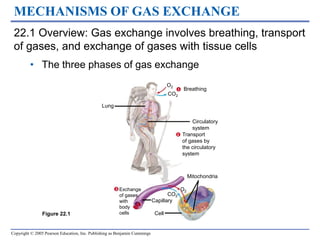
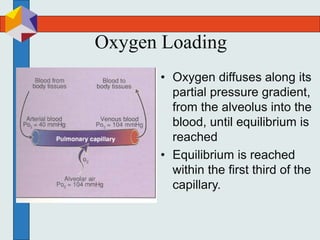
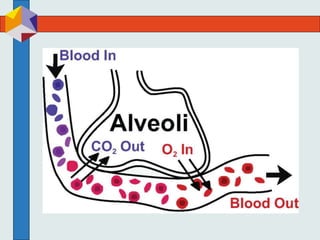
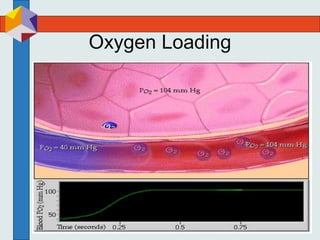
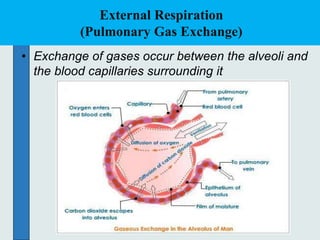
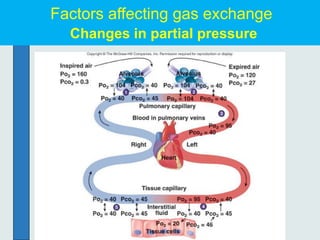
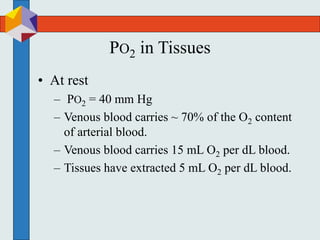
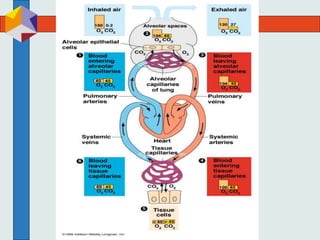
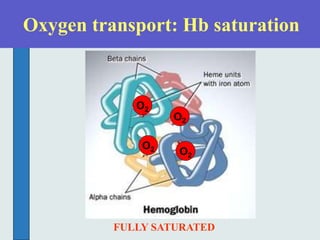
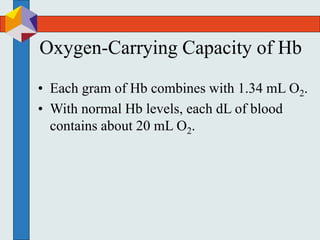
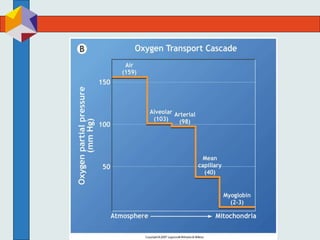
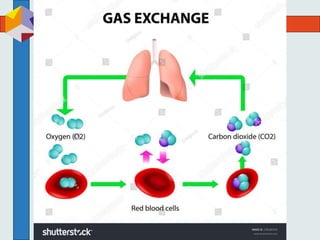
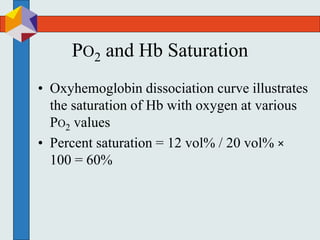
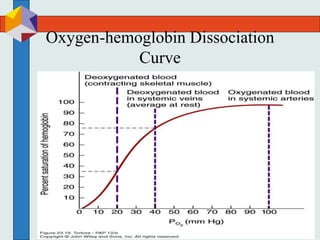
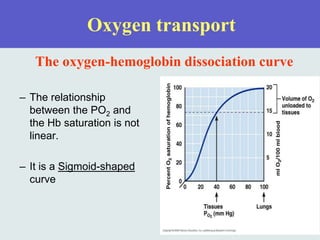
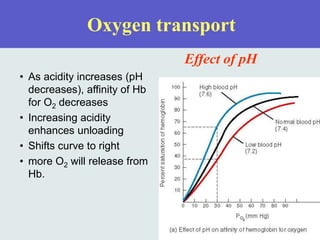
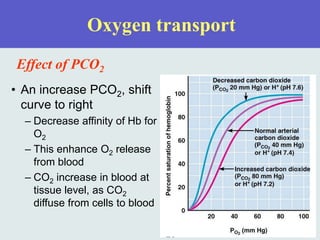
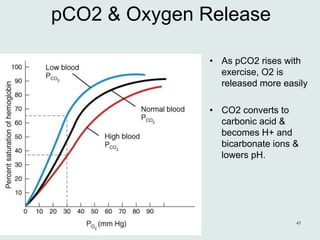
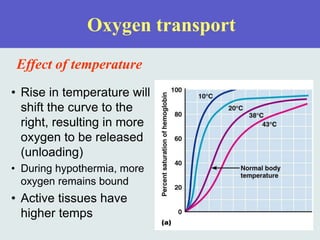
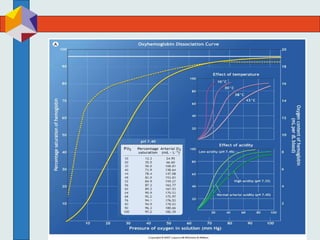
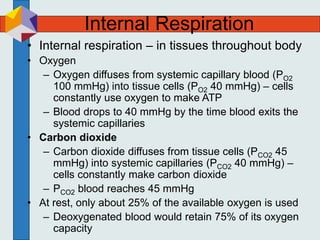
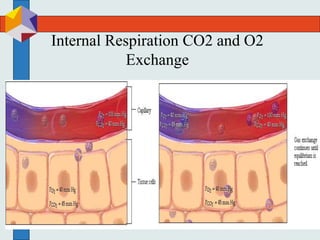
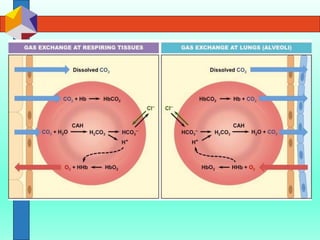
The three phases of gas exchange are breathing, transport of gases via the circulatory system, and exchange of gases with body cells. Oxygen diffuses from the alveoli into pulmonary capillaries based on the partial pressure gradient, then binds to hemoglobin and is transported to tissues where it diffuses into cells. Carbon dioxide diffuses in the opposite direction, from tissues into blood and from pulmonary capillaries into alveoli. Various factors like pH, temperature, carbon dioxide levels, and 2,3-DPG can shift the oxygen-hemoglobin dissociation curve right or left, facilitating oxygen loading or unloading in the lungs or tissues.