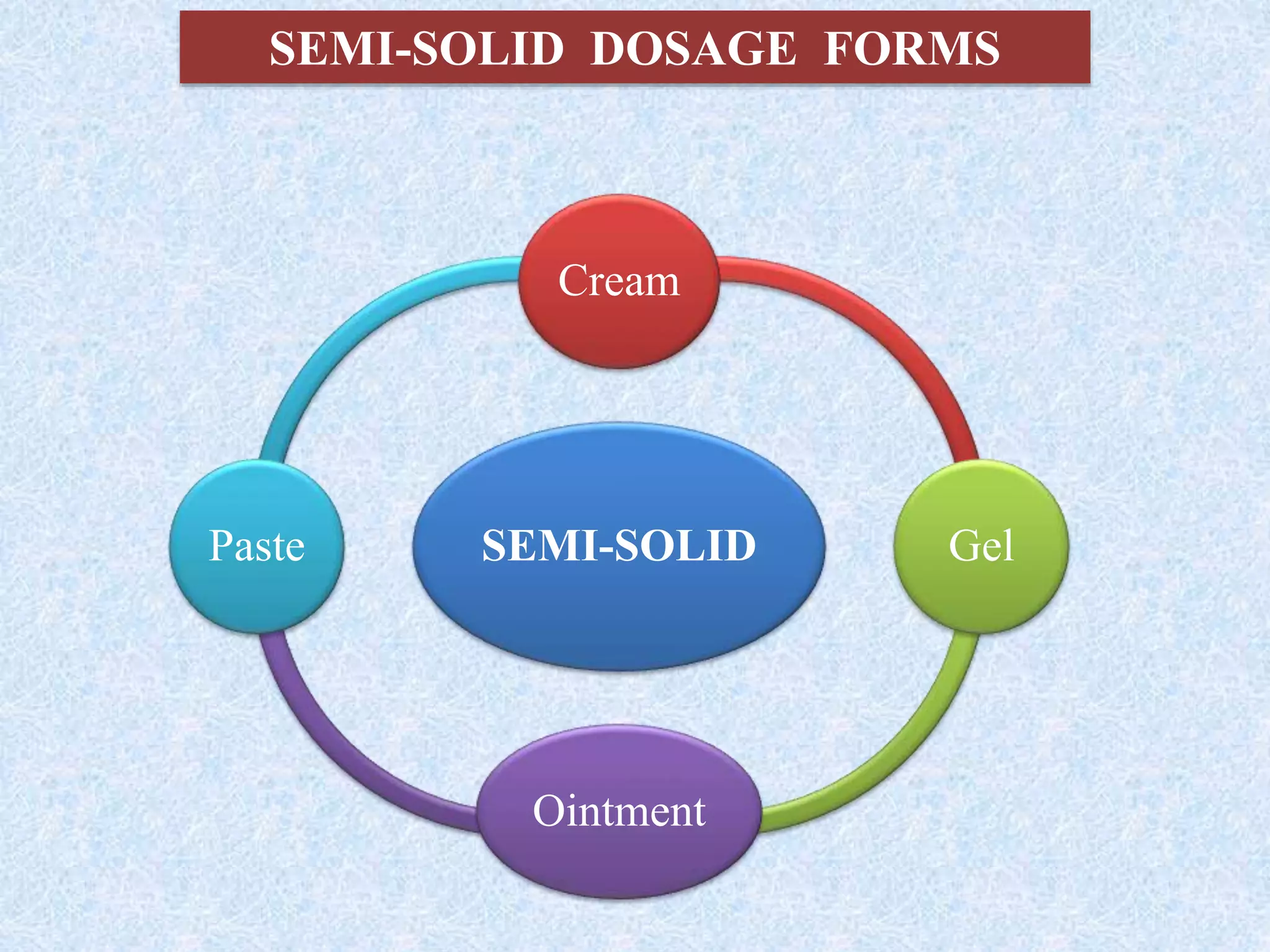
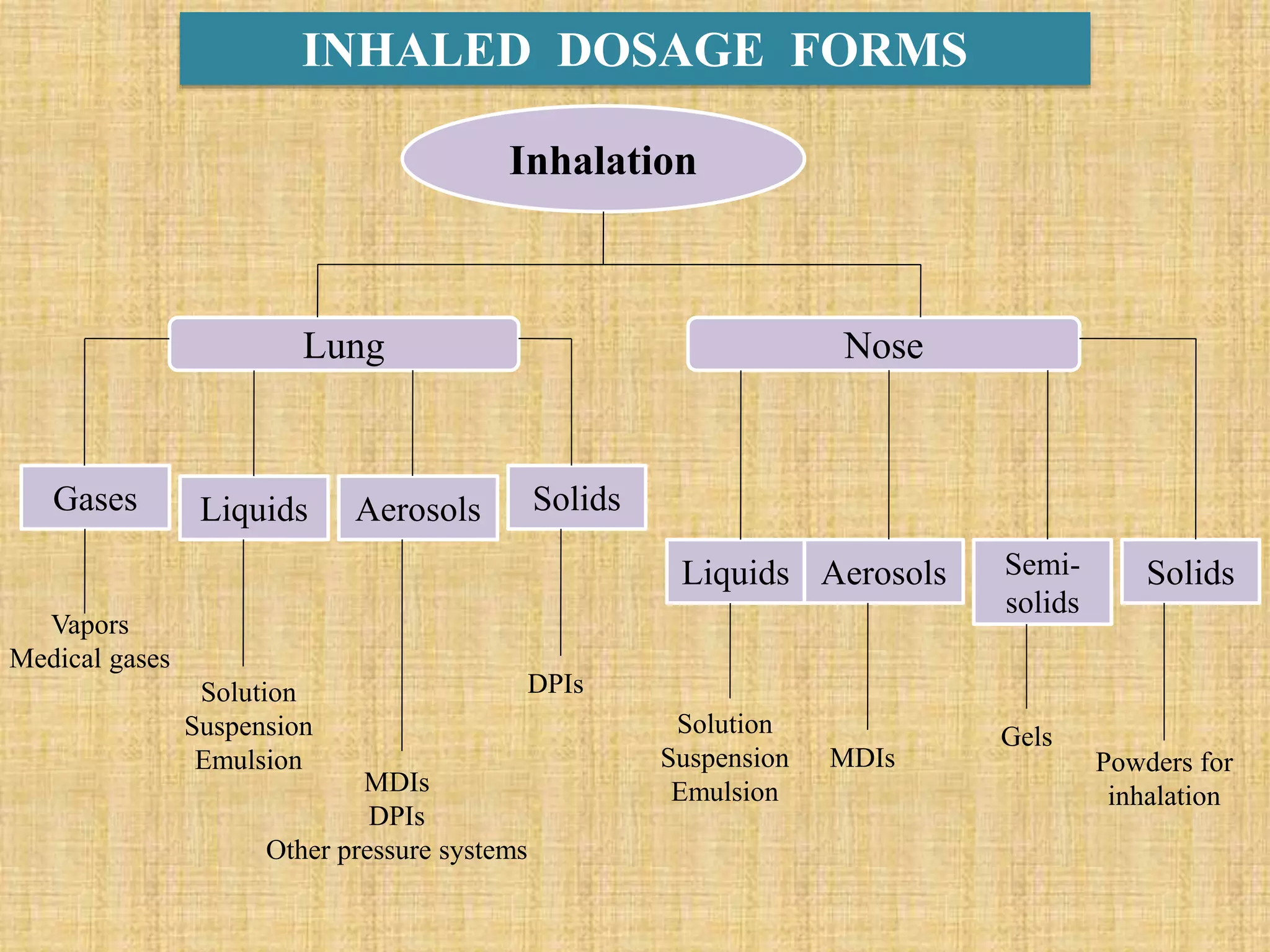
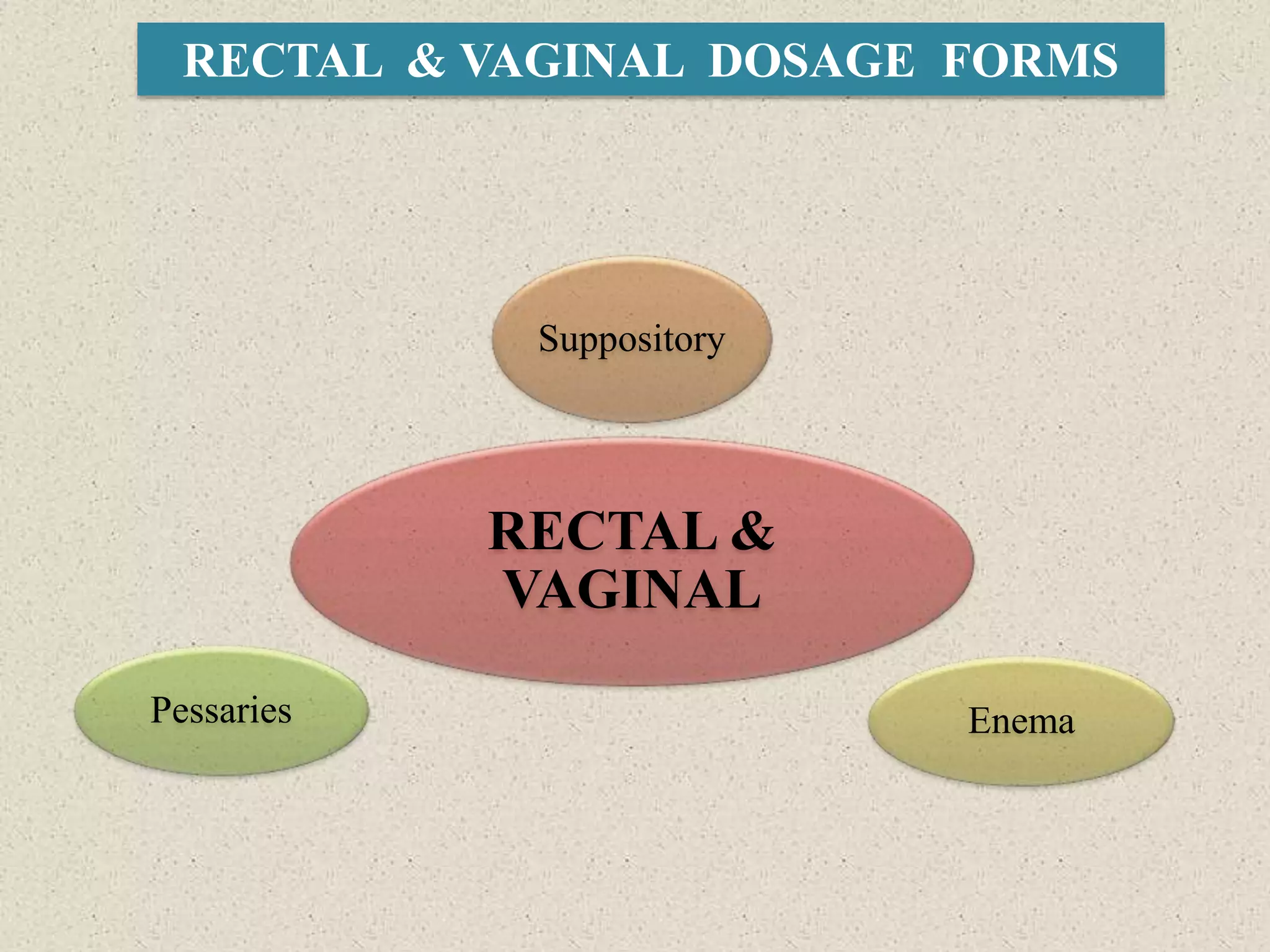
