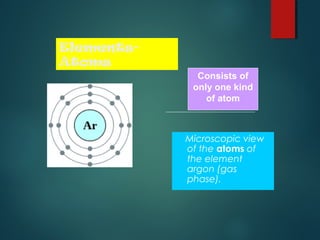
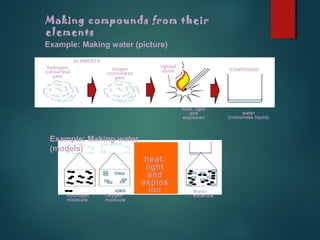
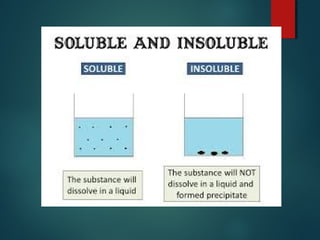
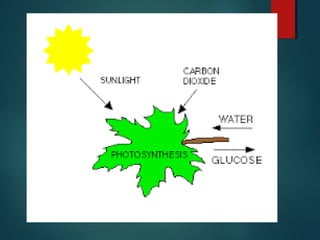
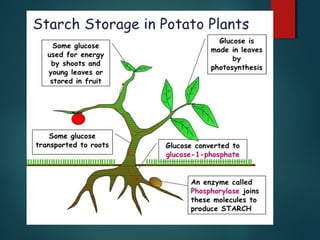
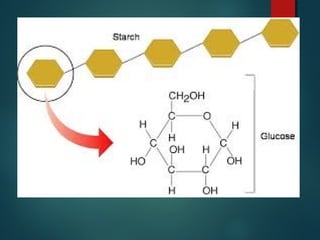
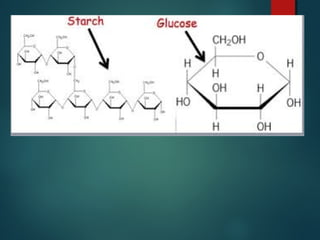
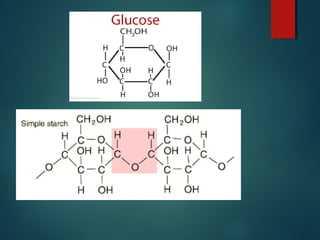
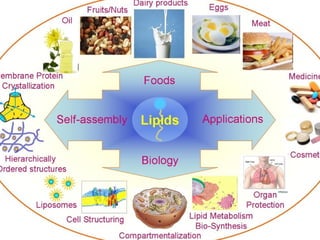
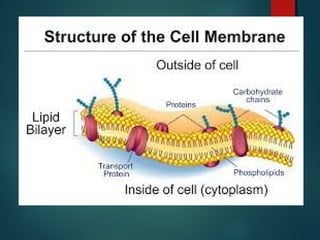
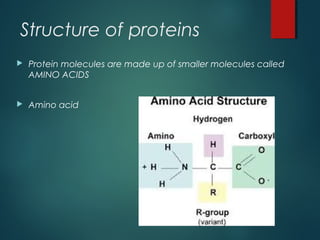
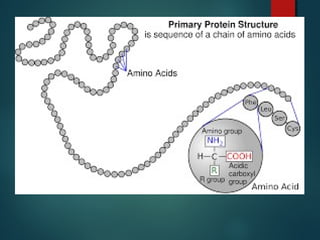
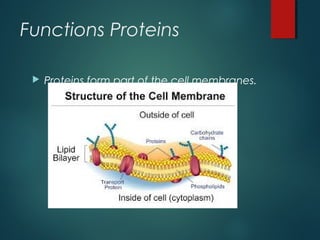
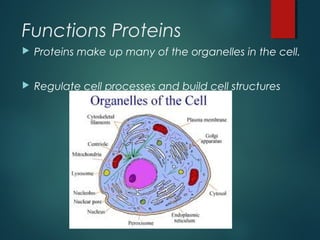
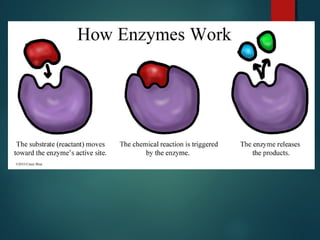
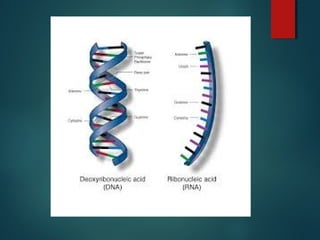
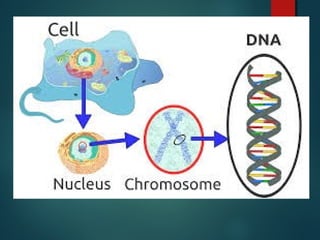
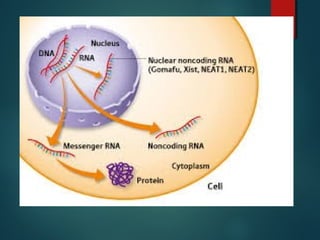
This document provides information about chemical compounds found in cells. It defines elements as substances made of only one type of atom that cannot be broken down further. Compounds contain two or more elements and have properties different from their components. The four main types of organic compounds found in cells are carbohydrates, lipids, proteins, and nucleic acids. Carbohydrates and lipids store energy, while proteins perform important functions and nucleic acids carry genetic information that directs cell activities.