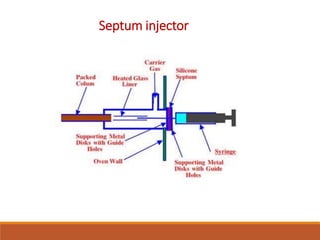
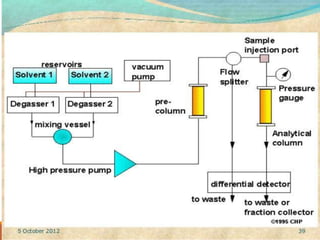
This document provides an overview of high performance liquid chromatography (HPLC). It discusses how HPLC refined traditional liquid chromatography by using smaller particle sizes, smaller column diameters, and high fluid pressures to provide enhanced separations over shorter periods of time. Key aspects of HPLC systems and processes are summarized, including the use of pumps to deliver mobile phases at high pressure through columns containing small stationary phase particles. Separation is achieved based on how sample components partition between the mobile and stationary phases. Various detectors are also outlined.