Tabla de Ka y pKa
•
1 like•24,634 views
Tabla de Ka y pKa nos muestra acidos con sus respectivas constantes de acides y pka's. En la tabla se ve el nombre del acido, su formula quimica, su acido conjugado su base conjugada, y la fuerza de estos.
Report
Share
Report
Share
Download to read offline
Recommended
Titulaciones REDOX y Titulaciones Potenciometricas

Capitulo 14 - Química Analítica - Gary
QFB UAM Xochimilco - Sexto Trimestre
Recommended
Titulaciones REDOX y Titulaciones Potenciometricas

Capitulo 14 - Química Analítica - Gary
QFB UAM Xochimilco - Sexto Trimestre
Volumetría de neutralización – mezcla de álcalis – hidróxido

Informe Escrito de la Titulación de una muestra de álcalis de hidróxido. En el anexo se encuentra el link del videotutorial que acompaña el trabajo.
Practica 8 (preparacion y estandarizacion del edta)

Química Analítica y Métodos Instrumentales
Equipo numero 1
Universidad Veracruzana
Ing. Química 302
Estimación de hierro (ii) por óxido reducción en suplementos comerciales

Estimación de Hierro(II) por oxido reducción en suplementos comerciales
More Related Content
What's hot
Volumetría de neutralización – mezcla de álcalis – hidróxido

Informe Escrito de la Titulación de una muestra de álcalis de hidróxido. En el anexo se encuentra el link del videotutorial que acompaña el trabajo.
Practica 8 (preparacion y estandarizacion del edta)

Química Analítica y Métodos Instrumentales
Equipo numero 1
Universidad Veracruzana
Ing. Química 302
Estimación de hierro (ii) por óxido reducción en suplementos comerciales

Estimación de Hierro(II) por oxido reducción en suplementos comerciales
What's hot (20)
Volumetría de neutralización – mezcla de álcalis – hidróxido

Volumetría de neutralización – mezcla de álcalis – hidróxido
Practica 8 (preparacion y estandarizacion del edta)

Practica 8 (preparacion y estandarizacion del edta)
Estimación de hierro (ii) por óxido reducción en suplementos comerciales

Estimación de hierro (ii) por óxido reducción en suplementos comerciales
ácidos polipróticos o polifuncionales (clase 05/02/2014)

ácidos polipróticos o polifuncionales (clase 05/02/2014)
Similar to Tabla de Ka y pKa
Strong weak acids and bases

Acids are divided into two categories based on the ease with which they can donate protons to the solvent: i) strong acids and ii) weak acids
Strong acids are acids that completely dissociate in water. The reaction of an acid with its solvent (typically H2O) is called an acid dissociation reaction.
Weak acids are acids that dissociate partially in water. The extent of dissociation is given by the equilibrium constant.
Note:
A measure of the relative strength of an acid is: i) the equilibrium constant ka of the dissociation reaction of the acid in water (depends on temperature) ii) the degree of dissociation α of the acid in water (depends on the concentration of the acid an on temperature).
Acid lonization Constants (K-) for Some Monoprotic Weak Acids at 25 ec.docx

Acid lonization Constants (K,) for Some Monoprotic Weak Acids at 25 ec s Formula Structural Formula lonization Reaction HCIO2 Chlorous acid 1.96 Ho (aq)CO2 (aq) Go(g) 1.110 Nitrous acicd HNO2 HNO2(aq) H20(,) H0 (aq)NO2 (aq) + 4.6 10 3.34 Hydrofluoric acid HF HF(ag) H200 H-F 3.5 104 3.46 H0 (aq)F(aq) Formic acid ????? HCHO2(ag)H200) - 3,74 H-0-C-H Ho (ag)CHO2 (aq) CHO g)1.8 101 ?? ? HC7H502 CH HC,H02(a)H20U) H-0-C-C Benzoic acid 6.510 4.19 H0 (a) C,MG02 (a0) HC,H30(aq) H20U) Acetic acid 1.8 x 10 4.74 H-0-C-CH H30 (aq) CH02 (aq) HOO(aq) H20(,) H30 (aq) = + 2.9 x 10 7.54 Hypochlorous acid HCIO CIO (aq) HCN(ag) H20U) 9.31 HCN Ho (aq)CN CN (ag)4.9 10 1 acid HC@H,0(aq) H200)- 1010 9.89 CH 1.3 x Phenol HCgHs0 ??-? H0(aq)CaHO (aq) KkK. (See tha dieeuesinn at the end of Section 15
Solution
1)
the given Ka = 3.16 x 10^-5
pKa = -log Ka
= -log (3.16 x 10^-5)
pKa = 4.50
the corresponding acid = Acetic acid .
acetic acid pKa = 4.74
this is very close to the given pKa value.
.
Similar to Tabla de Ka y pKa (20)
Acid lonization Constants (K-) for Some Monoprotic Weak Acids at 25 ec.docx

Acid lonization Constants (K-) for Some Monoprotic Weak Acids at 25 ec.docx
Recently uploaded
Multi-source connectivity as the driver of solar wind variability in the heli...

The ambient solar wind that flls the heliosphere originates from multiple
sources in the solar corona and is highly structured. It is often described
as high-speed, relatively homogeneous, plasma streams from coronal
holes and slow-speed, highly variable, streams whose source regions are
under debate. A key goal of ESA/NASA’s Solar Orbiter mission is to identify
solar wind sources and understand what drives the complexity seen in the
heliosphere. By combining magnetic feld modelling and spectroscopic
techniques with high-resolution observations and measurements, we show
that the solar wind variability detected in situ by Solar Orbiter in March
2022 is driven by spatio-temporal changes in the magnetic connectivity to
multiple sources in the solar atmosphere. The magnetic feld footpoints
connected to the spacecraft moved from the boundaries of a coronal hole
to one active region (12961) and then across to another region (12957). This
is refected in the in situ measurements, which show the transition from fast
to highly Alfvénic then to slow solar wind that is disrupted by the arrival of
a coronal mass ejection. Our results describe solar wind variability at 0.5 au
but are applicable to near-Earth observatories.
Structural Classification Of Protein (SCOP)

A brief information about the SCOP protein database used in bioinformatics.
The Structural Classification of Proteins (SCOP) database is a comprehensive and authoritative resource for the structural and evolutionary relationships of proteins. It provides a detailed and curated classification of protein structures, grouping them into families, superfamilies, and folds based on their structural and sequence similarities.
Astronomy Update- Curiosity’s exploration of Mars _ Local Briefs _ leadertele...

Article written for leader telegram
Citrus Greening Disease and its Management

Citrus Greening was one of the major causes of decline in the citrus production. So, effective management cultural practices should be incorporated
insect morphology and physiology of insect

the documents provide complete information of insect morphology and their structure
Structures and textures of metamorphic rocks

It is useful for the Under Graduating students for easy understanding and it's useful for the exam preparations.
SCHIZOPHRENIA Disorder/ Brain Disorder.pdf

This pdf is about the Schizophrenia.
For more details visit on YouTube; @SELF-EXPLANATORY;
https://www.youtube.com/channel/UCAiarMZDNhe1A3Rnpr_WkzA/videos
Thanks...!
The ASGCT Annual Meeting was packed with exciting progress in the field advan...

The ASGCT Annual Meeting was packed with exciting progress in the field advancing efforts to deliver highly promising therapies to more patients.
Unveiling the Energy Potential of Marshmallow Deposits.pdf

Unveiling the Energy Potential of Marshmallow Deposits: A Revolutionary
Breakthrough in Sustainable Energy Science
Recently uploaded (20)
platelets- lifespan -Clot retraction-disorders.pptx

platelets- lifespan -Clot retraction-disorders.pptx
Circulatory system_ Laplace law. Ohms law.reynaults law,baro-chemo-receptors-...

Circulatory system_ Laplace law. Ohms law.reynaults law,baro-chemo-receptors-...
PRESENTATION ABOUT PRINCIPLE OF COSMATIC EVALUATION

PRESENTATION ABOUT PRINCIPLE OF COSMATIC EVALUATION
Multi-source connectivity as the driver of solar wind variability in the heli...

Multi-source connectivity as the driver of solar wind variability in the heli...
Astronomy Update- Curiosity’s exploration of Mars _ Local Briefs _ leadertele...

Astronomy Update- Curiosity’s exploration of Mars _ Local Briefs _ leadertele...
Lateral Ventricles.pdf very easy good diagrams comprehensive

Lateral Ventricles.pdf very easy good diagrams comprehensive
The ASGCT Annual Meeting was packed with exciting progress in the field advan...

The ASGCT Annual Meeting was packed with exciting progress in the field advan...
ESR_factors_affect-clinic significance-Pathysiology.pptx

ESR_factors_affect-clinic significance-Pathysiology.pptx
Unveiling the Energy Potential of Marshmallow Deposits.pdf

