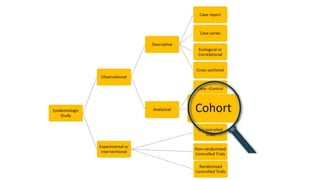
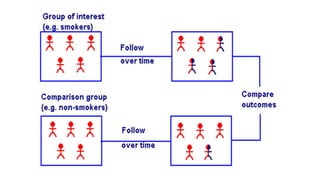
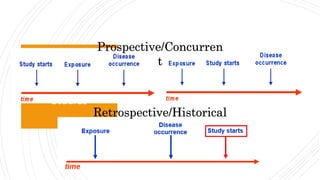
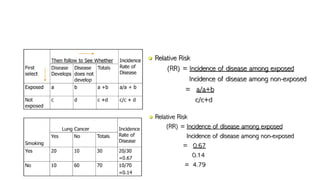
The document outlines the cohort study design, including its overview, advantages, disadvantages, and potential sources of bias. It details the steps in conducting a cohort study, such as selection of the study population, defining exposure groups, and determining outcomes. Examples illustrate the approach, emphasizing the importance of proper methodology to minimize bias and accurately assess health outcomes.