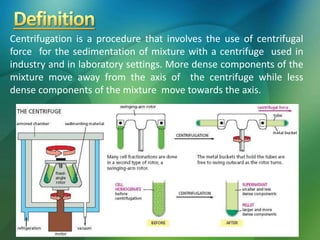
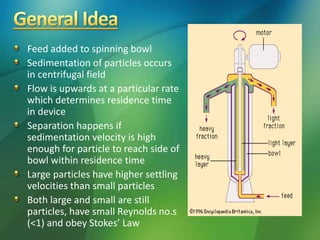
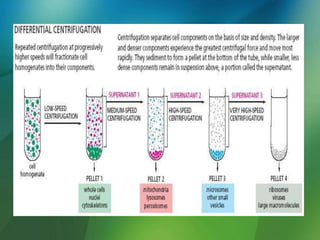
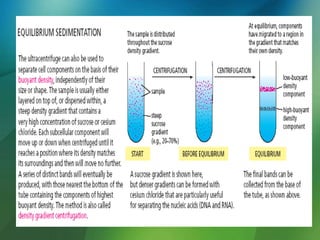
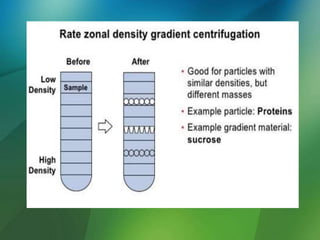
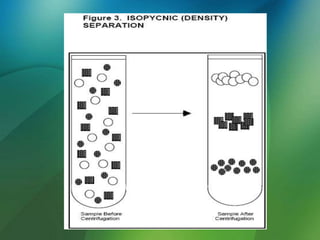
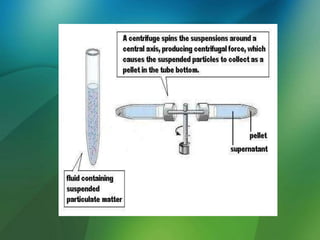
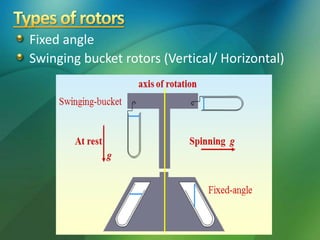
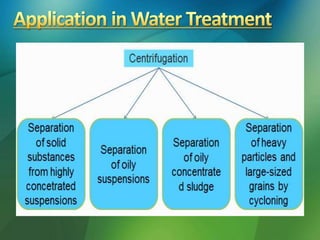
The document discusses centrifuges and centrifugation. It begins by summarizing the early history of centrifuges, including inventions in the 18th and 19th centuries. It then provides definitions and explanations of key terms like centrifuge, centrifugation, and relative centrifugal force. The rest of the document details different types of centrifuges, components of centrifuges, separation techniques, and rotors and tubes used in centrifugation.