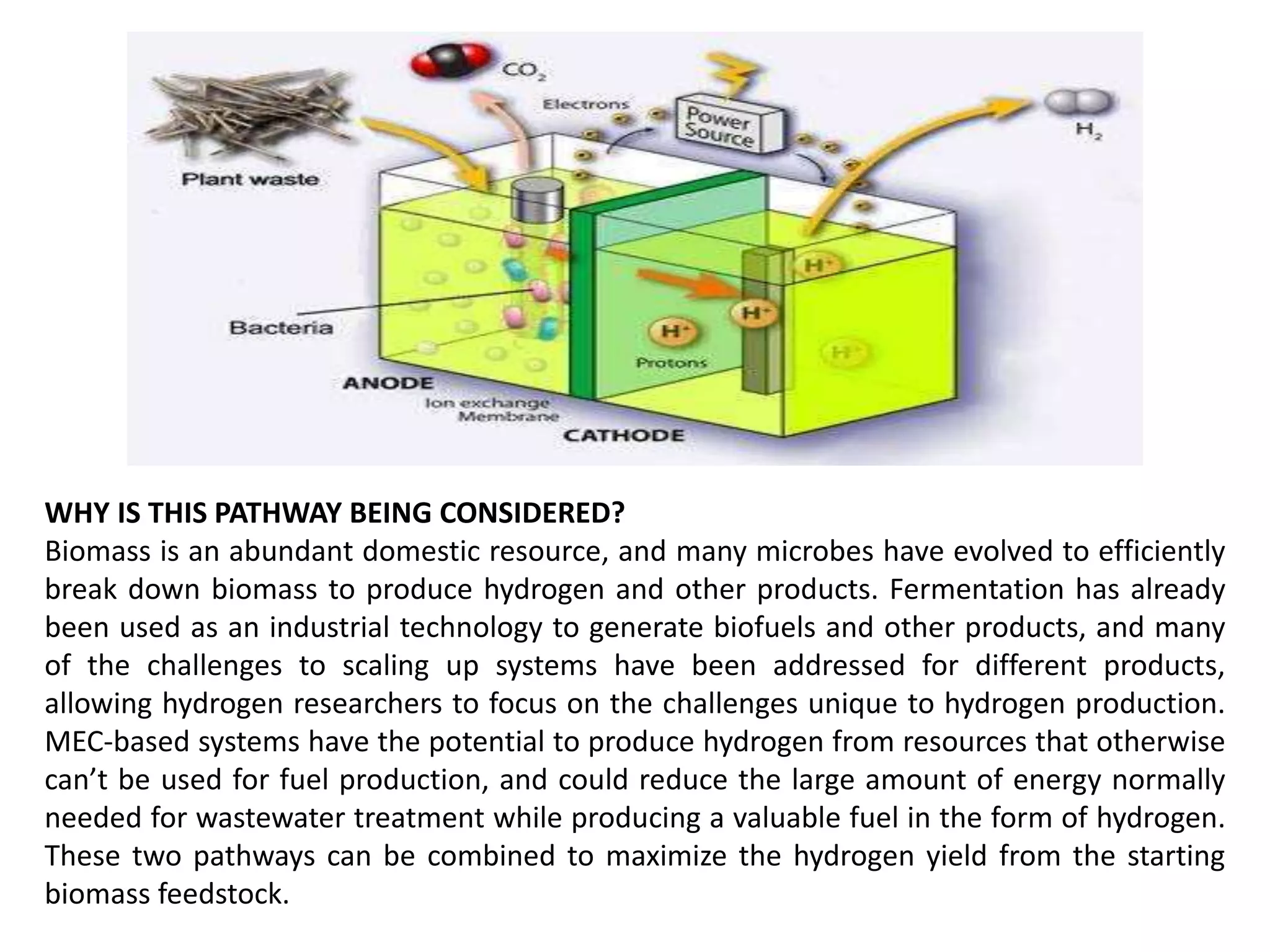
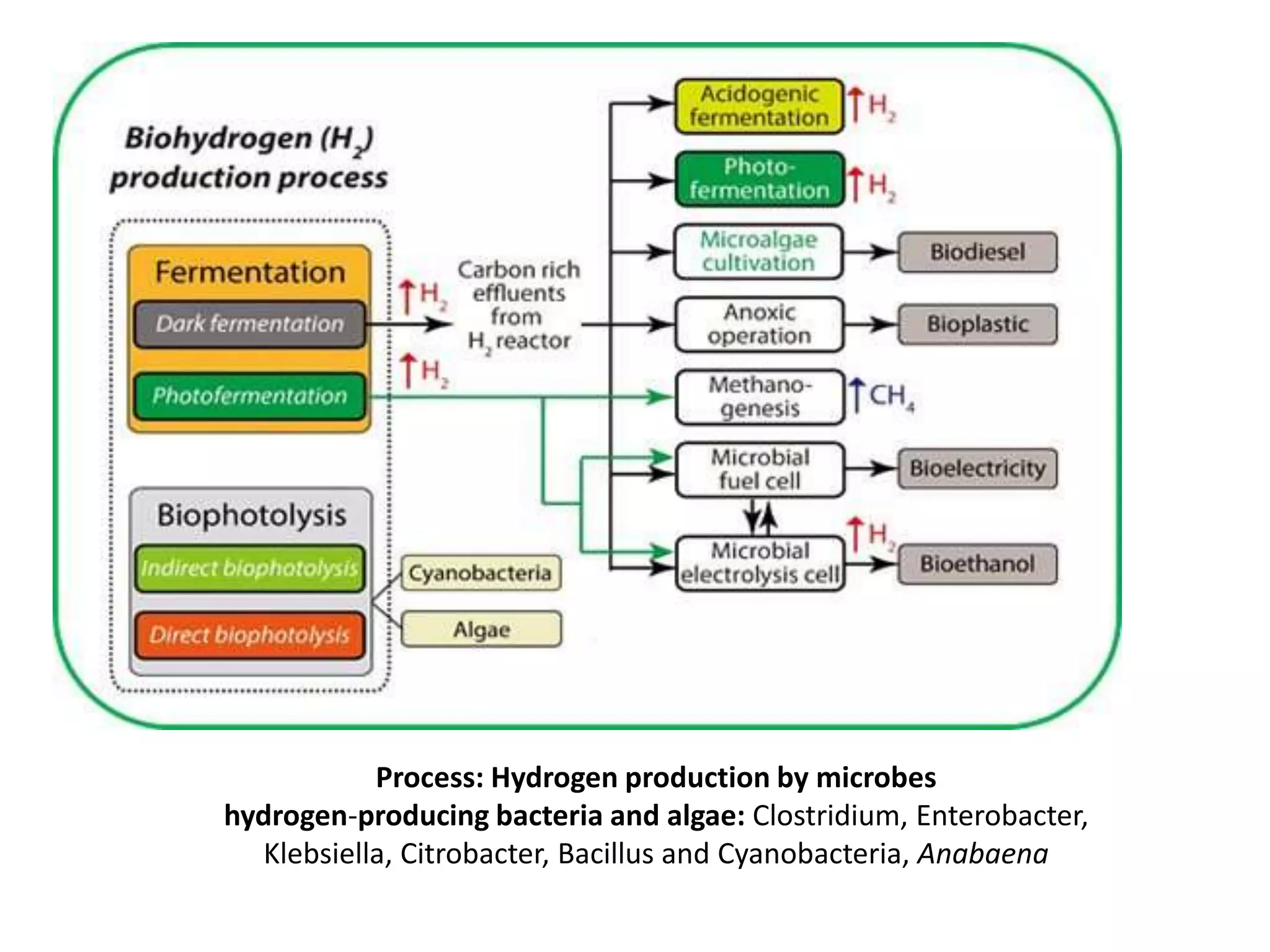
The document discusses hydrogen production through microbial biomass conversion, emphasizing the capabilities of microorganisms to digest biomass and generate hydrogen via fermentation and microbial electrolysis cells (MECs). It highlights strategies to optimize production rates and yields, as well as the potential for scaling up these technologies using various feedstocks. Additionally, specific examples of hydrogen-producing bacteria and research on enhancing efficiency and production methods are mentioned.