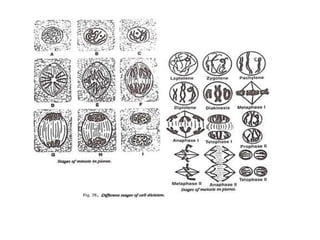
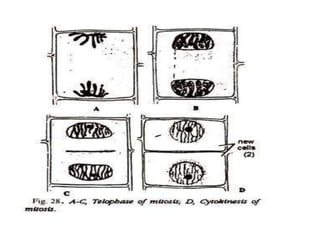
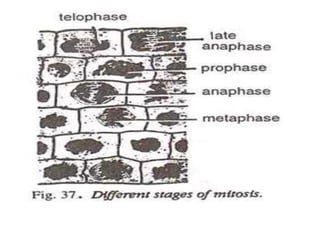
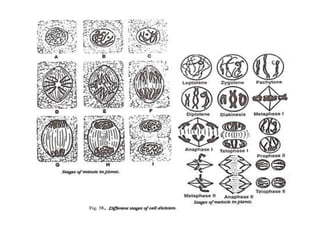
The document describes several experiments on cell biology involving plant and animal cells. The first experiment examines the effect of colchicine on onion root tip cells by treating them with a colchicine solution and observing abnormalities like polyploidy under a microscope. The second experiment prepares bone marrow squash from mice to observe the different stages of mitosis. The third experiment prepares slides of meiotic stages using plant materials like maize flowers. The remaining experiments involve preparing slides of root tip or bud cells using different staining techniques to observe mitosis or meiosis under a microscope.