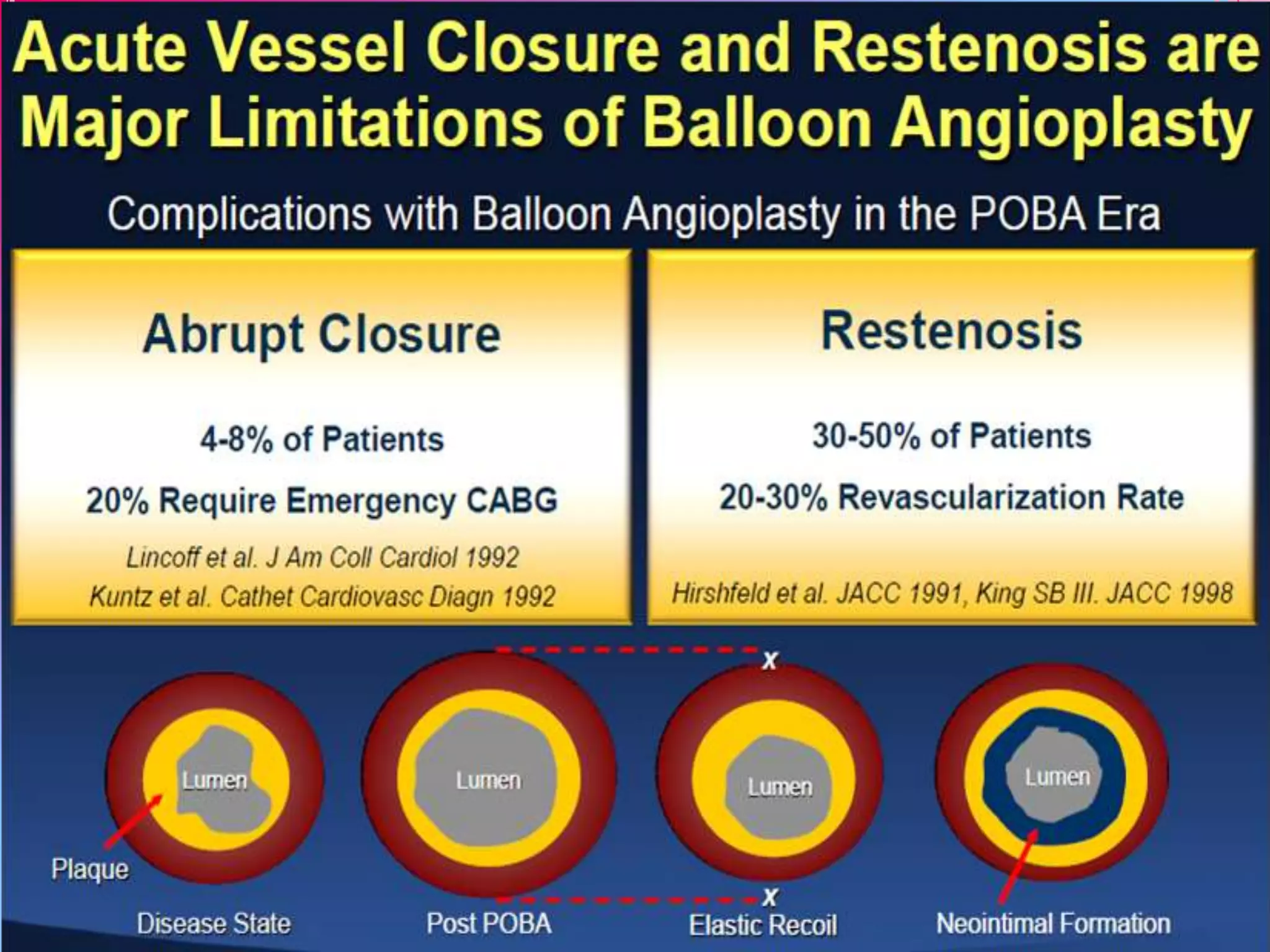
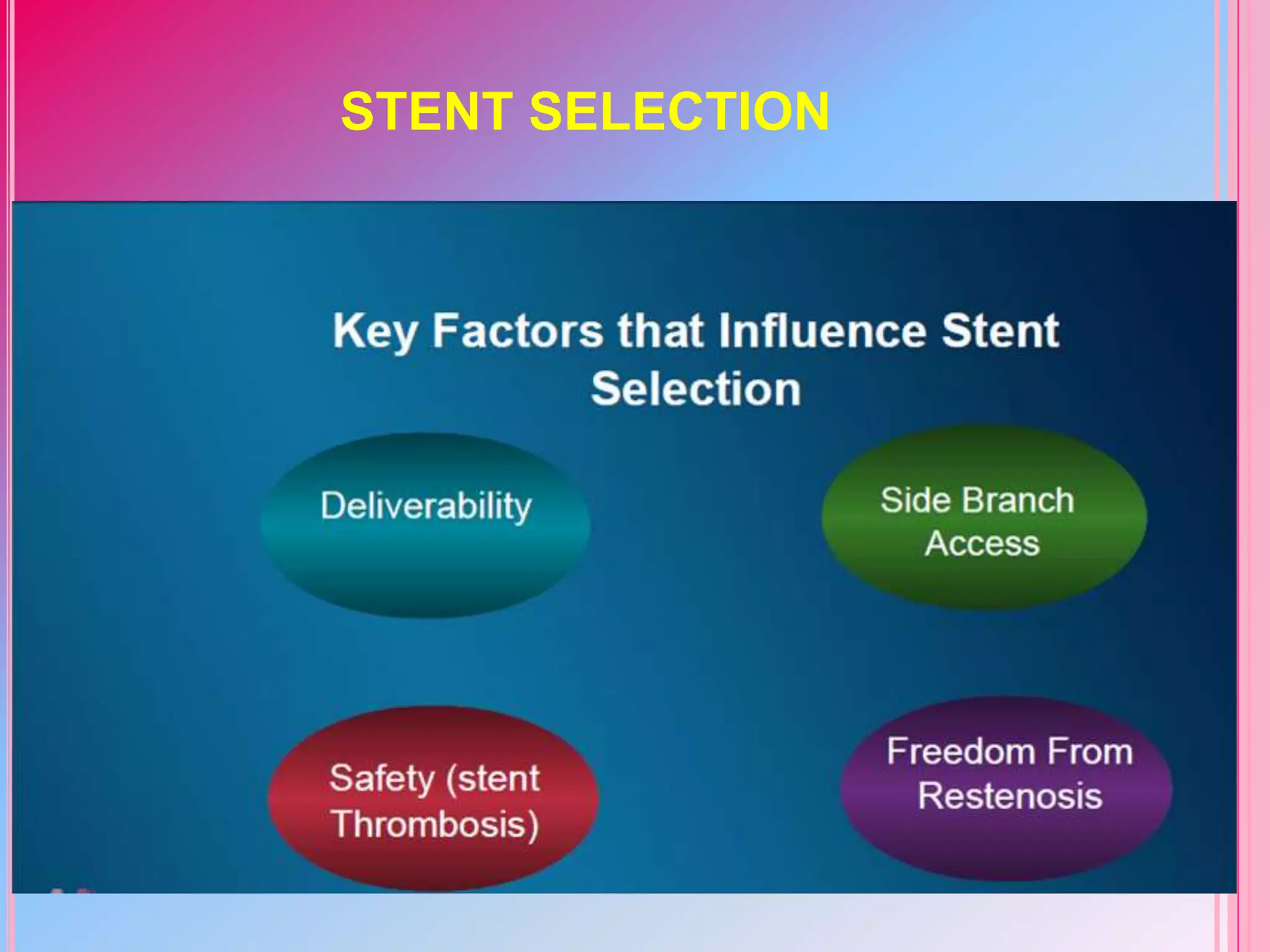
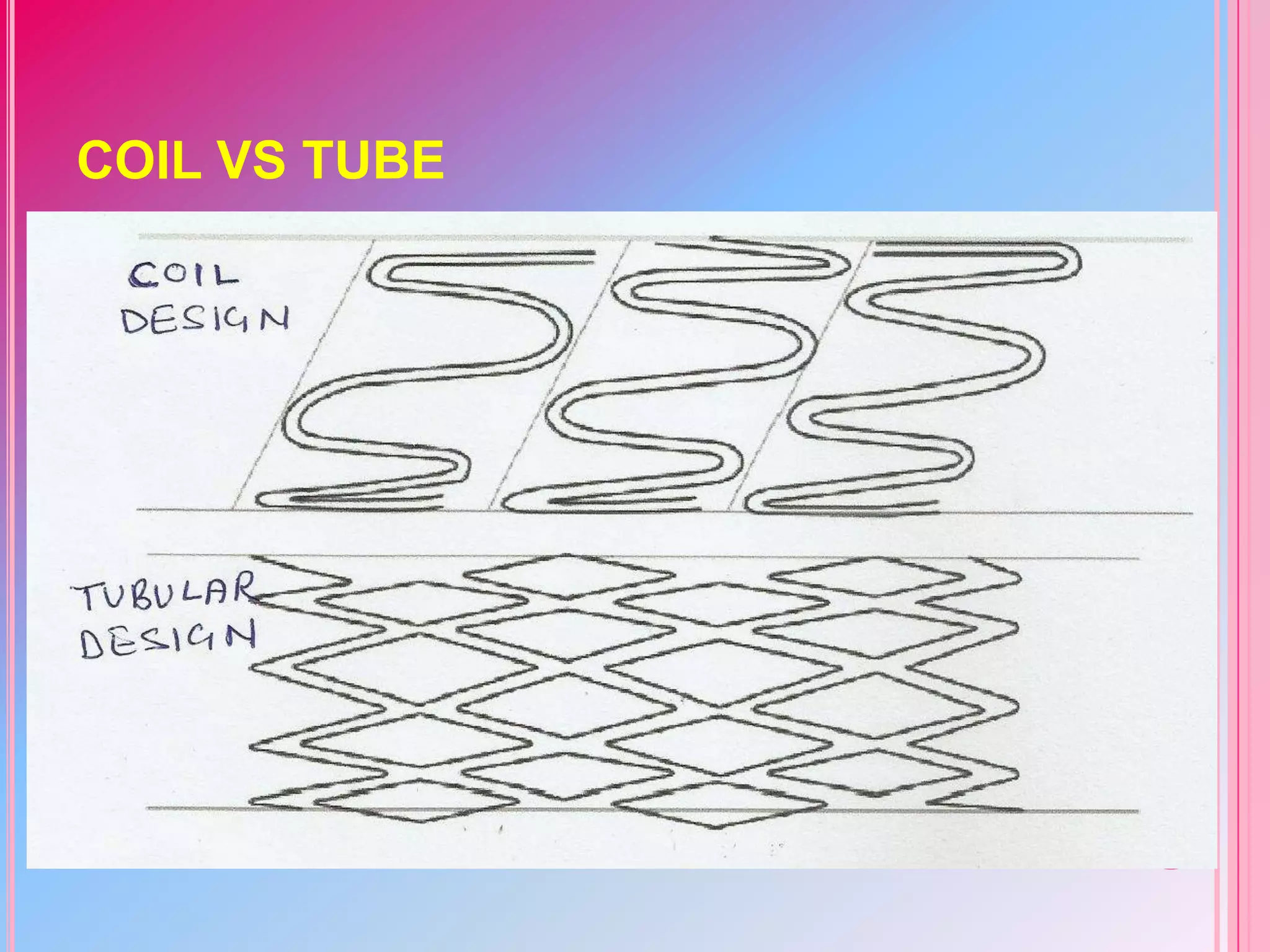
