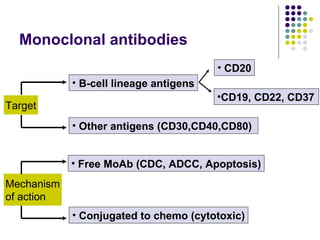
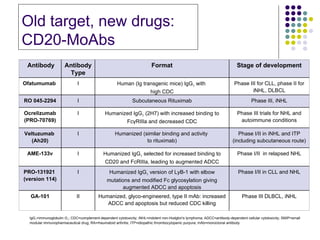
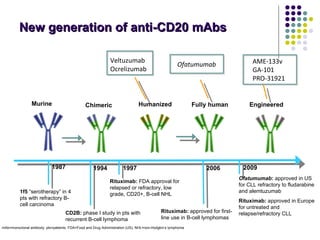
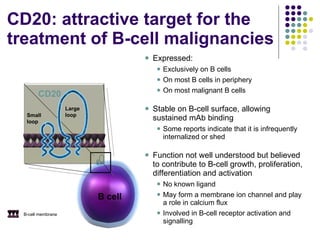
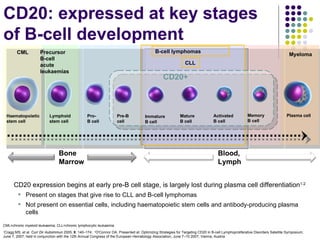
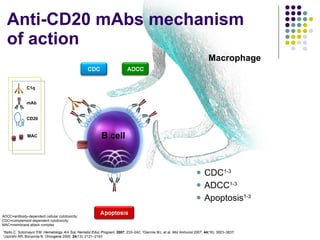
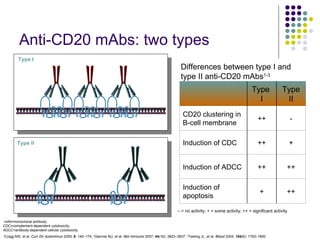
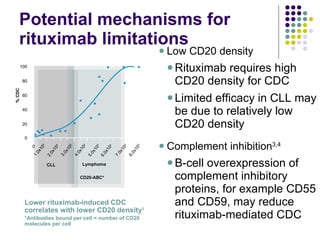
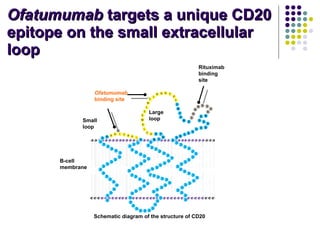
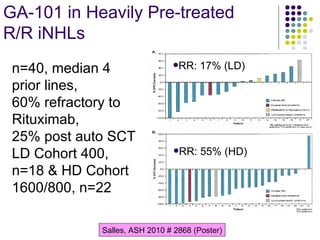
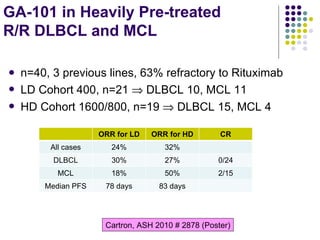
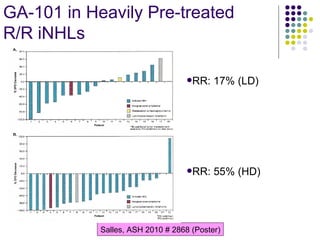
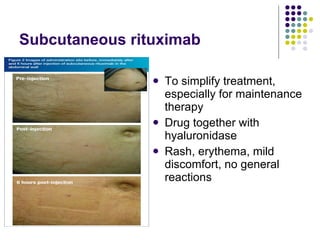
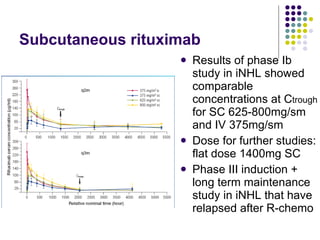
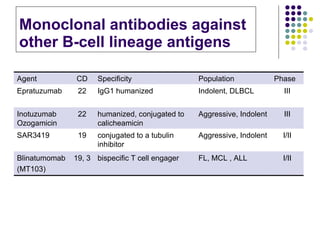
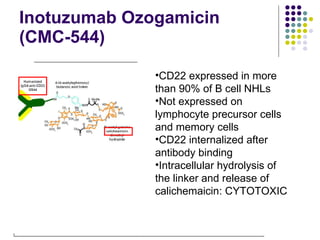
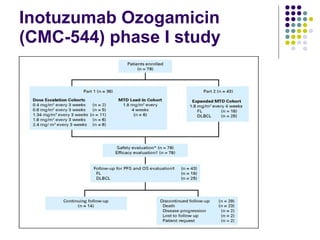
