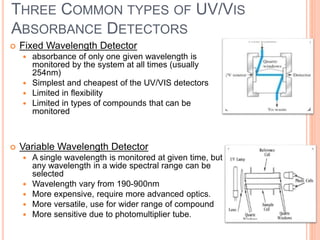
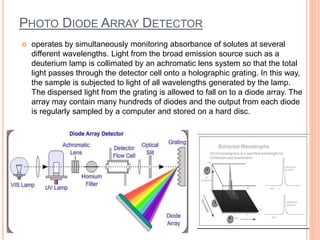
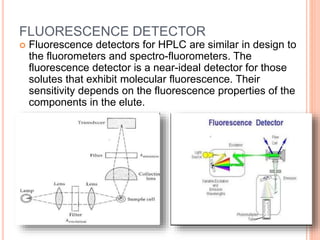
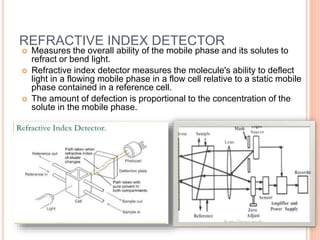
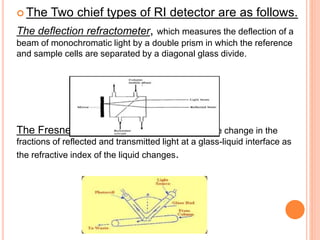
Detectors are used in HPLC to sense individual components as they leave the chromatography column. There are two main types of detectors: selective detectors that respond to a particular property of the solute and universal detectors that measure differences in bulk properties between the solute and mobile phase. Common detectors include UV-Vis absorbance, fluorescence, refractive index, electrochemical, and mass spectrometry detectors. An ideal detector has properties such as good stability, linear response, and high sensitivity.