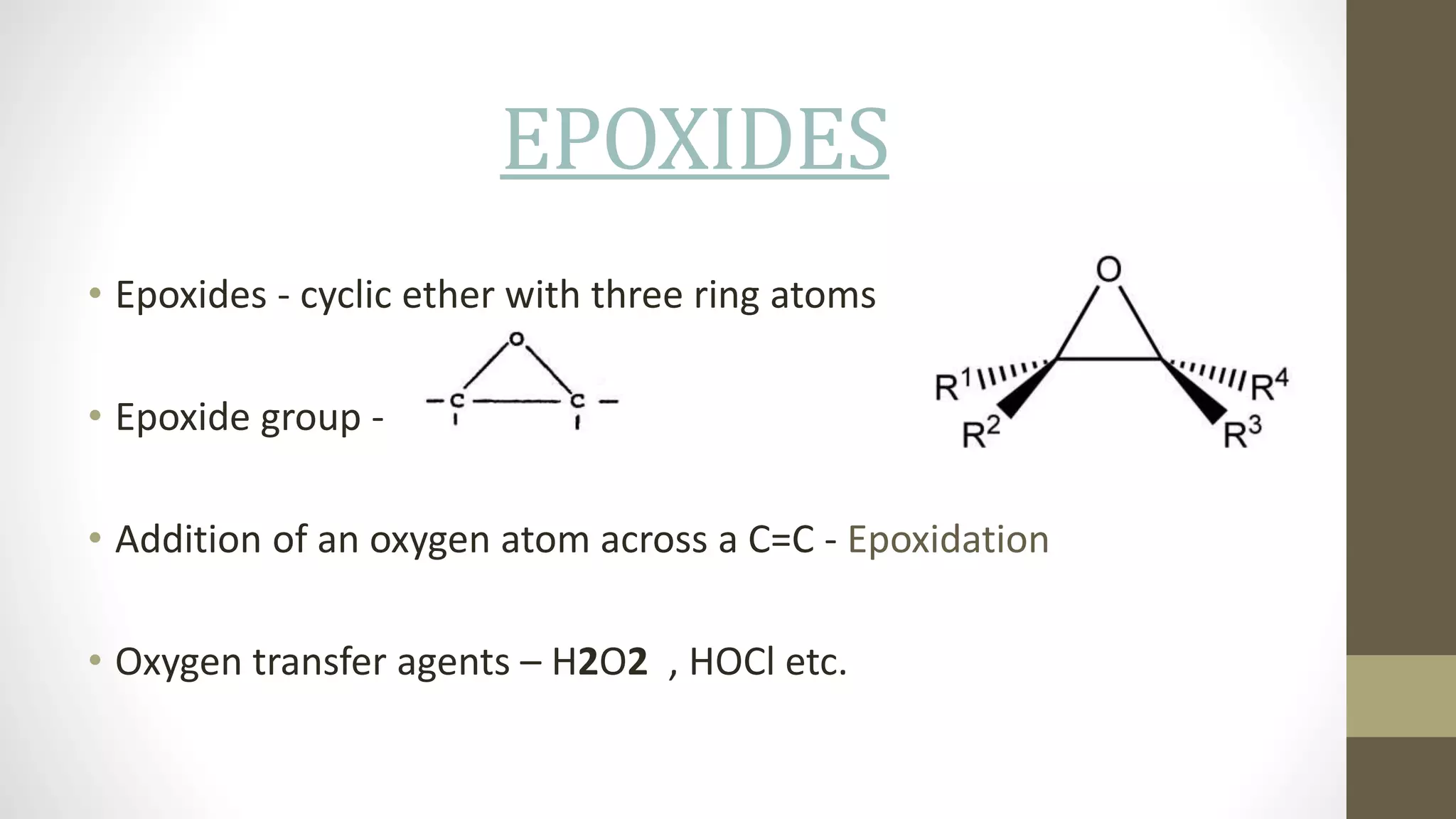
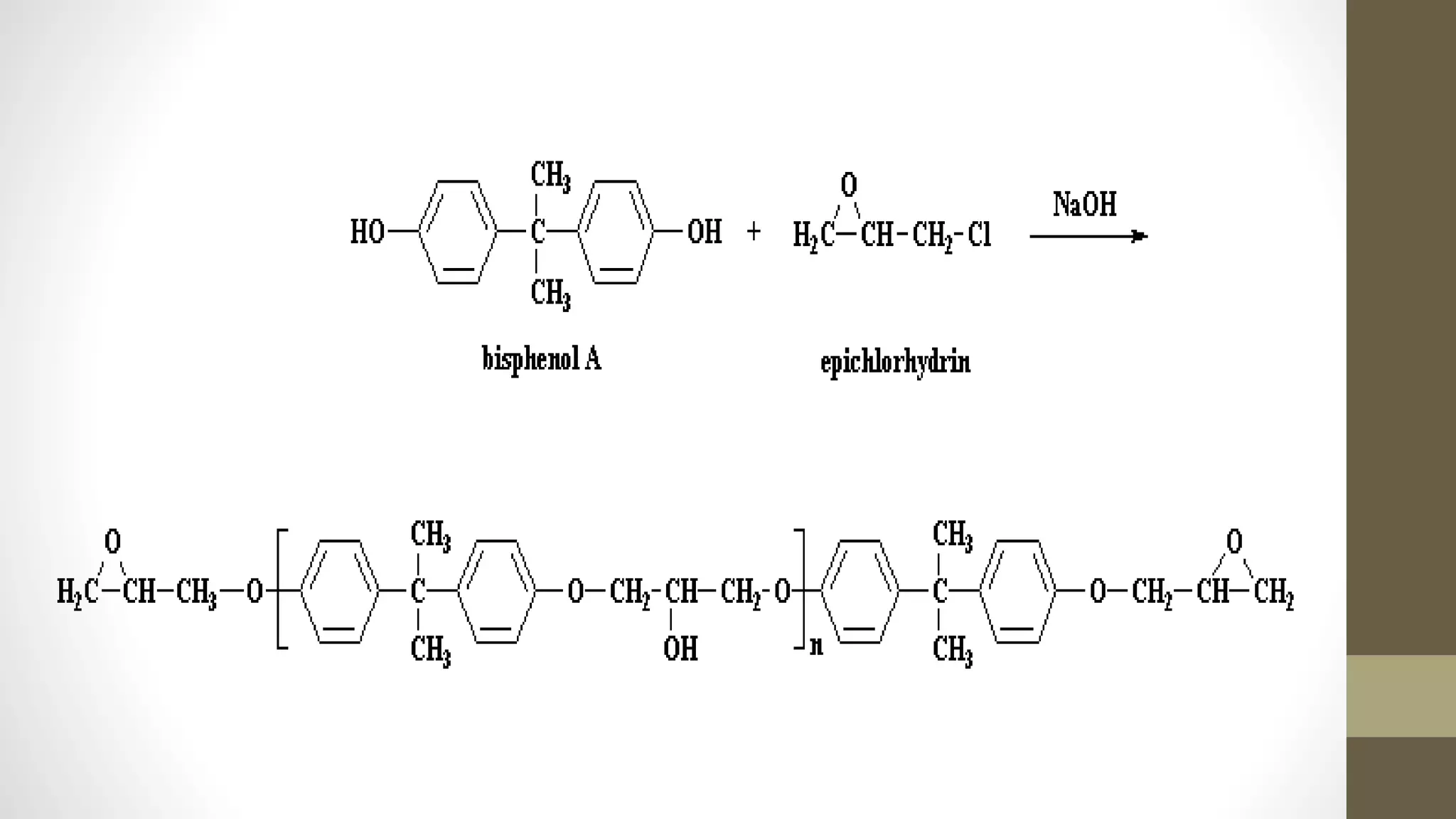
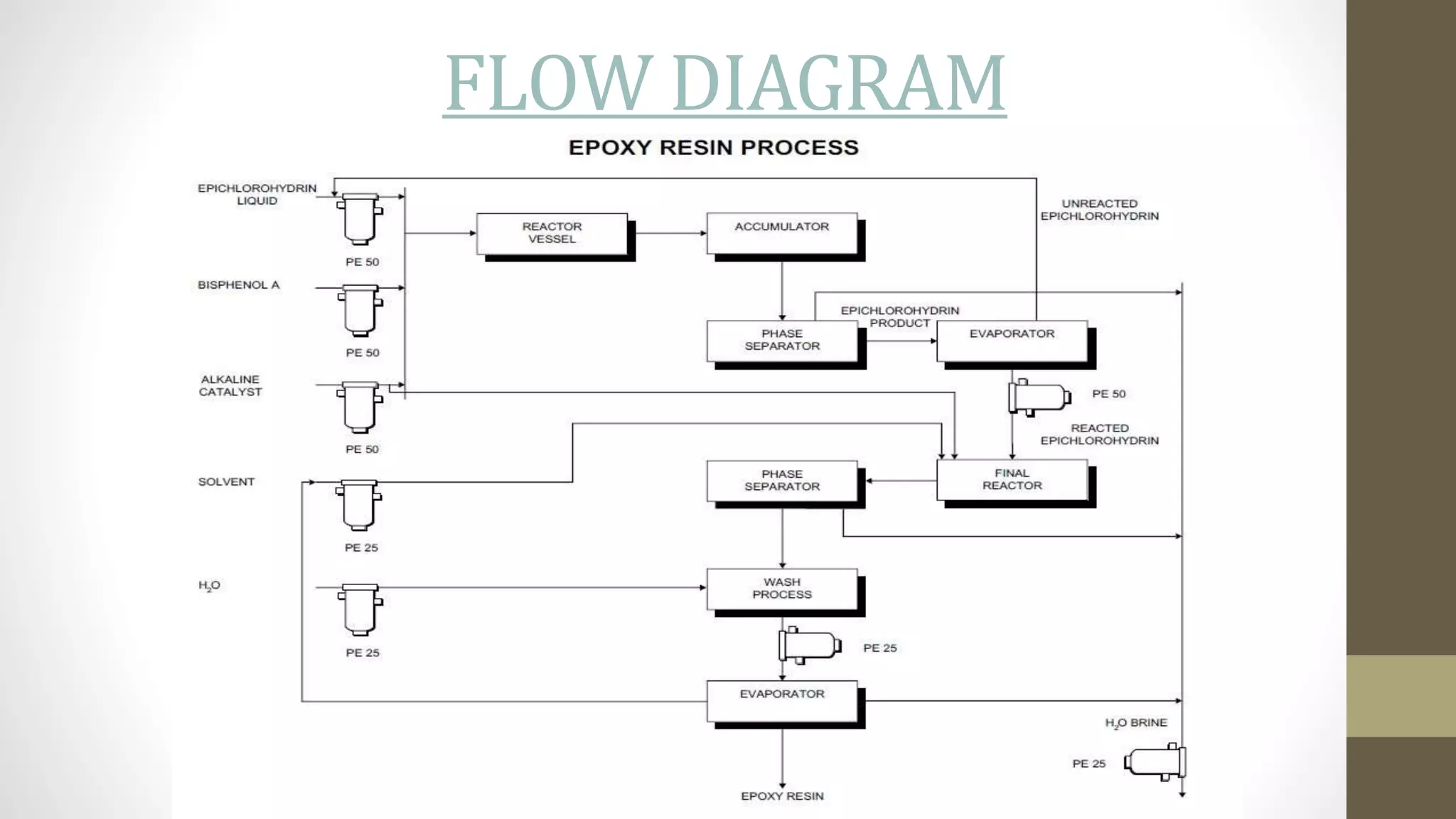
This document discusses epoxy resins, including their production and properties. Epoxy resins are reactive polymers that contain epoxide groups and can undergo crosslinking reactions through curing. They are produced from petroleum or plant-derived raw materials like epichlorohydrin and bisphenol A. Epoxy resins have properties like chemical stability, reactivity, adhesion and electrical insulation that make them useful for applications like structural adhesives, coatings, and electronics. Their production involves chemical reactions between epoxide monomers and crosslinking during curing.