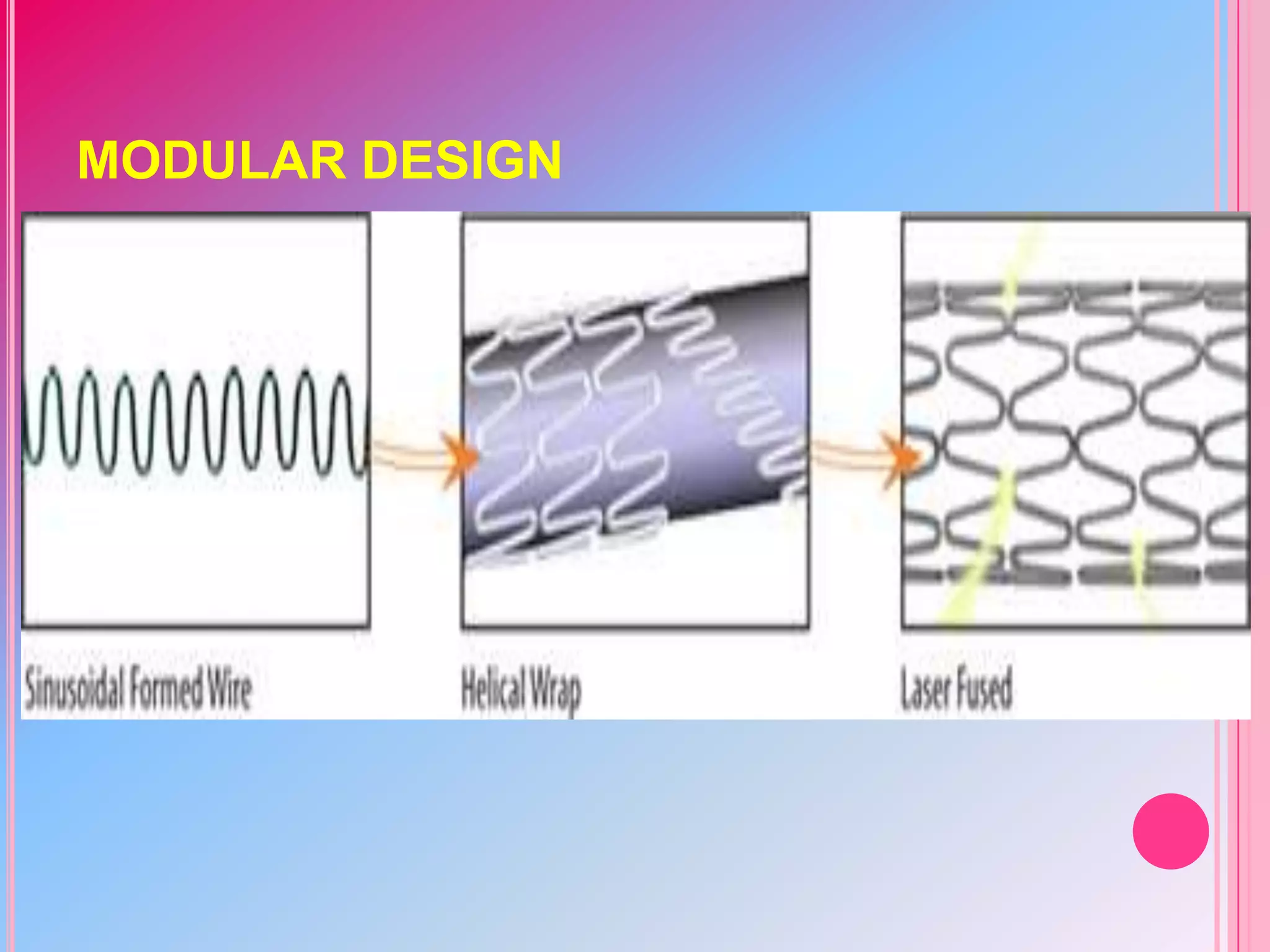
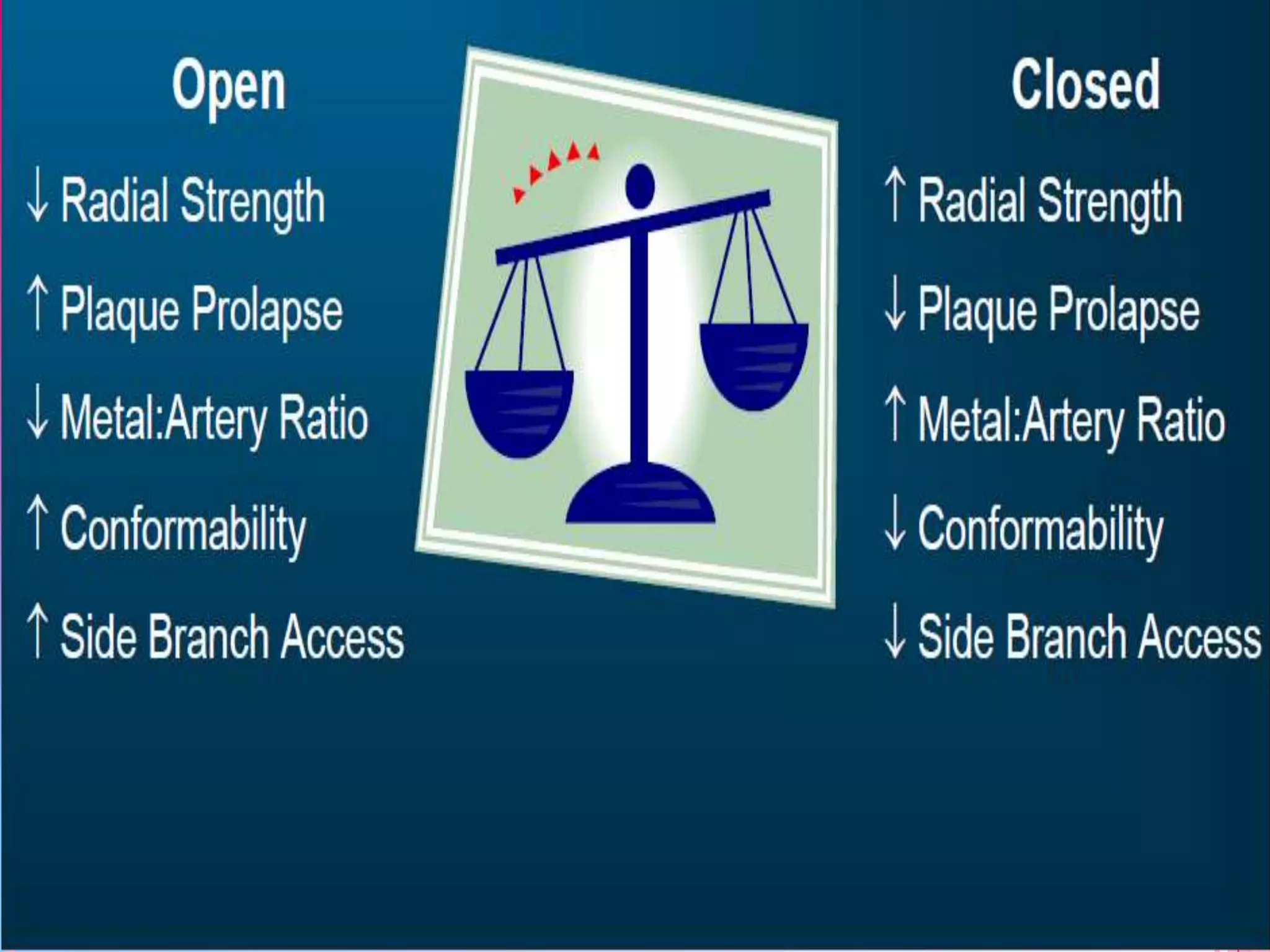
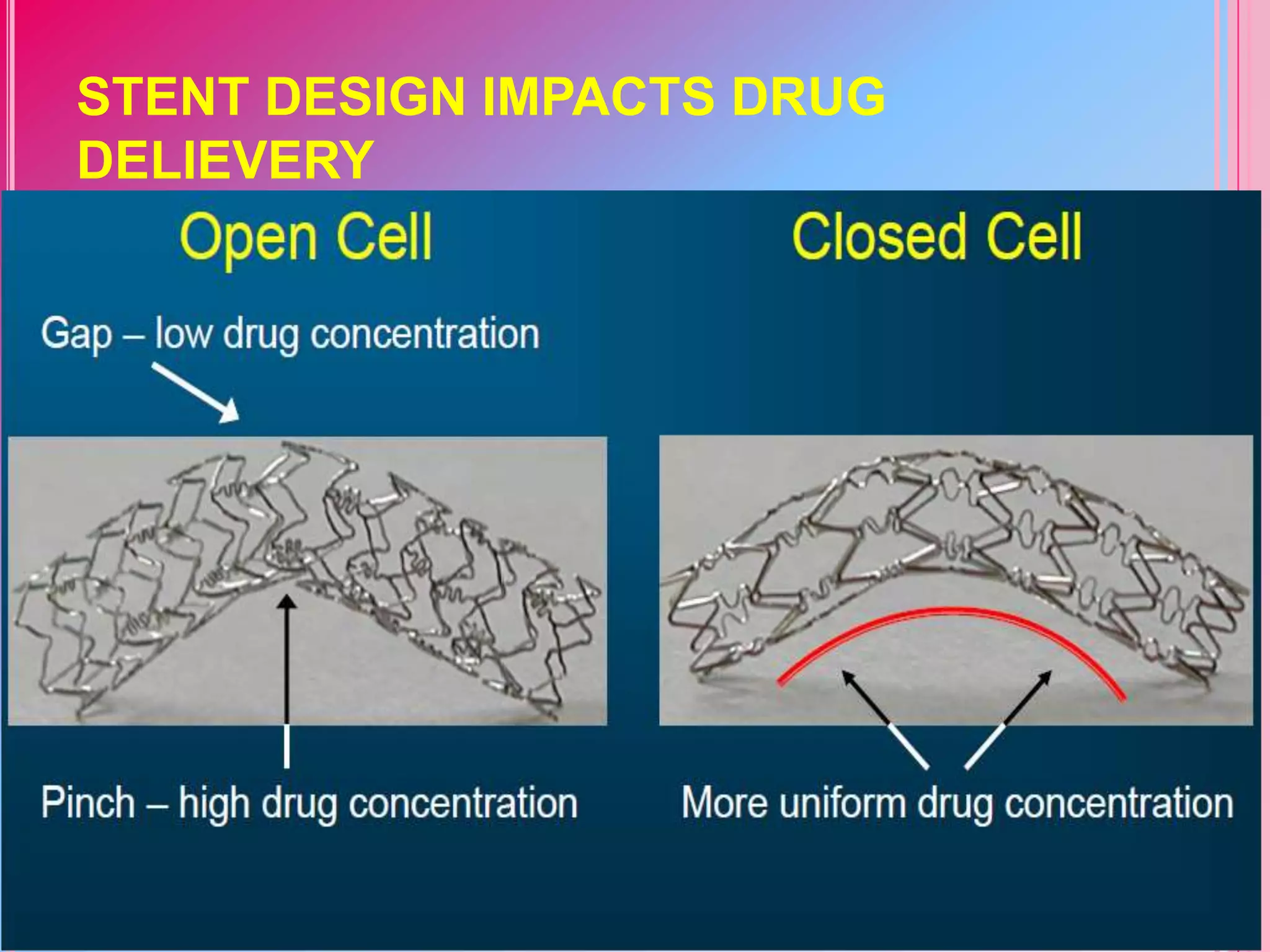
