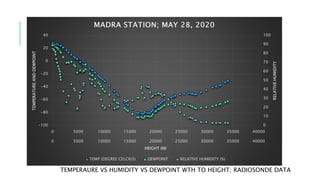
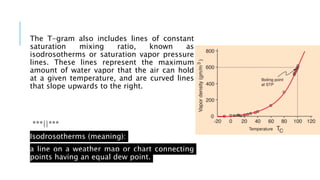
The document provides an overview of the laws of thermodynamics, detailing the zeroth through third laws and their implications, particularly in atmospheric thermodynamics. It explains how the first law is foundational for understanding energy conservation in open systems like the atmosphere, influencing weather patterns and phenomena such as cloud formation and temperature changes. Additionally, it covers the concepts of adiabatic lapse rates and the Clausius-Clapeyron equation, which are critical for meteorology and understanding the dynamics of temperature and vapor pressure in the atmosphere.