Unveiling the Energy Potential of Marshmallow Deposits.pdf
Tabla de Ka y pKa
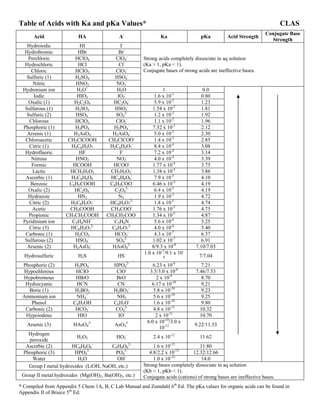
- 1. Table of Acids with Ka and pKa Values* CLAS Acid HA A- Ka pKa Acid Strength Conjugate Base Strength Hydroiodic HI I- Hydrobromic HBr Br- Perchloric HClO4 ClO4 - Hydrochloric HCl Cl- Chloric HClO3 ClO3 - Sulfuric (1) H2SO4 HSO4 - Nitric HNO3 NO3 - Strong acids completely dissociate in aq solution (Ka > 1, pKa < 1). Conjugate bases of strong acids are ineffective bases. Hydronium ion H3O+ H2O 1 0.0 Iodic HIO3 IO3 - 1.6 x 10-1 0.80 Oxalic (1) H2C2O4 HC2O4 - 5.9 x 10-2 1.23 Sulfurous (1) H2SO3 HSO3 - 1.54 x 10-2 1.81 Sulfuric (2) HSO4 - SO4 2- 1.2 x 10-2 1.92 Chlorous HClO2 ClO2 - 1.1 x 10-2 1.96 Phosphoric (1) H3PO4 H2PO4 - 7.52 x 10-3 2.12 Arsenic (1) H3AsO4 H2AsO4 - 5.0 x 10-3 2.30 Chloroacetic CH2ClCOOH CH2ClCOO- 1.4 x 10-3 2.85 Citric (1) H3C6H5O7 H2C6H5O7 - 8.4 x 10-4 3.08 Hydrofluoric HF F- 7.2 x 10-4 3.14 Nitrous HNO2 NO2 - 4.0 x 10-4 3.39 Formic HCOOH HCOO- 1.77 x 10-4 3.75 Lactic HCH3H5O3 CH3H5O3 - 1.38 x 10-4 3.86 Ascorbic (1) H2C6H6O6 HC6H6O6 - 7.9 x 10-5 4.10 Benzoic C6H5COOH C6H5COO- 6.46 x 10-5 4.19 Oxalic (2) HC2O4 - C2O4 2- 6.4 x 10-5 4.19 Hydrazoic HN3 N3 - 1.9 x 10-5 4.72 Citric (2) H2C6H5O7 - HC6H5O7 2- 1.8 x 10-5 4.74 Acetic CH3COOH CH3COO- 1.76 x 10-5 4.75 Propionic CH3CH2COOH CH3CH2COO- 1.34 x 10-5 4.87 Pyridinium ion C5H4NH+ C5H4N 5.6 x 10-6 5.25 Citric (3) HC6H5O7 2- C6H5O7 3- 4.0 x 10-6 5.40 Carbonic (1) H2CO3 HCO3 - 4.3 x 10-7 6.37 Sulfurous (2) HSO4 - SO4 2- 1.02 x 10-7 6.91 Arsenic (2) H2AsO4 - HAsO4 2- 8/9.3 x 10-8 7.10/7.03 Hydrosulfuric H2S HS- 1.0 x 10-7 /9.1 x 10- 8 7/7.04 Phosphoric (2) H2PO4 - HPO4 2- 6.23 x 10-8 7.21 Hypochlorous HClO ClO- 3.5/3.0 x 10-8 7.46/7.53 Hypobromous HBrO BrO- 2 x 10-9 8.70 Hydrocyanic HCN CN- 6.17 x 10-10 9.21 Boric (1) H3BO3 H2BO3 - 5.8 x 10-10 9.23 Ammonium ion NH4 + NH3 5.6 x 10-10 9.25 Phenol C6H5OH C6H5O- 1.6 x 10-10 9.80 Carbonic (2) HCO3 - CO3 2- 4.8 x 10-11 10.32 Hypoiodous HIO IO- 2 x 10-11 10.70 Arsenic (3) HAsO4 2- AsO4 3- 6.0 x 10-10 /3.0 x 10-12 9.22/11.53 Hydrogen peroxide H2O2 HO2 - 2.4 x 10-12 11.62 Ascorbic (2) HC6H6O6 - C6H6O6 2- 1.6 x 10-12 11.80 Phosphoric (3) HPO4 2- PO4 3- 4.8/2.2 x 10-13 12.32/12.66 Water H2O OH- 1.0 x 10-14 14.0 Group I metal hydroxides (LiOH, NaOH, etc.) Group II metal hydroxides (Mg(OH)2, Ba(OH)2, etc.) Strong bases completely dissociate in aq solution (Kb > 1, pKb < 1). Conjugate acids (cations) of strong bases are ineffective bases. * Compiled from Appendix 5 Chem 1A, B, C Lab Manual and Zumdahl 6th Ed. The pKa values for organic acids can be found in Appendix II of Bruice 5th Ed.
- 2. Table of Acids with Ka and pKa Values* CLAS * Compiled from Appendix 5 Chem 1A, B, C Lab Manual and Zumdahl 6th Ed. The pKa values for organic acids can be found in Appendix II of Bruice 5th Ed.