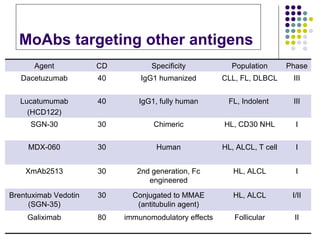
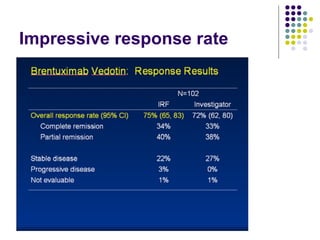
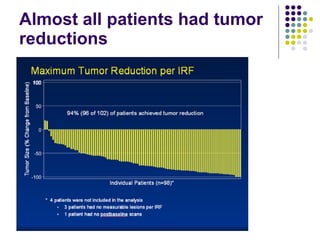
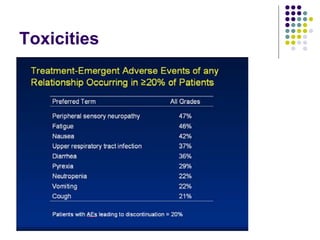
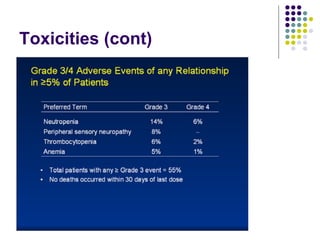
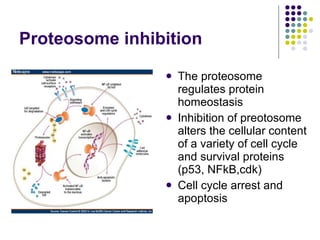
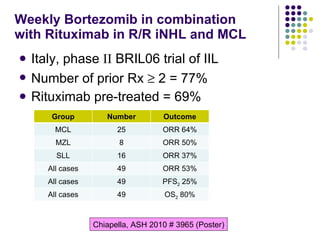
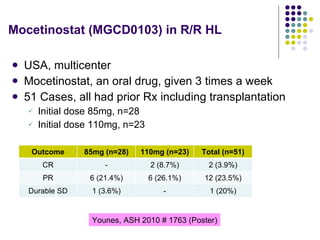
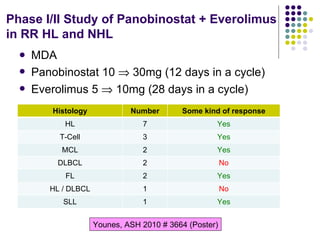
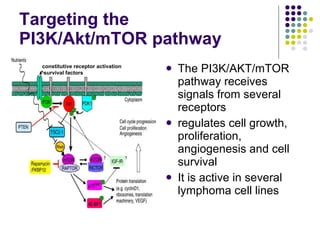
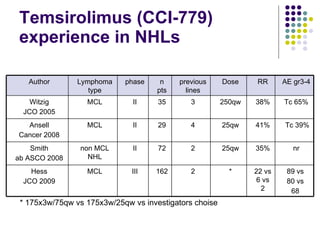
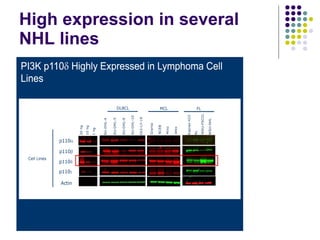
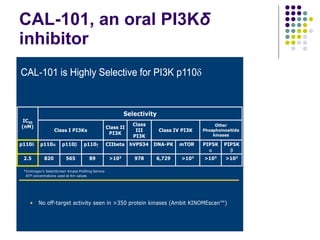
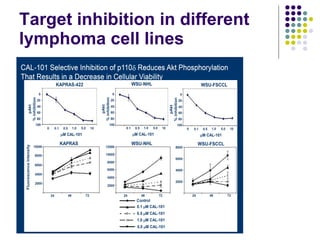
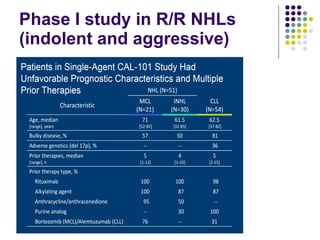
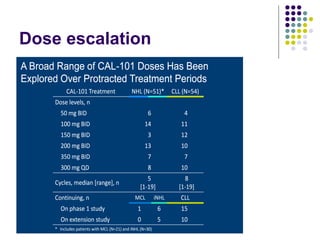
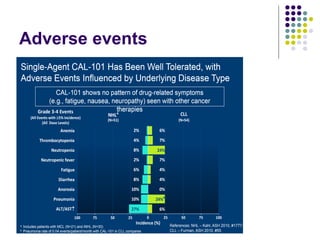
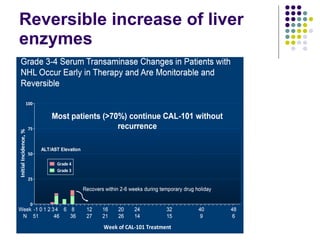
