Classification of matter
•
1 like•2,081 views
Learn about the different Classifications and Properties of Matter.
Report
Share
Report
Share
Download to read offline
Recommended
Recommended
More Related Content
What's hot
What's hot (20)
Chemical bonds- Properties of Ionic and Covalent compounds

Chemical bonds- Properties of Ionic and Covalent compounds
Transfer of heat: Conduction & Convection - Class 7

Transfer of heat: Conduction & Convection - Class 7
Similar to Classification of matter
Similar to Classification of matter (20)
Topic 1 Mass relationship in chemistry (2021.01.14) (Updated).pdf

Topic 1 Mass relationship in chemistry (2021.01.14) (Updated).pdf
aaa. Chem 326L – Experiment 2 Solubility Kickoff.pptx

aaa. Chem 326L – Experiment 2 Solubility Kickoff.pptx
5 1.1 Matter Powerpoint Part A Classification Of Matter

5 1.1 Matter Powerpoint Part A Classification Of Matter
Unit b matter and chemical change notes (classifying matter)

Unit b matter and chemical change notes (classifying matter)
Recently uploaded
Intravital microscopy (IVM) is a powerful tool utilized to study cellular behavior over time and space in vivo. Much of our understanding of cell biology has been accomplished using various in vitro and ex vivo methods; however, these studies do not necessarily reflect the natural dynamics of biological processes. Unlike traditional cell culture or fixed tissue imaging, IVM allows for the ultra-fast high-resolution imaging of cellular processes over time and space and were studied in its natural environment. Real-time visualization of biological processes in the context of an intact organism helps maintain physiological relevance and provide insights into the progression of disease, response to treatments or developmental processes.
In this webinar we give an overview of advanced applications of the IVM system in preclinical research. IVIM technology is a provider of all-in-one intravital microscopy systems and solutions optimized for in vivo imaging of live animal models at sub-micron resolution. The system’s unique features and user-friendly software enables researchers to probe fast dynamic biological processes such as immune cell tracking, cell-cell interaction as well as vascularization and tumor metastasis with exceptional detail. This webinar will also give an overview of IVM being utilized in drug development, offering a view into the intricate interaction between drugs/nanoparticles and tissues in vivo and allows for the evaluation of therapeutic intervention in a variety of tissues and organs. This interdisciplinary collaboration continues to drive the advancements of novel therapeutic strategies.
(May 29th, 2024) Advancements in Intravital Microscopy- Insights for Preclini...

(May 29th, 2024) Advancements in Intravital Microscopy- Insights for Preclini...Scintica Instrumentation
Recently uploaded (20)
BLOOD AND BLOOD COMPONENT- introduction to blood physiology

BLOOD AND BLOOD COMPONENT- introduction to blood physiology
insect taxonomy importance systematics and classification

insect taxonomy importance systematics and classification
GEOLOGICAL FIELD REPORT On Kaptai Rangamati Road-Cut Section.pdf

GEOLOGICAL FIELD REPORT On Kaptai Rangamati Road-Cut Section.pdf
PRESENTATION ABOUT PRINCIPLE OF COSMATIC EVALUATION

PRESENTATION ABOUT PRINCIPLE OF COSMATIC EVALUATION
Gliese 12 b, a temperate Earth-sized planet at 12 parsecs discovered with TES...

Gliese 12 b, a temperate Earth-sized planet at 12 parsecs discovered with TES...
(May 29th, 2024) Advancements in Intravital Microscopy- Insights for Preclini...

(May 29th, 2024) Advancements in Intravital Microscopy- Insights for Preclini...
GLOBAL AND LOCAL SCENARIO OF FOOD AND NUTRITION.pptx

GLOBAL AND LOCAL SCENARIO OF FOOD AND NUTRITION.pptx
Cancer cell metabolism: special Reference to Lactate Pathway

Cancer cell metabolism: special Reference to Lactate Pathway
Predicting property prices with machine learning algorithms.pdf

Predicting property prices with machine learning algorithms.pdf
Classification of matter
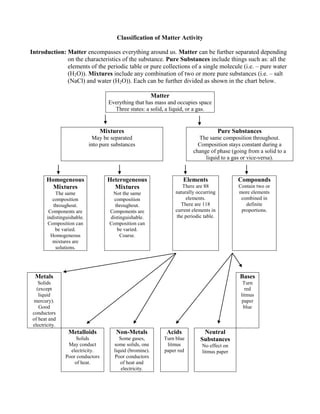
- 1. Classification of Matter Activity Introduction: Matter encompasses everything around us. Matter can be further separated depending on the characteristics of the substance. Pure Substances include things such as: all the elements of the periodic table or pure collections of a single molecule (i.e. – pure water (H2O)). Mixtures include any combination of two or more pure substances (i.e. – salt (NaCl) and water (H2O)). Each can be further divided as shown in the chart below. Matter Everything that has mass and occupies space Three states: a solid, a liquid, or a gas. Mixtures May be separated into pure substances Pure Substances The same composition throughout. Composition stays constant during a change of phase (going from a solid to a liquid to a gas or vice-versa). Homogeneous Mixtures The same composition throughout. Components are indistinguishable. Composition can be varied. Homogeneous mixtures are solutions. Heterogeneous Mixtures Not the same composition throughout. Components are distinguishable. Composition can be varied. Coarse. Elements There are 88 naturally occurring elements. There are 118 current elements in the periodic table. Compounds Contain two or more elements combined in definite proportions. Metals Solids (except liquid mercury). Good conductors of heat and electricity. Bases Turn red litmus paper blue Metalloids Solids May conduct electricity. Poor conductors of heat. Non-Metals Some gases, some solids, one liquid (bromine). Poor conductors of heat and electricity. Acids Turn blue litmus paper red Neutral Substances No effect on litmus paper
- 2. Purpose: This activity will allow you to classify matter as a Pure Substance or as a Mixture. If you classify the substance as a mixture, you can further classify the substance as a Homogeneous mixture or a Heterogeneous mixture. Materials: Ten vials of matter each containing ONLY: Cupric Acetate Cupric Acetate & Water ( & H2O) Distilled Water (H2O) Glucose (C6H12O6) Glucose & Water (C6H12O6 & H2O) Gravel & Water (Gravel & H2O) Gravel & Zinc (Gravel & Zn) Salt (NaCl & H2O) Salt & Water (NaCl & H2O) Starch & Water ((C6H11O5)n & H2O) Procedure: 1: Obtain one of the 10 vials listed above. 2: Look at the vial closely, read its contents, and decide whether the vial contains a Pure Substance or a Mixture. 3: Dependent on your answer to (2) above, decide whether the contents fall into one of the following three categories: Element/Compound, Homogeneous mixture or a Heterogeneous mixture. 4: Create a table similar to the one below to summarize your results for all ten vials. Include a column to give a reason for your placement of the contents of each vial. Give your table a title. 5: Repeat steps 1 to 3 for all 10 vials.
- 3. Title for Table: Vial Name Qualitative Observations (state, colour, etc) Pure Substance or Mixture Element/Compound or Homogeneous or Heterogeneous Reason for Classification 1: 2: 3: 4: 5:
- 4. 6: 7: 8: 9: 10: