Acid base table
•
1 like•3,175 views
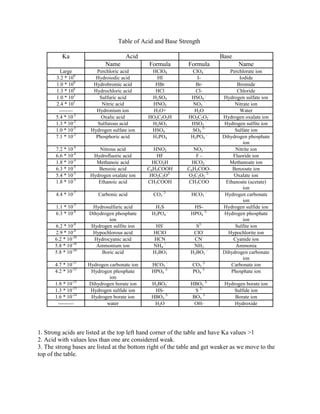
1. The document presents a table listing the acid dissociation constants (Ka) of various acids from strongest to weakest. 2. The strongest acids such as perchloric acid and hydroiodic acid have Ka values greater than 1 and are listed at the top left of the table. 3. Weaker acids with Ka values less than 1 such as sulfuric acid and nitric acid are also included.
Report
Share
Report
Share
Download to read offline
Recommended
Solid State

The document discusses various topics related to crystal structures including:
1. The seven possible shapes of unit cells - cubic, orthorhombic, rhombohedral, tetragonal, triclinic, hexagonal, monoclinic.
2. The four possible arrangements of spheres in unit cells - primitive, body centered, face centered, end centered.
3. Close packing of spheres in cubic unit cells and the coordination numbers and packing fractions for each arrangement.
4. Structures of ionic compounds like NaCl, ZnS, CaF2, and relationships between ion sizes and structures.
Recrystallisation

Recrystallization is a technique used to purify solids based on differences in their solubility, involving dissolving the impure solid in a hot solvent, filtering to remove insoluble materials, and obtaining pure crystals during controlled cooling as the solvent crystallizes out of solution. The process works best when an appropriate solvent is selected that dissolves the compound at high temperatures but causes it to crystallize upon cooling, allowing purification through multiple recrystallization attempts if needed.
Crystal structure

This document summarizes a seminar on crystal structures presented by Madhusmita Sethy at Dr. Hari Singh Gour Vishwavidyalaya University. It defines crystals as solids composed of atoms or molecules arranged periodically in three dimensions. Crystals are made up of basic building blocks including unit cells that repeat through translation, lattices consisting of periodic arrays of points, and motifs or basis of one or more atoms located in a specific way. The document outlines six major crystal systems and discusses crystal structures, Pauling's rules of crystal chemistry, polymorphism, and the significance of understanding crystal structures.
Common ion effect

This document is a chemistry assignment about applying the common ion effect to separate ionic compounds. It discusses how the common ion effect decreases the solubility of an ionic precipitate by adding a soluble compound that shares an ion with the precipitate. As an example, it explains that the solubility of calcium fluoride decreases if additional calcium ions are added from another source, since both share the calcium ion. The student then proposes using the common ion effect to separate lithium hydroxide by adding carbonate ions, which will form less soluble lithium carbonate precipitate.
Crystal structure

This is very basics of crystal structure ..with so many figures of examples...so that u will get proper idea about the crystal structure....thank u..
Triphenylmethane and derivatives

This document discusses triphenylmethane, which is a colorless solid hydrocarbon that serves as the basic skeleton for many synthetic dyes called triarylmethane dyes. It can be synthesized through various reactions including the Friedel-Crafts reaction of benzene and chloroform. As a triarylmethane, it is more acidic than other hydrocarbons due to delocalization of charge over three phenyl rings. Examples provided of triarylmethane dyes include bromocresol green and malachite green.
Preparation of tris(ethylenediamine)nickel(ii) chloride complex.

The document describes the procedure for preparing tris(ethylenediamine)nickel(II) chloride complex. Nickel chloride hexahydrate is dissolved in water and reacted with ethylenediamine solution. Acetone is added to precipitate the complex, which is then filtered, washed, dried and weighed to calculate the percentage yield. The nickel ion in the complex is in the +2 oxidation state and ethylenediamine acts as a chelating ligand, binding to nickel in two positions to form an octahedral complex that is optically active.
Structure and bonding of chemistry ppt

- Atoms consist of electrons surrounding a positively charged nucleus made up of protons and neutrons. Different types of atoms have different numbers of protons, electrons, and neutrons.
- There are two main types of chemical bonding: ionic bonding and covalent bonding. Ionic bonding involves the transfer of electrons between atoms to form oppositely charged ions that are attracted to each other. Covalent bonding involves the sharing of electrons between atoms.
- Covalent bonding allows for many different molecular structures and isomers. Molecules can be nonpolar or polar depending on electron distribution. Metals exhibit metallic bonding where electrons are delocalized and shared among many neighboring atoms. Hydrogen bonding is a weaker interaction between polar molecules that
Recommended
Solid State

The document discusses various topics related to crystal structures including:
1. The seven possible shapes of unit cells - cubic, orthorhombic, rhombohedral, tetragonal, triclinic, hexagonal, monoclinic.
2. The four possible arrangements of spheres in unit cells - primitive, body centered, face centered, end centered.
3. Close packing of spheres in cubic unit cells and the coordination numbers and packing fractions for each arrangement.
4. Structures of ionic compounds like NaCl, ZnS, CaF2, and relationships between ion sizes and structures.
Recrystallisation

Recrystallization is a technique used to purify solids based on differences in their solubility, involving dissolving the impure solid in a hot solvent, filtering to remove insoluble materials, and obtaining pure crystals during controlled cooling as the solvent crystallizes out of solution. The process works best when an appropriate solvent is selected that dissolves the compound at high temperatures but causes it to crystallize upon cooling, allowing purification through multiple recrystallization attempts if needed.
Crystal structure

This document summarizes a seminar on crystal structures presented by Madhusmita Sethy at Dr. Hari Singh Gour Vishwavidyalaya University. It defines crystals as solids composed of atoms or molecules arranged periodically in three dimensions. Crystals are made up of basic building blocks including unit cells that repeat through translation, lattices consisting of periodic arrays of points, and motifs or basis of one or more atoms located in a specific way. The document outlines six major crystal systems and discusses crystal structures, Pauling's rules of crystal chemistry, polymorphism, and the significance of understanding crystal structures.
Common ion effect

This document is a chemistry assignment about applying the common ion effect to separate ionic compounds. It discusses how the common ion effect decreases the solubility of an ionic precipitate by adding a soluble compound that shares an ion with the precipitate. As an example, it explains that the solubility of calcium fluoride decreases if additional calcium ions are added from another source, since both share the calcium ion. The student then proposes using the common ion effect to separate lithium hydroxide by adding carbonate ions, which will form less soluble lithium carbonate precipitate.
Crystal structure

This is very basics of crystal structure ..with so many figures of examples...so that u will get proper idea about the crystal structure....thank u..
Triphenylmethane and derivatives

This document discusses triphenylmethane, which is a colorless solid hydrocarbon that serves as the basic skeleton for many synthetic dyes called triarylmethane dyes. It can be synthesized through various reactions including the Friedel-Crafts reaction of benzene and chloroform. As a triarylmethane, it is more acidic than other hydrocarbons due to delocalization of charge over three phenyl rings. Examples provided of triarylmethane dyes include bromocresol green and malachite green.
Preparation of tris(ethylenediamine)nickel(ii) chloride complex.

The document describes the procedure for preparing tris(ethylenediamine)nickel(II) chloride complex. Nickel chloride hexahydrate is dissolved in water and reacted with ethylenediamine solution. Acetone is added to precipitate the complex, which is then filtered, washed, dried and weighed to calculate the percentage yield. The nickel ion in the complex is in the +2 oxidation state and ethylenediamine acts as a chelating ligand, binding to nickel in two positions to form an octahedral complex that is optically active.
Structure and bonding of chemistry ppt

- Atoms consist of electrons surrounding a positively charged nucleus made up of protons and neutrons. Different types of atoms have different numbers of protons, electrons, and neutrons.
- There are two main types of chemical bonding: ionic bonding and covalent bonding. Ionic bonding involves the transfer of electrons between atoms to form oppositely charged ions that are attracted to each other. Covalent bonding involves the sharing of electrons between atoms.
- Covalent bonding allows for many different molecular structures and isomers. Molecules can be nonpolar or polar depending on electron distribution. Metals exhibit metallic bonding where electrons are delocalized and shared among many neighboring atoms. Hydrogen bonding is a weaker interaction between polar molecules that
Point defect in solids

point defects are the irregularities from ideal arrangement around a point in a crystalline substance.
Phy351 ch 3

This document discusses crystal structure and properties of solids. It begins by defining long-range order and crystalline structure, noting that most metals, semiconductors, and some other materials have crystalline structure with atoms arranged in a repetitive grid pattern. It then discusses basic terms like unit cell and space lattice, and describes the seven crystal systems and 14 Bravais lattices. Common metallic crystal structures of body-centered cubic, face-centered cubic, and hexagonal close-packed are discussed. Finally, it touches on X-ray diffraction techniques used to analyze crystal structure.
Complexometric titration

INTRODUCTION,LIGAND AND ITS TYPES,DETECTION OF END POINT,TYPES OF COMPLEXOMETRIC TITRATIONS AND ITS APPLICATIONS
X ray spectroscopy. ppt 

This document provides an overview of X-ray spectroscopy techniques, including X-ray absorption and fluorescence. It discusses the production of X-rays, the principles of X-ray absorption spectroscopy and X-ray fluorescence spectroscopy, and their applications. Key topics covered include X-ray sources like X-ray tubes and synchrotrons, Beer's law and how it relates to X-ray absorption, X-ray absorption near edge structure (XANES) and extended X-ray absorption fine structure (EXAFS), and the use of these techniques in fields like chemistry, physics and materials science.
Grade 10 Organic Chemistry

1) Crude oil is a mixture of hydrocarbons formed from the remains of ancient plants and animals over millions of years under high pressure and temperature conditions underground.
2) Fractional distillation is used to separate crude oil into different hydrocarbon fractions like gasoline, kerosene and diesel in a fractionating column based on their boiling points.
3) Short hydrocarbon fractions are collected at the top of the fractionating column while longer fractions are collected at the bottom during the fractional distillation process.
Crystalline Solids

This document discusses different types of crystalline solids. It defines a crystalline solid as having a well-ordered structure with definite arrangements of particles. Crystalline solids are made up of repeating units called unit cells, which together form a crystal lattice. The document describes the different packing arrangements of particles in unit cells and classifies crystalline solids into four main types - ionic, covalent, metallic and molecular crystals - based on the type of bonding forces between particles. Each type of crystalline solid is characterized by distinct properties like melting point, conductivity, hardness and thermal stability.
Nmr spectroscopy of fluorine 19

This document discusses 19F NMR spectroscopy. It begins by explaining that 19F NMR can be used to identify fluorine-containing compounds and describes the nuclear properties of 19F that make it responsive to NMR measurements. It then discusses the reference standards used for 19F NMR, solvent effects, isotopic effects, coupling constants, and provides examples of spin systems and virtual coupling. The document also discusses organofluorine compounds and their applications as well as some environmental and health issues.
Anistropy

Anistropy is an important feature of the materials that needs to be kept in view for processing. The strength of materials may depend on the Anisotropic behavior of the material.
Noble Gases

The document summarizes the properties of the noble gases: helium, neon, argon, krypton, xenon, and radon. It provides their atomic numbers, melting and boiling points, densities, dates and discoverers of discovery, name origins, common uses, sources, and safety information. Some noble gases like xenon can form compounds while others like helium, neon and argon cannot be made into compounds.
Engineering Physics - Crystal structure - Dr. Victor Vedanayakam.S

Engineering Physics - Crystal structure - Dr. Victor Vedanayakam.SMadanapalle Institute of Technology and Science
The study of crystal geometry helps to understand the behaviour of solids and their
mechanical,
electrical,
magnetic
optical and
Metallurgical properties
Stereochemistry manik 2

Geometric isomerism of alkenes, cyclic compounds: cis-trans and (E)-(Z) system of
nomenclature
b) Conformational isomers: Open chain and cyclic system
c) Chirality of molecules: Enantiomers, diastereomers, racemic modification, Meso
compound, R & S configuration, sequence rule, Optical rotation
d) Asymmetric synthesis: Preparation of enantiomers by asymmetric synthesis & optical
resolution method
e) Stereo selective and stereo specific reaction
f) Pharmaceutical importance of studding stereochemistry
Complexometric Titration

more chemistry contents are available
1. pdf file on Termmate: https://www.termmate.com/rabia.aziz
2. YouTube: https://www.youtube.com/channel/UCKxWnNdskGHnZFS0h1QRTEA
3. Facebook: https://web.facebook.com/Chemist.Rabia.Aziz/
4. Blogger: https://chemistry-academy.blogspot.com/
EDTA Titration
Structure of Crystal Lattice - K Adithi Prabhu

The document discusses the structure of crystal lattices. It begins by defining a crystal as a solid whose constituents are arranged in a highly ordered microscopic structure called a crystal lattice. There are seven basic crystal systems based on symmetry elements. The document then discusses various types of unit cells including primitive and centered unit cells. It describes the 14 Bravais lattices and provides examples of body centered cubic, face centered cubic, and end centered cubic lattices. Finally, it summarizes the structures of sodium chloride and calcium fluoride crystals including their chemical formulas and coordination numbers.
Radio-chemical method Pdf

This document provides an overview of radiation and radioactive elements. It discusses the classification of radiation into ionizing (alpha, beta, gamma) and non-ionizing radiation. It lists many naturally occurring radioactive elements and their properties. The document describes different types of radioactive decay including alpha, beta, and gamma decay. It discusses applications of radioisotopes in medicine, industry, and research. Nuclear fusion and fission are also summarized. The document provides information on radioactivity, radiopharmaceuticals, and detection of radiation.
Statics presentation ppt(1)

This document provides an introduction to statistical mechanics and different types of statistics. It discusses classical statistics, which includes Maxwell-Boltzmann statistics, and quantum statistics, which includes Bose-Einstein (B-E) and Fermi-Dirac (F-D) statistics. Maxwell-Boltzmann statistics treats particles as distinguishable and applies to ideal gases, while B-E and F-D statistics treat particles as indistinguishable and apply to photons/bosons and electrons/fermions, respectively. The key differences between the statistics are whether particles can occupy the same state (B-E allows multiple occupancy, F-D allows only single occupancy) and the formulas that describe the most probable distribution of particles
MOLECULAR SPECTROSCOPY.pptx

This document provides an overview of molecular spectroscopy. It discusses Maxwell's theory of electromagnetic radiation and how spectroscopy can be divided into absorption and emission types. Key concepts in UV-visible spectroscopy are explained, including Lambert Beer's law, electronic transitions between orbitals, and factors that influence absorption maxima wavelengths. The document also covers infrared spectroscopy, discussing active and inactive molecular vibrations, different types of vibrations, and the fingerprint region.
Acid base titration

1) The document describes acid-base titration techniques, including defining terms like equivalence point and end point.
2) It discusses different types of titrations including strong acid-strong base, weak acid-strong base, and constructing titration curves.
3) Key points are made about calculating pH values before, at, and after the equivalence point for different titration scenarios. The document provides examples of constructing titration curves step-by-step.
Lect. 8. conductometric titration

Conductometric titration. various types, Applications of Conductometric titration and its advantages
Crystallization

Crystals are solid structures with repeating patterns of atoms or molecules. They consist of a lattice, which defines points in space that repeat periodically, and a motif or basis made of one or more atoms associated with each lattice point. Different crystal structures are defined by their lattice type (e.g. cubic, hexagonal) and motif. Common 3D crystal structures include simple cubic, body-centered cubic, and face-centered cubic lattices combined with different motifs like single atoms or groups of atoms. The document provides examples of crystal structures for various elements and discusses key properties like coordination number and atomic packing factor.
Nanosecond photolysis

Photolysis is a chemical process where molecules are broken down by light absorption. Flash photolysis is commonly used to study short-lived intermediates in photochemical reactions, employing a photolysis flash to initiate reactions followed by a monitoring flash to measure absorption spectra. To study processes in the nanosecond time range, lasers can be used to generate photolysis pulses less than 20 nanoseconds, allowing observation of excited singlet state lifetimes and other fast reactions. Laser flash photolysis systems employ a laser pulse to synchronize a photolysis spark and provide pulses to initiate reactions and monitor absorption on nanosecond timescales, enabling identification of transient intermediates and insight into fast reaction mechanisms.
Tabla de Ka y pKa 

Tabla de Ka y pKa nos muestra acidos con sus respectivas constantes de acides y pka's. En la tabla se ve el nombre del acido, su formula quimica, su acido conjugado su base conjugada, y la fuerza de estos.
Theme 5 inorganic chemistry 2

The document discusses different types of acids and salts. It describes oxacid acids as compounds formed from hydrogen, oxygen, and a non-metal element through the addition of water to an oxide. Hydracid acids are combinations of hydrogen and a non-metal. It provides examples of naming conventions for oxacid acids, hydracid acids, and the salts derived from them. Exercises are included for naming and writing formulas for various acids and salts.
More Related Content
What's hot
Point defect in solids

point defects are the irregularities from ideal arrangement around a point in a crystalline substance.
Phy351 ch 3

This document discusses crystal structure and properties of solids. It begins by defining long-range order and crystalline structure, noting that most metals, semiconductors, and some other materials have crystalline structure with atoms arranged in a repetitive grid pattern. It then discusses basic terms like unit cell and space lattice, and describes the seven crystal systems and 14 Bravais lattices. Common metallic crystal structures of body-centered cubic, face-centered cubic, and hexagonal close-packed are discussed. Finally, it touches on X-ray diffraction techniques used to analyze crystal structure.
Complexometric titration

INTRODUCTION,LIGAND AND ITS TYPES,DETECTION OF END POINT,TYPES OF COMPLEXOMETRIC TITRATIONS AND ITS APPLICATIONS
X ray spectroscopy. ppt 

This document provides an overview of X-ray spectroscopy techniques, including X-ray absorption and fluorescence. It discusses the production of X-rays, the principles of X-ray absorption spectroscopy and X-ray fluorescence spectroscopy, and their applications. Key topics covered include X-ray sources like X-ray tubes and synchrotrons, Beer's law and how it relates to X-ray absorption, X-ray absorption near edge structure (XANES) and extended X-ray absorption fine structure (EXAFS), and the use of these techniques in fields like chemistry, physics and materials science.
Grade 10 Organic Chemistry

1) Crude oil is a mixture of hydrocarbons formed from the remains of ancient plants and animals over millions of years under high pressure and temperature conditions underground.
2) Fractional distillation is used to separate crude oil into different hydrocarbon fractions like gasoline, kerosene and diesel in a fractionating column based on their boiling points.
3) Short hydrocarbon fractions are collected at the top of the fractionating column while longer fractions are collected at the bottom during the fractional distillation process.
Crystalline Solids

This document discusses different types of crystalline solids. It defines a crystalline solid as having a well-ordered structure with definite arrangements of particles. Crystalline solids are made up of repeating units called unit cells, which together form a crystal lattice. The document describes the different packing arrangements of particles in unit cells and classifies crystalline solids into four main types - ionic, covalent, metallic and molecular crystals - based on the type of bonding forces between particles. Each type of crystalline solid is characterized by distinct properties like melting point, conductivity, hardness and thermal stability.
Nmr spectroscopy of fluorine 19

This document discusses 19F NMR spectroscopy. It begins by explaining that 19F NMR can be used to identify fluorine-containing compounds and describes the nuclear properties of 19F that make it responsive to NMR measurements. It then discusses the reference standards used for 19F NMR, solvent effects, isotopic effects, coupling constants, and provides examples of spin systems and virtual coupling. The document also discusses organofluorine compounds and their applications as well as some environmental and health issues.
Anistropy

Anistropy is an important feature of the materials that needs to be kept in view for processing. The strength of materials may depend on the Anisotropic behavior of the material.
Noble Gases

The document summarizes the properties of the noble gases: helium, neon, argon, krypton, xenon, and radon. It provides their atomic numbers, melting and boiling points, densities, dates and discoverers of discovery, name origins, common uses, sources, and safety information. Some noble gases like xenon can form compounds while others like helium, neon and argon cannot be made into compounds.
Engineering Physics - Crystal structure - Dr. Victor Vedanayakam.S

Engineering Physics - Crystal structure - Dr. Victor Vedanayakam.SMadanapalle Institute of Technology and Science
The study of crystal geometry helps to understand the behaviour of solids and their
mechanical,
electrical,
magnetic
optical and
Metallurgical properties
Stereochemistry manik 2

Geometric isomerism of alkenes, cyclic compounds: cis-trans and (E)-(Z) system of
nomenclature
b) Conformational isomers: Open chain and cyclic system
c) Chirality of molecules: Enantiomers, diastereomers, racemic modification, Meso
compound, R & S configuration, sequence rule, Optical rotation
d) Asymmetric synthesis: Preparation of enantiomers by asymmetric synthesis & optical
resolution method
e) Stereo selective and stereo specific reaction
f) Pharmaceutical importance of studding stereochemistry
Complexometric Titration

more chemistry contents are available
1. pdf file on Termmate: https://www.termmate.com/rabia.aziz
2. YouTube: https://www.youtube.com/channel/UCKxWnNdskGHnZFS0h1QRTEA
3. Facebook: https://web.facebook.com/Chemist.Rabia.Aziz/
4. Blogger: https://chemistry-academy.blogspot.com/
EDTA Titration
Structure of Crystal Lattice - K Adithi Prabhu

The document discusses the structure of crystal lattices. It begins by defining a crystal as a solid whose constituents are arranged in a highly ordered microscopic structure called a crystal lattice. There are seven basic crystal systems based on symmetry elements. The document then discusses various types of unit cells including primitive and centered unit cells. It describes the 14 Bravais lattices and provides examples of body centered cubic, face centered cubic, and end centered cubic lattices. Finally, it summarizes the structures of sodium chloride and calcium fluoride crystals including their chemical formulas and coordination numbers.
Radio-chemical method Pdf

This document provides an overview of radiation and radioactive elements. It discusses the classification of radiation into ionizing (alpha, beta, gamma) and non-ionizing radiation. It lists many naturally occurring radioactive elements and their properties. The document describes different types of radioactive decay including alpha, beta, and gamma decay. It discusses applications of radioisotopes in medicine, industry, and research. Nuclear fusion and fission are also summarized. The document provides information on radioactivity, radiopharmaceuticals, and detection of radiation.
Statics presentation ppt(1)

This document provides an introduction to statistical mechanics and different types of statistics. It discusses classical statistics, which includes Maxwell-Boltzmann statistics, and quantum statistics, which includes Bose-Einstein (B-E) and Fermi-Dirac (F-D) statistics. Maxwell-Boltzmann statistics treats particles as distinguishable and applies to ideal gases, while B-E and F-D statistics treat particles as indistinguishable and apply to photons/bosons and electrons/fermions, respectively. The key differences between the statistics are whether particles can occupy the same state (B-E allows multiple occupancy, F-D allows only single occupancy) and the formulas that describe the most probable distribution of particles
MOLECULAR SPECTROSCOPY.pptx

This document provides an overview of molecular spectroscopy. It discusses Maxwell's theory of electromagnetic radiation and how spectroscopy can be divided into absorption and emission types. Key concepts in UV-visible spectroscopy are explained, including Lambert Beer's law, electronic transitions between orbitals, and factors that influence absorption maxima wavelengths. The document also covers infrared spectroscopy, discussing active and inactive molecular vibrations, different types of vibrations, and the fingerprint region.
Acid base titration

1) The document describes acid-base titration techniques, including defining terms like equivalence point and end point.
2) It discusses different types of titrations including strong acid-strong base, weak acid-strong base, and constructing titration curves.
3) Key points are made about calculating pH values before, at, and after the equivalence point for different titration scenarios. The document provides examples of constructing titration curves step-by-step.
Lect. 8. conductometric titration

Conductometric titration. various types, Applications of Conductometric titration and its advantages
Crystallization

Crystals are solid structures with repeating patterns of atoms or molecules. They consist of a lattice, which defines points in space that repeat periodically, and a motif or basis made of one or more atoms associated with each lattice point. Different crystal structures are defined by their lattice type (e.g. cubic, hexagonal) and motif. Common 3D crystal structures include simple cubic, body-centered cubic, and face-centered cubic lattices combined with different motifs like single atoms or groups of atoms. The document provides examples of crystal structures for various elements and discusses key properties like coordination number and atomic packing factor.
Nanosecond photolysis

Photolysis is a chemical process where molecules are broken down by light absorption. Flash photolysis is commonly used to study short-lived intermediates in photochemical reactions, employing a photolysis flash to initiate reactions followed by a monitoring flash to measure absorption spectra. To study processes in the nanosecond time range, lasers can be used to generate photolysis pulses less than 20 nanoseconds, allowing observation of excited singlet state lifetimes and other fast reactions. Laser flash photolysis systems employ a laser pulse to synchronize a photolysis spark and provide pulses to initiate reactions and monitor absorption on nanosecond timescales, enabling identification of transient intermediates and insight into fast reaction mechanisms.
What's hot (20)
Engineering Physics - Crystal structure - Dr. Victor Vedanayakam.S