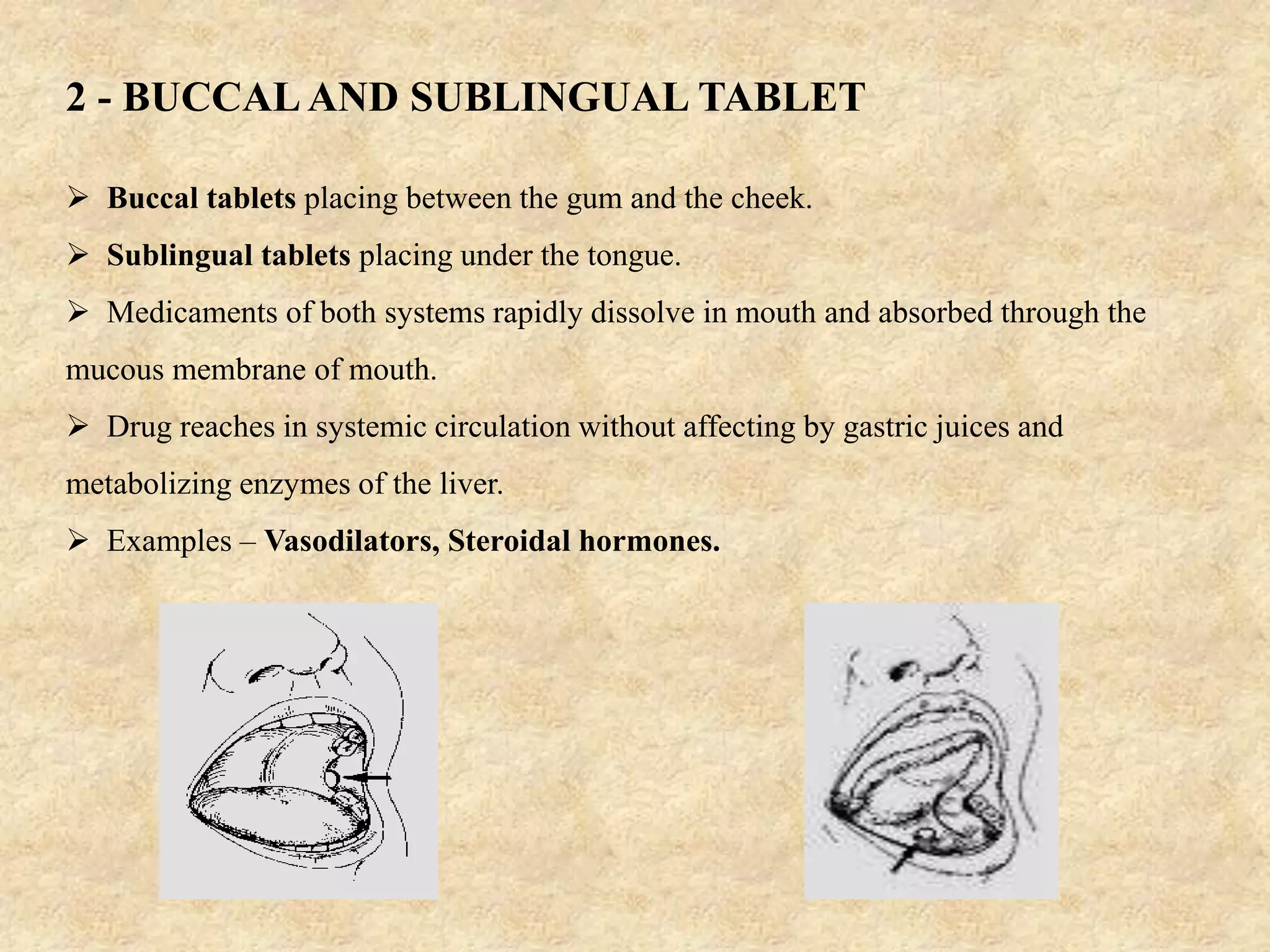
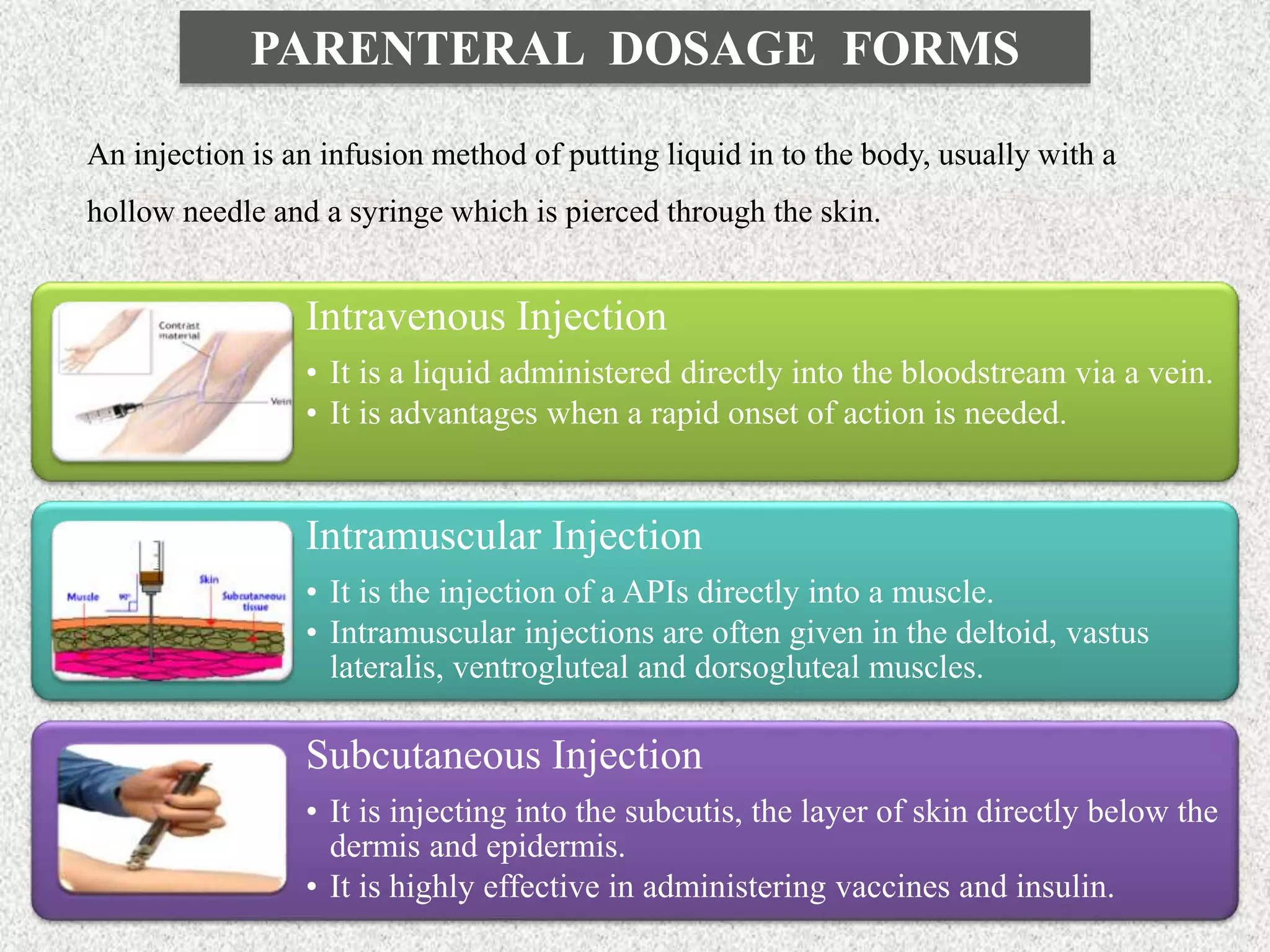
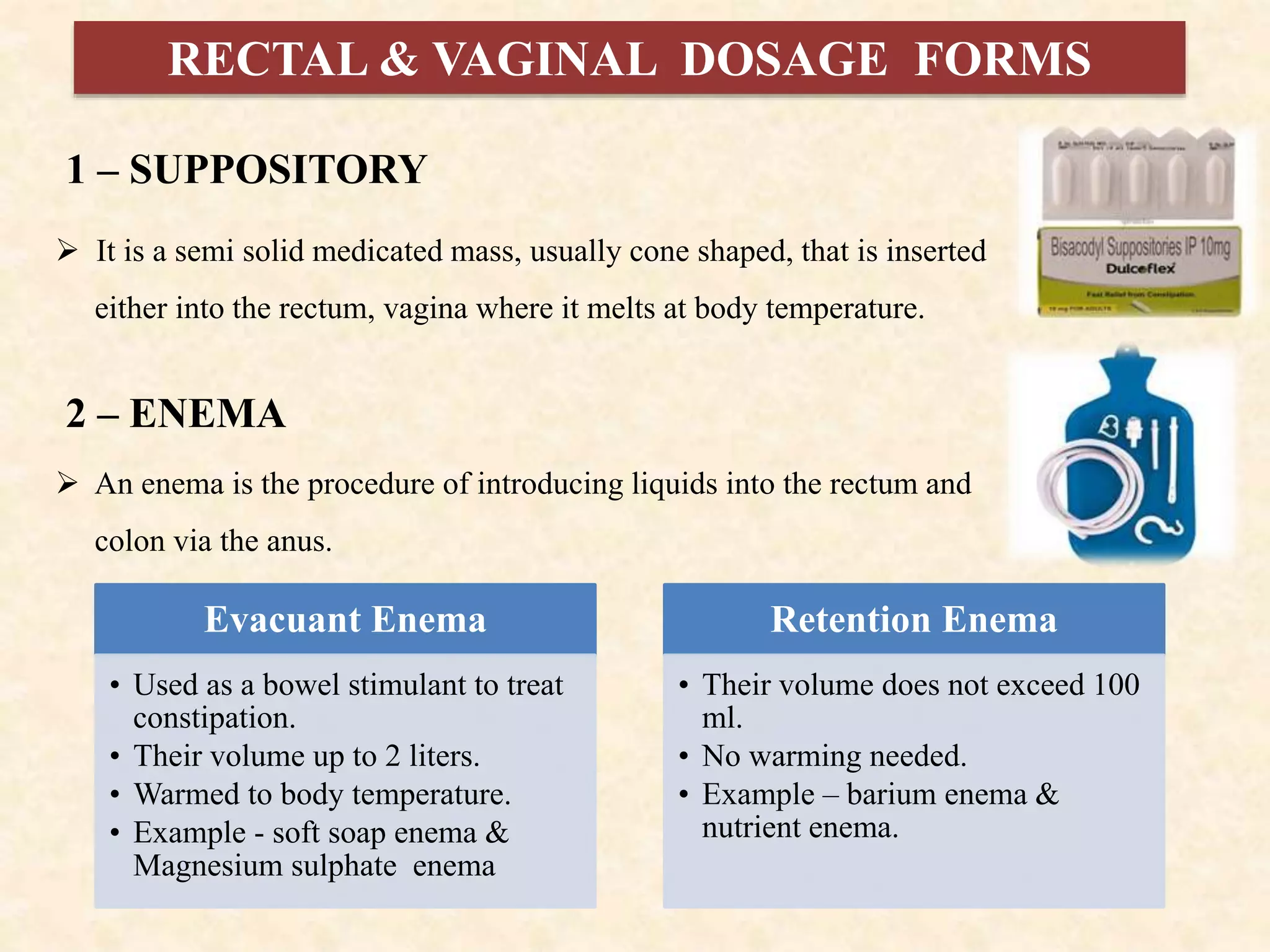
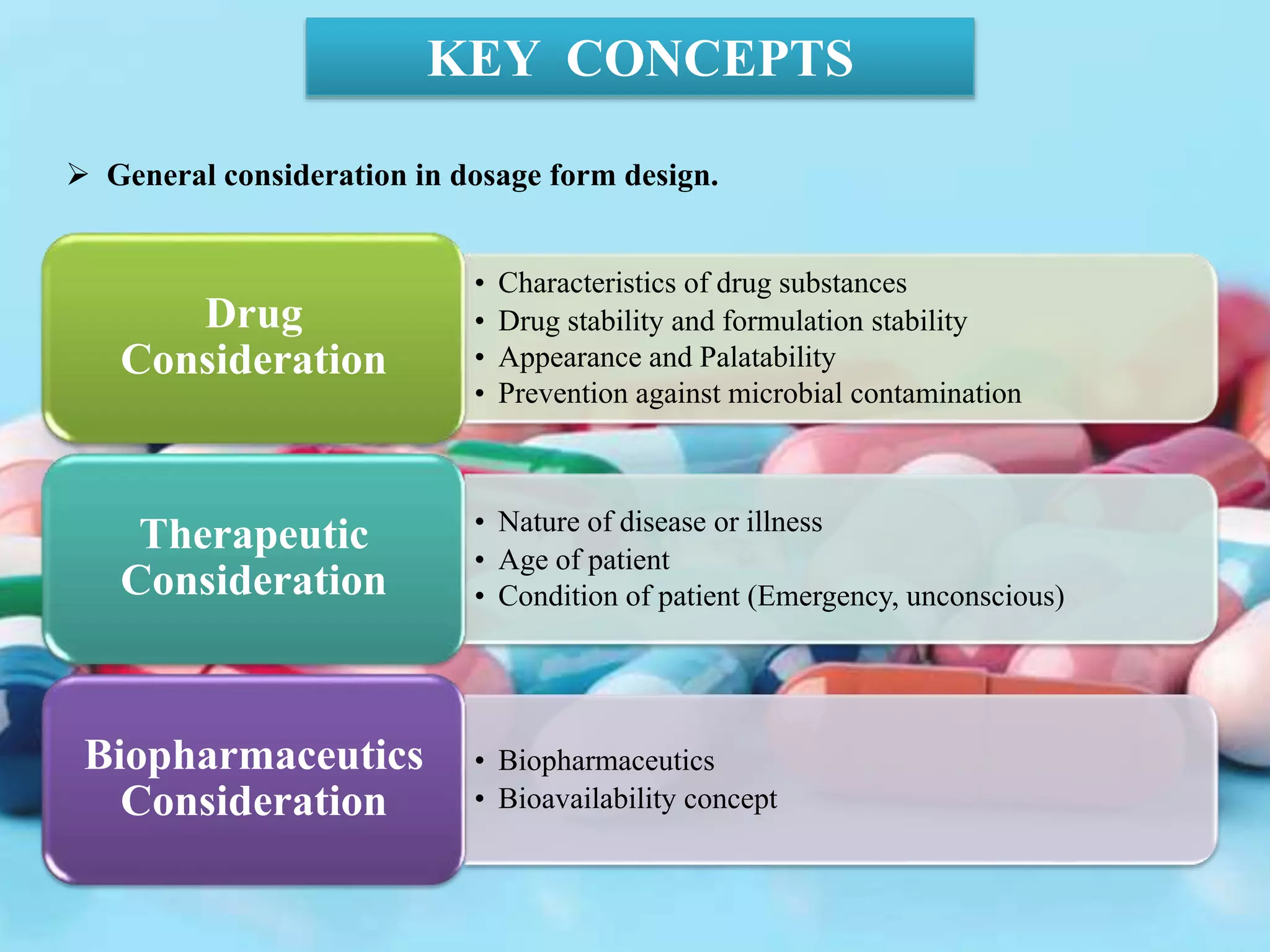
The document discusses various dosage forms and drug delivery systems. It begins by defining dosage forms as means of delivering drug molecules to sites of action. It then covers different types of solid, liquid, semi-solid, inhaled, rectal and vaginal dosage forms. Key points include classifications based on route of administration (oral, parenteral, etc.) and physical form (solid, liquid, semi-solid). Common examples are provided for different dosage forms like tablets, capsules, solutions, suspensions, creams, inhalers, and suppositories.