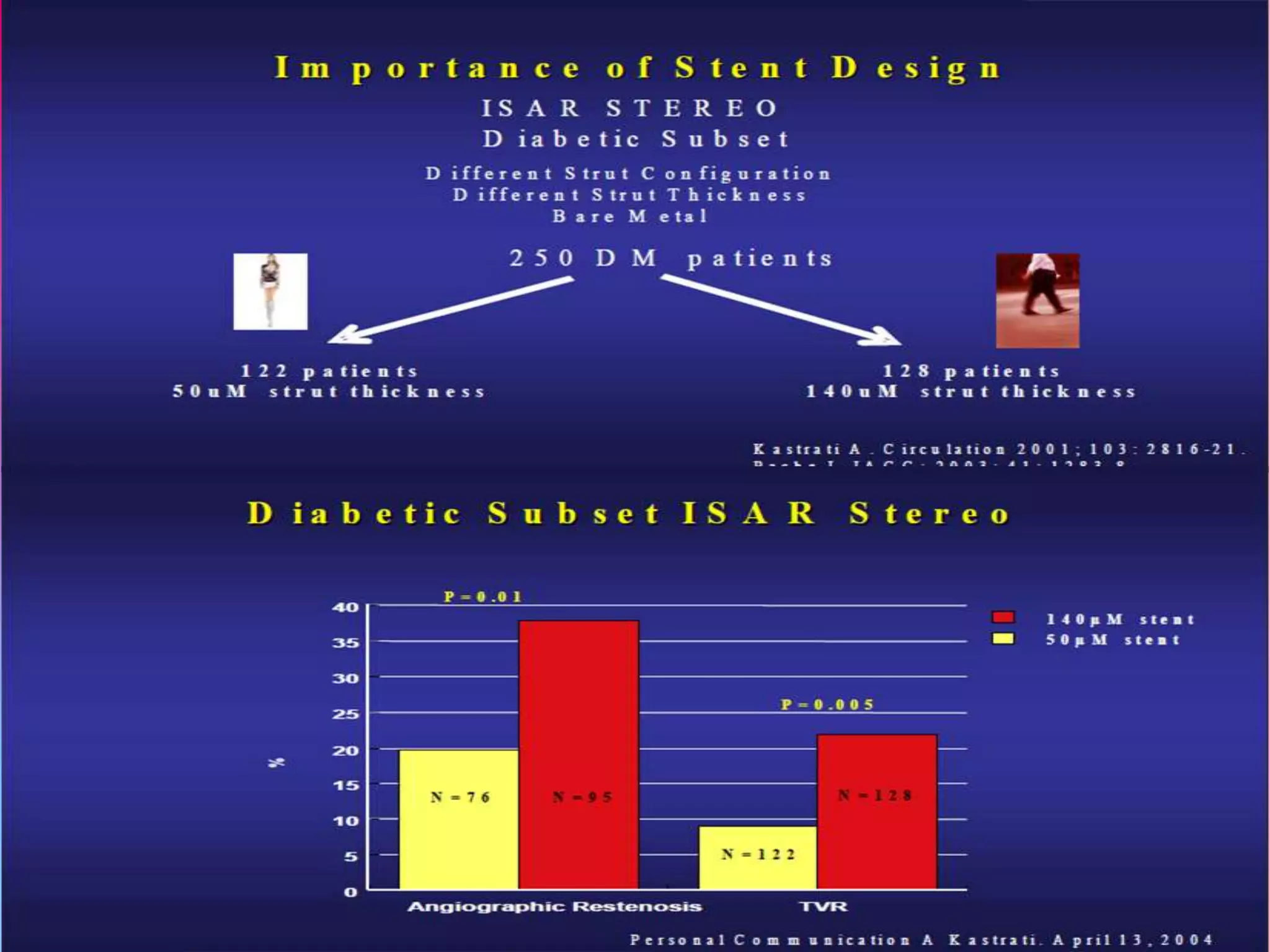
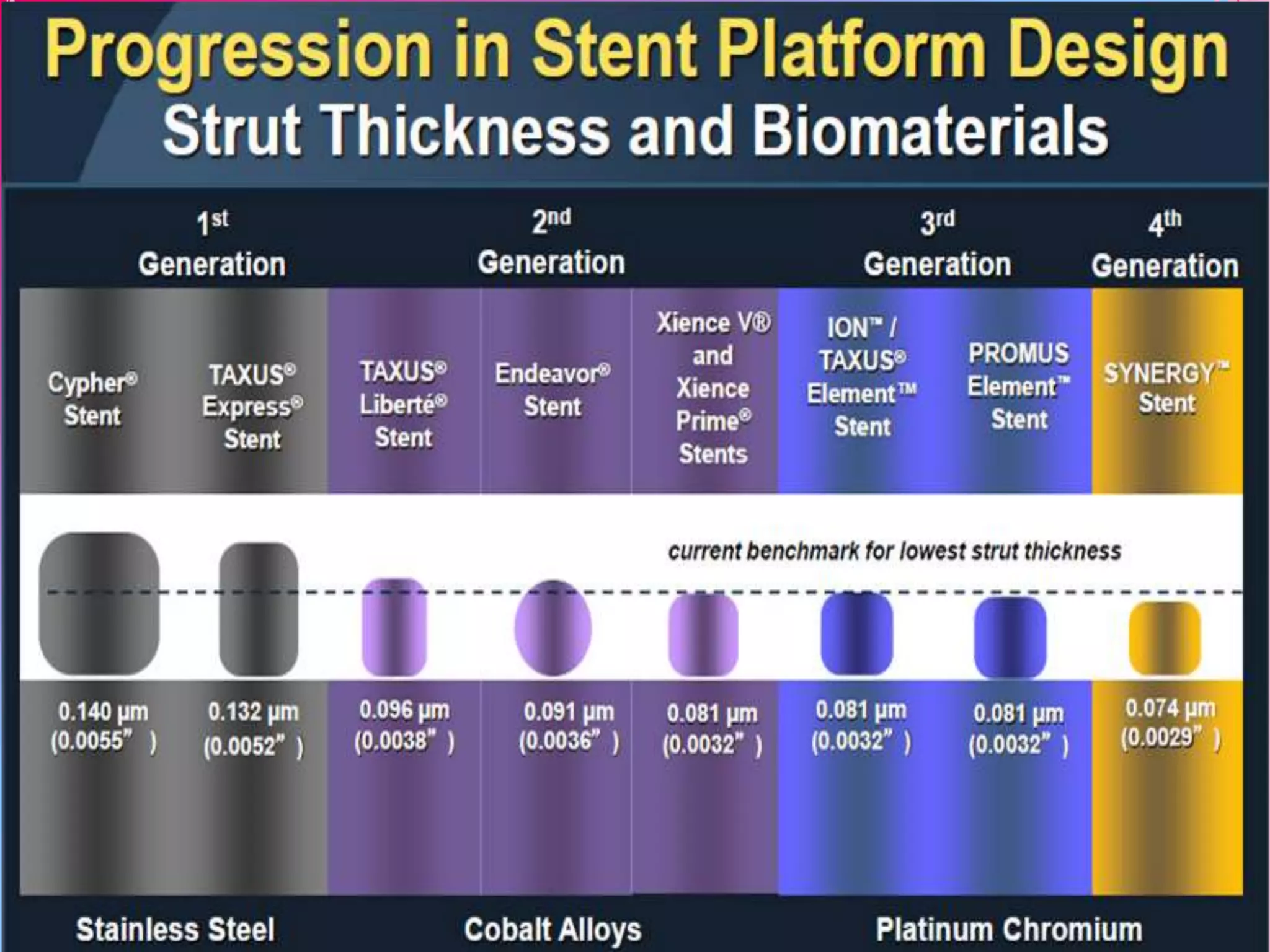
This document discusses different designs of coronary stents. It begins by providing background on the development of coronary stents and their approval for use. It then describes some of the earliest stent designs, including the Gianturco-Roubin coil stent and the Palmaz-Schatz slotted tube stent. The document goes on to discuss various aspects of stent design that can impact performance, such as the geometric configuration, materials used, coatings, and drug-eluting capabilities. Key design considerations like strut thickness, number of struts, and mechanical properties are also reviewed.

























