Engineering Physics - Crystal structure - Dr. Victor Vedanayakam.S
Similar to Acid base table
Tabla de Ka y pKa 

Tabla de Ka y pKa nos muestra acidos con sus respectivas constantes de acides y pka's. En la tabla se ve el nombre del acido, su formula quimica, su acido conjugado su base conjugada, y la fuerza de estos.
Theme 5 inorganic chemistry 2

The document discusses different types of acids and salts. It describes oxacid acids as compounds formed from hydrogen, oxygen, and a non-metal element through the addition of water to an oxide. Hydracid acids are combinations of hydrogen and a non-metal. It provides examples of naming conventions for oxacid acids, hydracid acids, and the salts derived from them. Exercises are included for naming and writing formulas for various acids and salts.
Ion list

This document lists common polyatomic ions, monatomic ions, and metals that can form multiple ions. It includes ions such as ammonium, hydroxide, cyanide, acetate, magnesium, calcium, iron, copper, and tin. It also notes the classical and stock names for metal ions like ferrous, ferric, cuprous, and cupric.
Blog.7 2.Acid And Base

The document defines acids and bases according to Arrhenius and Brønsted-Lowry theories. Acids are substances that produce hydrogen (H+) or hydronium (H3O+) ions in water according to Arrhenius, and are proton donors according to Brønsted-Lowry. Bases are substances that produce hydroxide (OH-) ions in water according to Arrhenius, and are proton acceptors according to Brønsted-Lowry. Conjugate acid-base pairs are also defined. Common acids, bases, and salts are listed along with their formulas and IUPAC names.
Chemical

The document lists the common, commercial, chemical and formula names of 22 compounds. It provides this identifying information in a table with 3 columns - the commercial name, chemical name and chemical formula of each compound. Some of the compounds listed include baking soda, bleaching powder, caustic soda, blue vitriol, chalk, gypsum, green vitriol, dry ice, white vitriol, carbolic acid, alcohol, bauxite, washing soda, acid salt, lauching gas, lime water, lime, oil of vitriol, lime stone, silica, T.N.T and vinegar.
Covalent bonds ch 9 2

This document provides information on naming binary molecular compounds and acids. It explains that binary molecular compounds contain two nonmetal elements, and the name indicates the elements and number of atoms using prefixes and suffixes like -ide. Acids are compounds that release hydrogen ions (H+) in water. Binary acids contain hydrogen and one other element, named with prefixes like hydro-, while oxyacids contain an oxyanion like nitrate and are named with suffixes like -ic or -ous acid. The document gives examples of naming compounds from their formulas and vice versa.
Química. Alejandra Cevallos 

This document contains a chemistry lesson on acids and their nomenclature in Spanish. It provides examples of common acids like nitric acid, sulfuric acid, and phosphoric acid along with their systematic, traditional, and other names in Spanish. It also gives formulas for compounds like selenious acid, boric acid, gold sulfide hydroxide, and cadmium zirconium sulfide.
Similar to Acid base table (7)
Recently uploaded
PCOS corelations and management through Ayurveda.

This presentation includes basic of PCOS their pathology and treatment and also Ayurveda correlation of PCOS and Ayurvedic line of treatment mentioned in classics.
RPMS TEMPLATE FOR SCHOOL YEAR 2023-2024 FOR TEACHER 1 TO TEACHER 3

RPMS Template 2023-2024 by: Irene S. Rueco
Your Skill Boost Masterclass: Strategies for Effective Upskilling

Your Skill Boost Masterclass: Strategies for Effective UpskillingExcellence Foundation for South Sudan
Strategies for Effective Upskilling is a presentation by Chinwendu Peace in a Your Skill Boost Masterclass organisation by the Excellence Foundation for South Sudan on 08th and 09th June 2024 from 1 PM to 3 PM on each day.BÀI TẬP BỔ TRỢ TIẾNG ANH 8 CẢ NĂM - GLOBAL SUCCESS - NĂM HỌC 2023-2024 (CÓ FI...

BÀI TẬP BỔ TRỢ TIẾNG ANH 8 CẢ NĂM - GLOBAL SUCCESS - NĂM HỌC 2023-2024 (CÓ FI...Nguyen Thanh Tu Collection
https://app.box.com/s/y977uz6bpd3af4qsebv7r9b7s21935vdবাংলাদেশ অর্থনৈতিক সমীক্ষা (Economic Review) ২০২৪ UJS App.pdf

