Embed presentation
Download to read offline


















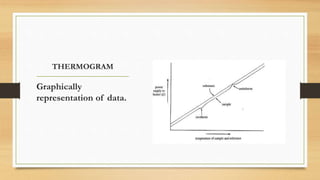


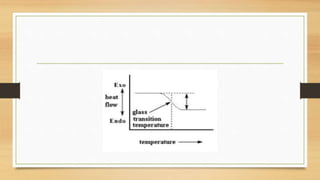

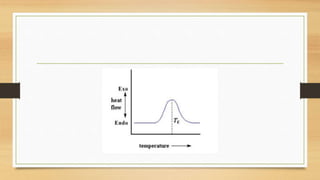

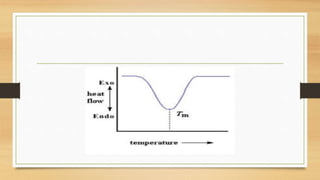

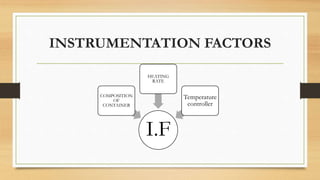









This presentation summarizes differential scanning calorimetry (DSC), which measures the heat flow into or out of a sample during heating or cooling. DSC can determine phase transitions like glass transitions, melting points, and crystallization temperatures. It works by heating a sample and reference simultaneously while measuring any heat differential. Factors like heating rate, sample size, and instrumentation can affect results. DSC is useful for characterizing polymers and other materials.




































