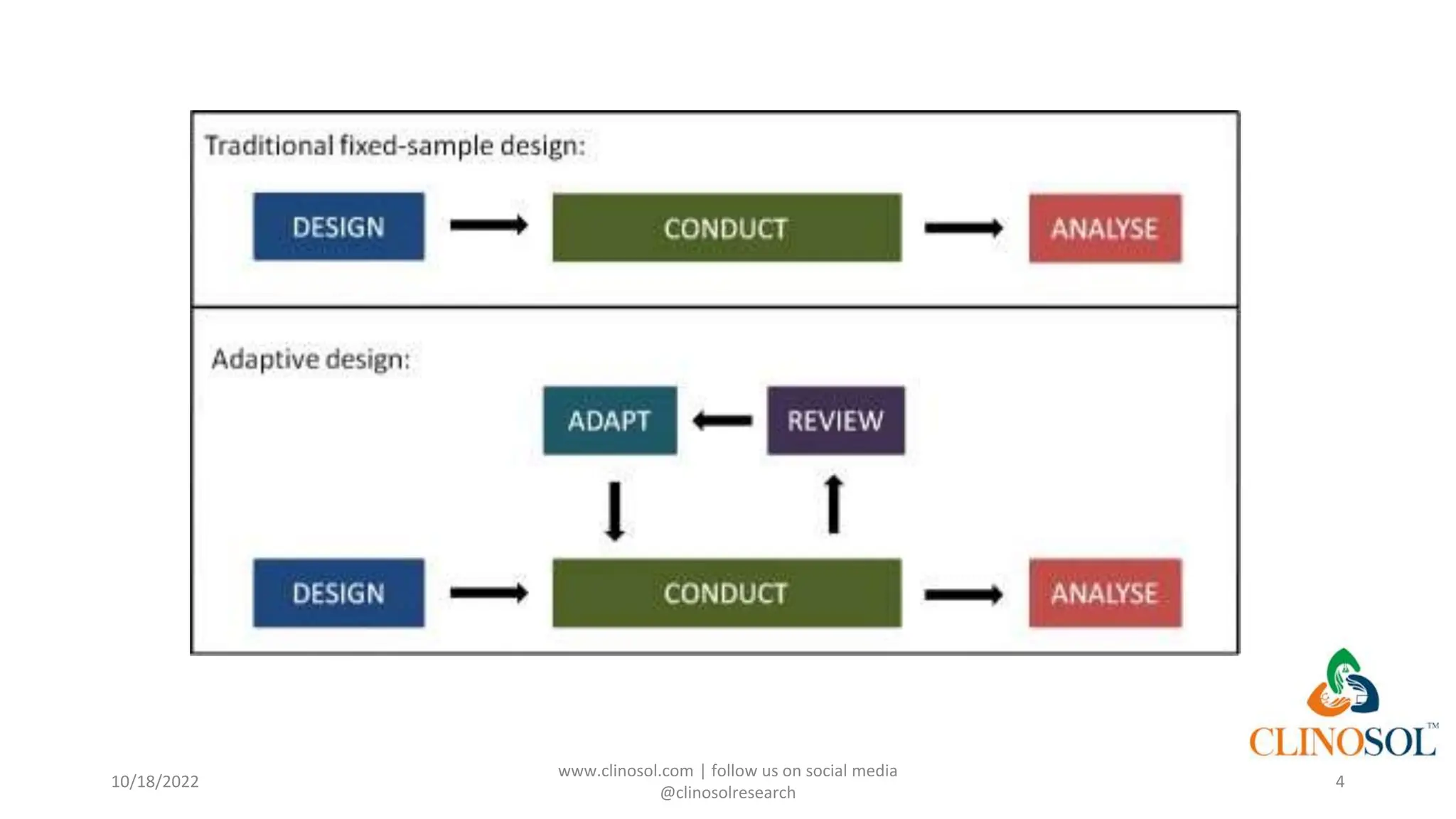
Adaptive clinical trials allow for modifications to trial design based on data collected during the study, enhancing flexibility and efficiency without compromising integrity. They can include various methodologies and regulatory acceptance, providing potential advantages like reduced resource waste and better allocation of participants. However, challenges such as the necessity for Bayesian statistical approaches and the risk of compromising study credibility due to ad hoc changes need to be considered.