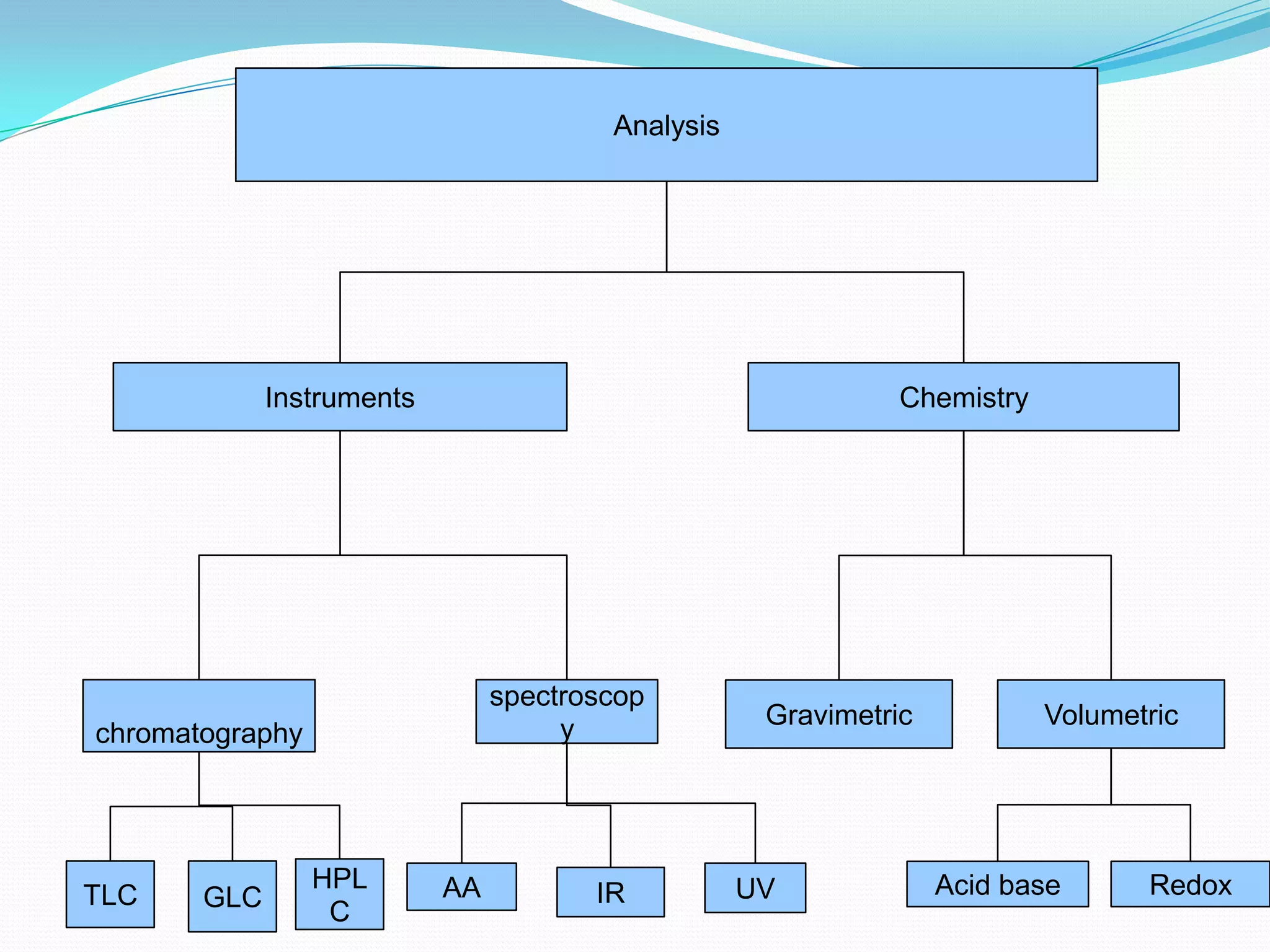
This document provides an overview of analytical chemistry, including qualitative and quantitative analysis techniques. It discusses various instruments and methods used in analytical chemistry, such as chromatography, spectroscopy, titration, and gravimetric analysis. Key concepts covered include units, molar mass, moles, stoichiometry, empirical formulas, and significant figures. Examples are provided to demonstrate calculations for composition percentages, empirical formulas, hydration states, and concentration determination through gravimetric analysis.