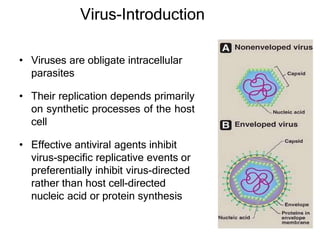
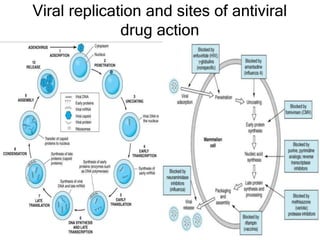
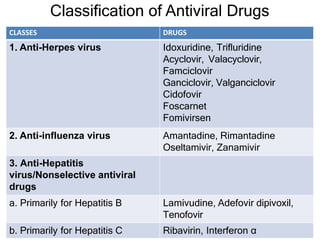
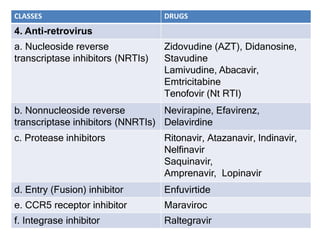
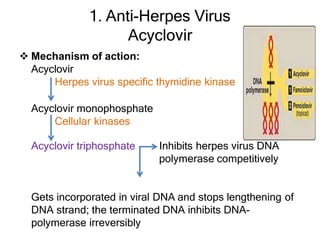
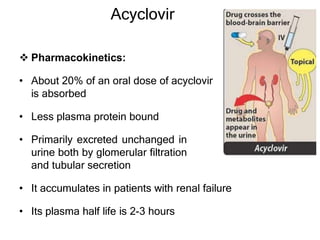
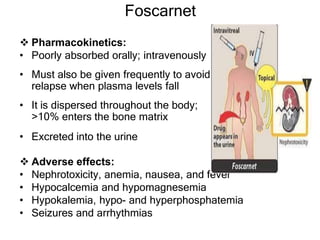
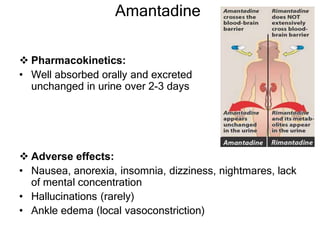
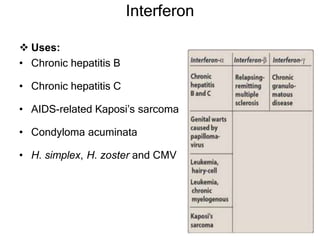
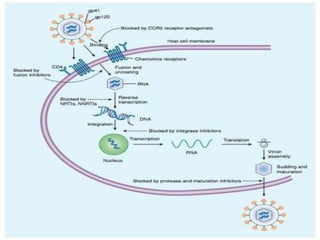
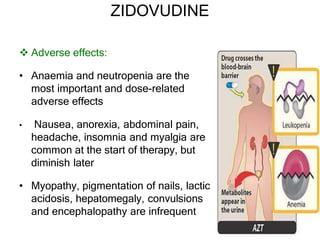
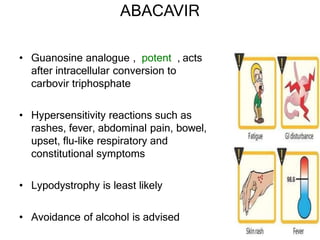
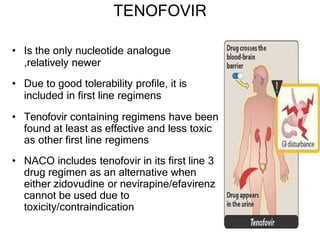
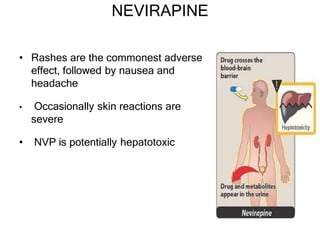
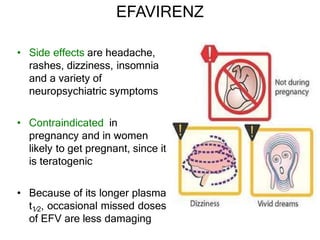
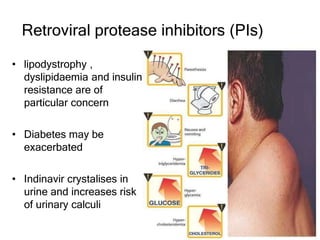
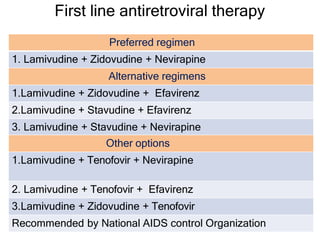
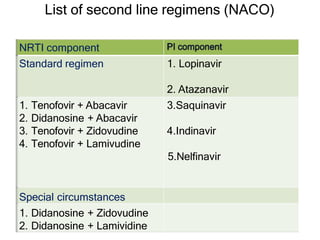
This document discusses various classes of antiviral drugs, including those that target herpes viruses, influenza virus, hepatitis viruses, and HIV. It provides details on the mechanism of action, pharmacokinetics, uses, and adverse effects of representative drugs in each class, such as acyclovir for herpes, amantadine/rimantadine for influenza, lamivudine/adefovir for hepatitis B, ribavirin/interferon for hepatitis C, and zidovudine as the prototype nucleoside reverse transcriptase inhibitor for HIV. The goal of antiviral treatment is to inhibit virus-specific replication events while minimizing effects on host cell processes.