বাংলাদেশের অর্থনৈতিক সমীক্ষা ২০২৪ [Bangladesh Economic Review 2024 Bangla.pdf] কম্পিউটার , ট্যাব ও স্মার্ট ফোন ভার্সন সহ সম্পূর্ণ বাংলা ই-বুক বা pdf বই " সুচিপত্র ...বুকমার্ক মেনু 🔖 ও হাইপার লিংক মেনু 📝👆 যুক্ত ..
আমাদের সবার জন্য খুব খুব গুরুত্বপূর্ণ একটি বই ..বিসিএস, ব্যাংক, ইউনিভার্সিটি ভর্তি ও যে কোন প্রতিযোগিতা মূলক পরীক্ষার জন্য এর খুব ইম্পরট্যান্ট একটি বিষয় ...তাছাড়া বাংলাদেশের সাম্প্রতিক যে কোন ডাটা বা তথ্য এই বইতে পাবেন ...
তাই একজন নাগরিক হিসাবে এই তথ্য গুলো আপনার জানা প্রয়োজন ...।
বিসিএস ও ব্যাংক এর লিখিত পরীক্ষা ...+এছাড়া মাধ্যমিক ও উচ্চমাধ্যমিকের স্টুডেন্টদের জন্য অনেক কাজে আসবে ...
Chapter 4 - Islamic Financial Institutions in Malaysia.pptx

Chapter 4 - Islamic Financial Institutions in Malaysia.pptxMohd Adib Abd Muin, Senior Lecturer at Universiti Utara Malaysia
This slide is special for master students (MIBS & MIFB) in UUM. Also useful for readers who are interested in the topic of contemporary Islamic banking.
The simplified electron and muon model, Oscillating Spacetime: The Foundation...

Discover the Simplified Electron and Muon Model: A New Wave-Based Approach to Understanding Particles delves into a groundbreaking theory that presents electrons and muons as rotating soliton waves within oscillating spacetime. Geared towards students, researchers, and science buffs, this book breaks down complex ideas into simple explanations. It covers topics such as electron waves, temporal dynamics, and the implications of this model on particle physics. With clear illustrations and easy-to-follow explanations, readers will gain a new outlook on the universe's fundamental nature.
Executive Directors Chat Leveraging AI for Diversity, Equity, and Inclusion

Let’s explore the intersection of technology and equity in the final session of our DEI series. Discover how AI tools, like ChatGPT, can be used to support and enhance your nonprofit's DEI initiatives. Participants will gain insights into practical AI applications and get tips for leveraging technology to advance their DEI goals.
Advanced Java[Extra Concepts, Not Difficult].docx![Advanced Java[Extra Concepts, Not Difficult].docx](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![Advanced Java[Extra Concepts, Not Difficult].docx](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
This is part 2 of my Java Learning Journey. This contains Hashing, ArrayList, LinkedList, Date and Time Classes, Calendar Class and more.
How to Build a Module in Odoo 17 Using the Scaffold Method

Odoo provides an option for creating a module by using a single line command. By using this command the user can make a whole structure of a module. It is very easy for a beginner to make a module. There is no need to make each file manually. This slide will show how to create a module using the scaffold method.
Main Java[All of the Base Concepts}.docx

This is part 1 of my Java Learning Journey. This Contains Custom methods, classes, constructors, packages, multithreading , try- catch block, finally block and more.
Walmart Business+ and Spark Good for Nonprofits.pdf

"Learn about all the ways Walmart supports nonprofit organizations.
You will hear from Liz Willett, the Head of Nonprofits, and hear about what Walmart is doing to help nonprofits, including Walmart Business and Spark Good. Walmart Business+ is a new offer for nonprofits that offers discounts and also streamlines nonprofits order and expense tracking, saving time and money.
The webinar may also give some examples on how nonprofits can best leverage Walmart Business+.
The event will cover the following::
Walmart Business + (https://business.walmart.com/plus) is a new shopping experience for nonprofits, schools, and local business customers that connects an exclusive online shopping experience to stores. Benefits include free delivery and shipping, a 'Spend Analytics” feature, special discounts, deals and tax-exempt shopping.
Special TechSoup offer for a free 180 days membership, and up to $150 in discounts on eligible orders.
Spark Good (walmart.com/sparkgood) is a charitable platform that enables nonprofits to receive donations directly from customers and associates.
Answers about how you can do more with Walmart!"
The Diamonds of 2023-2024 in the IGRA collection

A review of the growth of the Israel Genealogy Research Association Database Collection for the last 12 months. Our collection is now passed the 3 million mark and still growing. See which archives have contributed the most. See the different types of records we have, and which years have had records added. You can also see what we have for the future.
Pollock and Snow "DEIA in the Scholarly Landscape, Session One: Setting Expec...

Pollock and Snow "DEIA in the Scholarly Landscape, Session One: Setting Expec...National Information Standards Organization (NISO)
This presentation was provided by Steph Pollock of The American Psychological Association’s Journals Program, and Damita Snow, of The American Society of Civil Engineers (ASCE), for the initial session of NISO's 2024 Training Series "DEIA in the Scholarly Landscape." Session One: 'Setting Expectations: a DEIA Primer,' was held June 6, 2024.Recently uploaded (20)
RPMS TEMPLATE FOR SCHOOL YEAR 2023-2024 FOR TEACHER 1 TO TEACHER 3

RPMS TEMPLATE FOR SCHOOL YEAR 2023-2024 FOR TEACHER 1 TO TEACHER 3
Your Skill Boost Masterclass: Strategies for Effective Upskilling

Your Skill Boost Masterclass: Strategies for Effective Upskilling
BÀI TẬP BỔ TRỢ TIẾNG ANH 8 CẢ NĂM - GLOBAL SUCCESS - NĂM HỌC 2023-2024 (CÓ FI...

