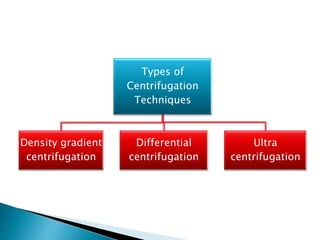
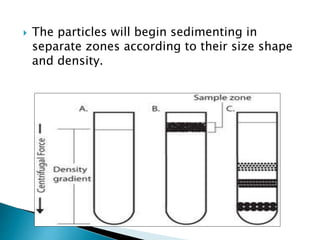
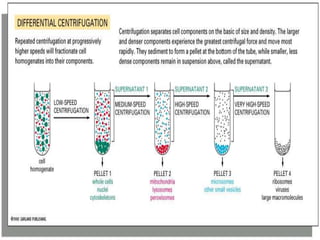
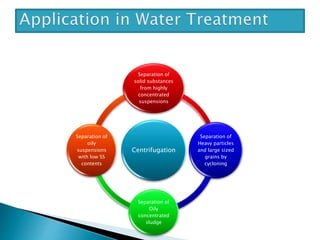
The document discusses centrifugation, including its definition, principles, classifications, construction, advantages/disadvantages, and applications. Centrifugation uses centrifugal force to separate mixtures based on density differences. It can separate components in industry, water treatment, and pharmaceutical/biological analysis. Centrifuges are classified by speed, temperature, and use. Common techniques include density gradient, differential, and ultra centrifugation. Industrial and laboratory centrifuges have various uses such as wastewater treatment and isolating cell components.