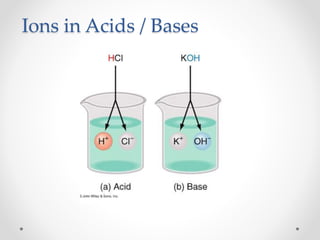
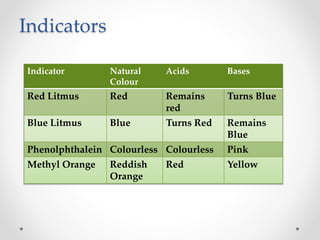
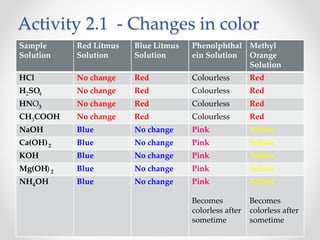
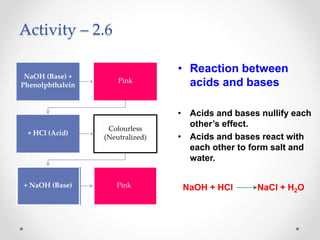
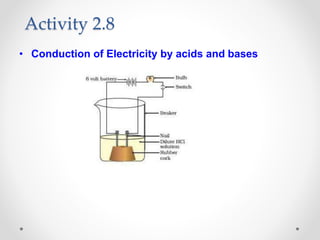
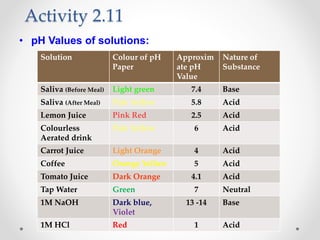
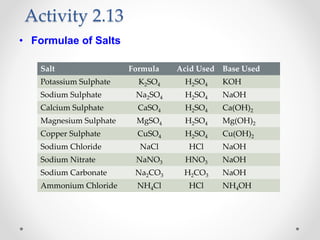
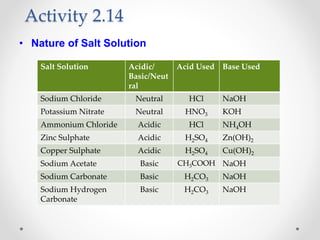
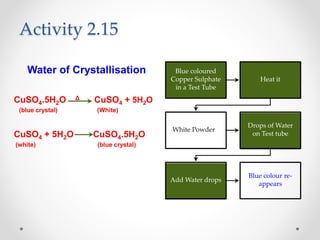
The document provides a comprehensive overview of acids, bases, and salts, detailing their properties, reactions, and various applications. It explains concepts such as ionization, pH scale, neutralization reactions, and the preparation of common compounds like sodium hydroxide and baking soda. Additionally, it includes activities demonstrating the chemical reactions involved and the use of indicators to classify substances as acidic or basic.