- Anaerobic bacteria require oxygen-free conditions to grow as oxygen is often toxic to them. There are different types including strict anaerobes, microaerophiles, aerotolerant anaerobes, and facultative anaerobes.
- Oxygen toxicity occurs as it is used in respiration, producing reactive oxygen species that can damage enzymes and cell structures in anaerobic bacteria. Special culturing techniques are needed to exclude oxygen like using vacuum, inert gas displacement, or chemical oxygen absorbers.
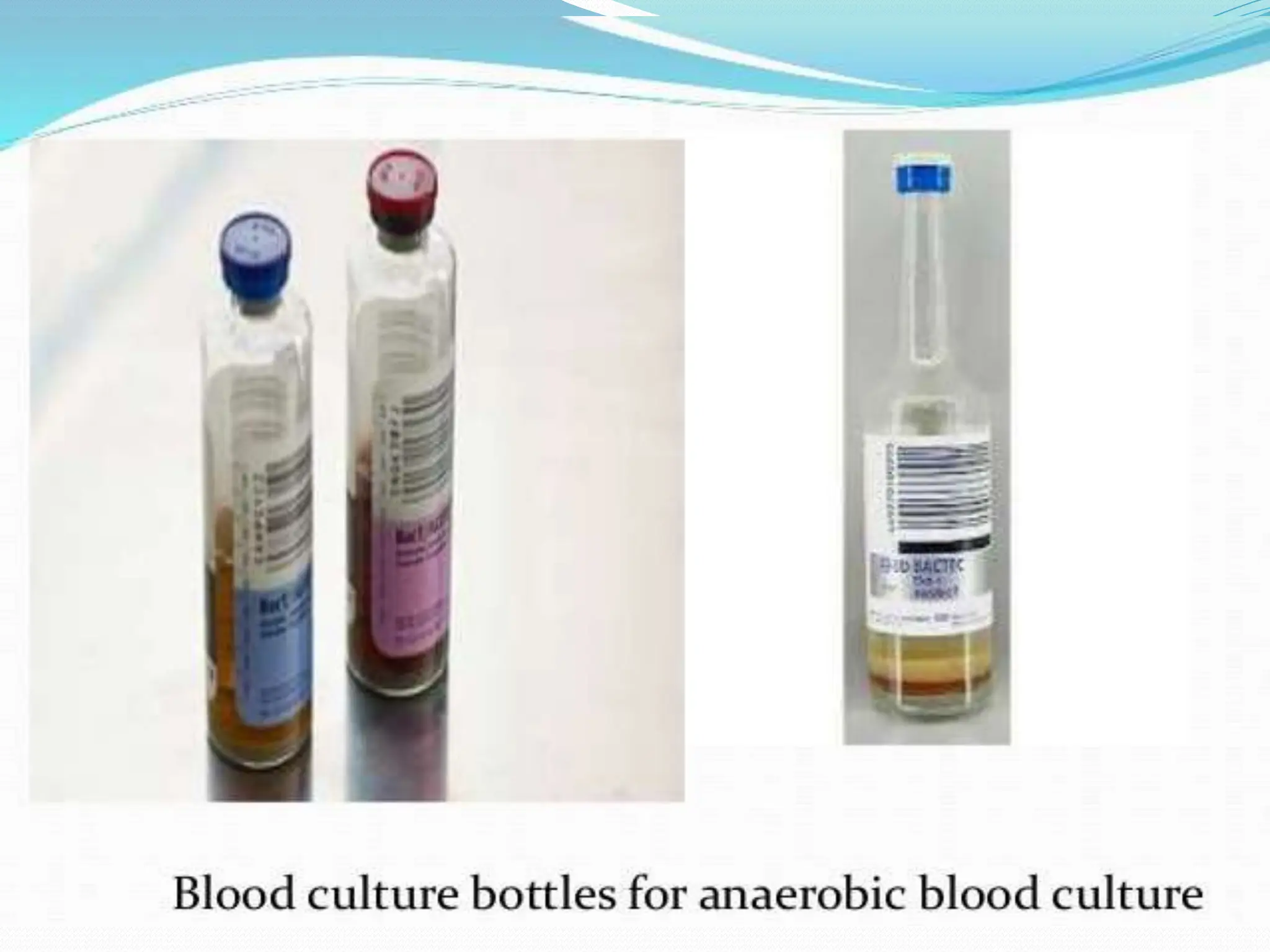
- Common culturing methods include the McIntosh jar using hydrogen gas and a catalyst to remove oxygen, or automated systems like the Anoxomat. Special reduced media and culturing