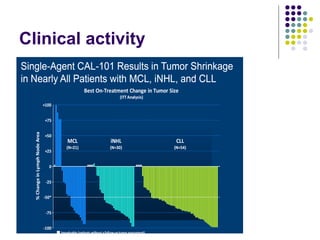
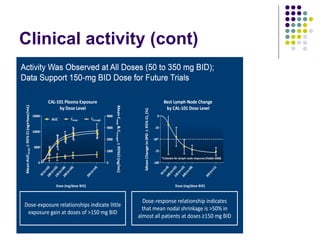
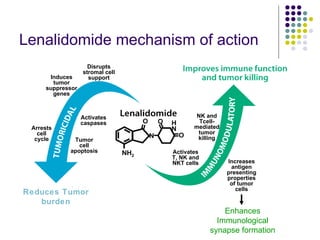
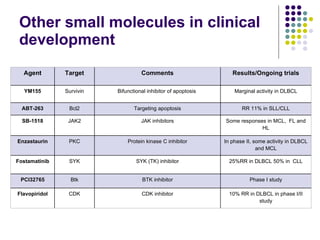
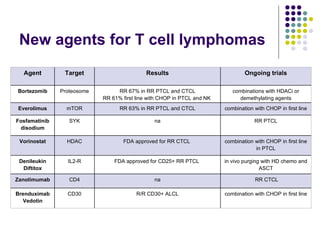
The document discusses new drugs for the treatment of lymphomas. It outlines several monoclonal antibodies that target antigens on B-cells, including CD20, CD19, CD22 and CD37. Ofatumumab and GA-101 are new anti-CD20 monoclonal antibodies that exhibit enhanced binding and cell-killing properties compared to Rituximab. Inotuzumab Ozogamicin is an antibody-drug conjugate targeting CD22 that is internalized and releases a cytotoxic drug, showing promising activity in early clinical trials.