BÀI TẬP BỔ TRỢ TIẾNG ANH 8 CẢ NĂM - GLOBAL SUCCESS - NĂM HỌC 2023-2024 (CÓ FI...
বাংলাদেশ অর্থনৈতিক সমীক্ষা (Economic Review) ২০২৪ UJS App.pdf

বাংলাদেশ অর্থনৈতিক সমীক্ষা (Economic Review) ২০২৪ UJS App.pdf
Chapter 4 - Islamic Financial Institutions in Malaysia.pptx

Chapter 4 - Islamic Financial Institutions in Malaysia.pptx
The simplified electron and muon model, Oscillating Spacetime: The Foundation...

The simplified electron and muon model, Oscillating Spacetime: The Foundation...
Pride Month Slides 2024 David Douglas School District

Pride Month Slides 2024 David Douglas School District
Executive Directors Chat Leveraging AI for Diversity, Equity, and Inclusion

Executive Directors Chat Leveraging AI for Diversity, Equity, and Inclusion
How to Build a Module in Odoo 17 Using the Scaffold Method

How to Build a Module in Odoo 17 Using the Scaffold Method
Film vocab for eal 3 students: Australia the movie

Film vocab for eal 3 students: Australia the movie
Digital Artefact 1 - Tiny Home Environmental Design

Digital Artefact 1 - Tiny Home Environmental Design
Walmart Business+ and Spark Good for Nonprofits.pdf

Walmart Business+ and Spark Good for Nonprofits.pdf
Pollock and Snow "DEIA in the Scholarly Landscape, Session One: Setting Expec...

Pollock and Snow "DEIA in the Scholarly Landscape, Session One: Setting Expec...
Acid base table
- 1. Table of Acid and Base Strength Ka Acid Base Name Formula Formula Name Large Perchloric acid HClO4 ClO4 - Perchlorate ion 3.2 * 109 Hydroiodic acid HI I- Iodide 1.0 * 109 Hydrobromic acid HBr Br- Bromide 1.3 * 106 Hydrochloric acid HCl Cl- Chloride 1.0 * 103 Sulfuric acid H2SO4 HSO4 - Hydrogen sulfate ion 2.4 * 101 Nitric acid HNO3 NO3 - Nitrate ion -------- Hydronium ion H3O+ H2O Water 5.4 * 10-2 Oxalic acid HO2C2O2H HO2C2O2 - Hydrogen oxalate ion 1.3 * 10-2 Sulfurous acid H2SO3 HSO3 - Hydrogen sulfite ion 1.0 * 10-2 Hydrogen sulfate ion HSO4 - SO4 2- Sulfate ion 7.1 * 10-3 Phosphoric acid H3PO4 H2PO4 - Dihydrogen phosphate ion 7.2 * 10-4 Nitrous acid HNO2 NO3 - Nitrite ion 6.6 * 10-4 Hydrofluoric acid HF F - Fluoride ion 1.8 * 10-4 Methanoic acid HCO2H HCO2 - Methanoate ion 6.3 * 10-5 Benzoic acid C6H5COOH C6H5COO- Benzoate ion 5.4 * 10-5 Hydrogen oxalate ion HO2C2O2- O2C2O2 2- Oxalate ion 1.8 * 10-5 Ethanoic acid CH3COOH CH3COO Ethanoate (acetate) ion 4.4 * 10-7 Carbonic acid CO3 2- HCO3 - Hydrogen carbonate ion 1.1 * 10-7 Hydrosulfuric acid H2S HS- Hydrogen sulfide ion 6.3 * 10-8 Dihydrogen phosphate ion H2PO4 - HPO4 2- Hydrogen phosphate ion 6.2 * 10-8 Hydrogen sulfite ion HS- S2- Sulfite ion 2.9 * 10-8 Hypochlorous acid HClO ClO- Hypochlorite ion 6.2 * 10-10 Hydrocyanic acid HCN CN- Cyanide ion 5.8 * 10-10 Ammonium ion NH4 + NH3 Ammonia 5.8 * 10-10 Boric acid H3BO3 H2BO3 - Dihydrogen carbonate ion 4.7 * 10-11 Hydrogen carbonate ion HCO3 - CO3 2- Carbonate ion 4.2 * 10-13 Hydrogen phosphate ion HPO4 2- PO4 3- Phosphate ion 1.8 * 10-13 Dihydrogen borate ion H2BO3 - HBO3 2- Hydrogen borate ion 1.3 * 10-13 Hydrogen sulfide ion HS- S 2- Sulfide ion 1.6 * 10-14 Hydrogen borate ion HBO3 2- BO3 3- Borate ion --------- water H2O OH- Hydroxide 1. Strong acids are listed at the top left hand corner of the table and have Ka values >1 2. Acid with values less than one are considered weak. 3. The strong bases are listed at the bottom right of the table and get weaker as we move to the top of the table.