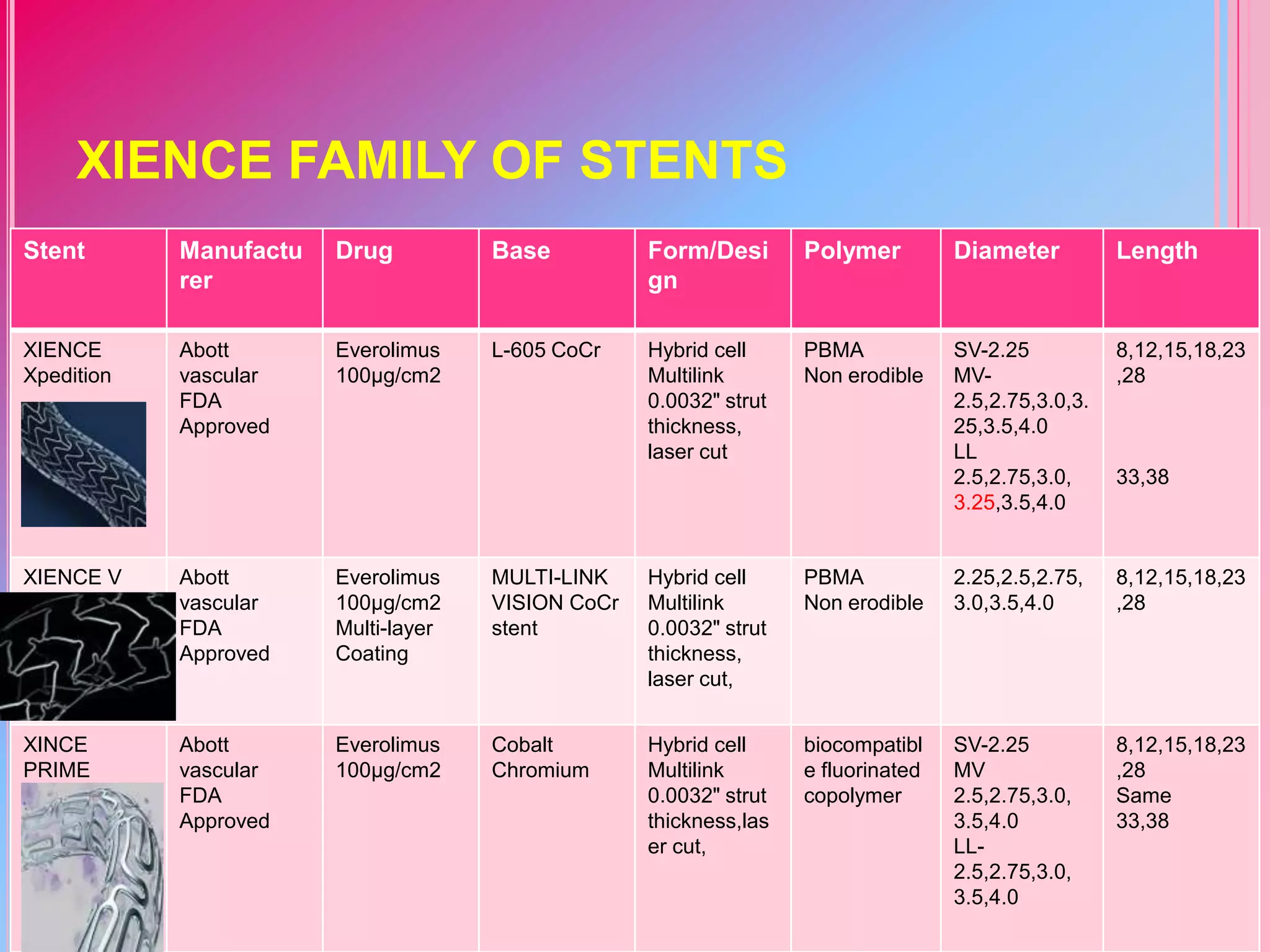


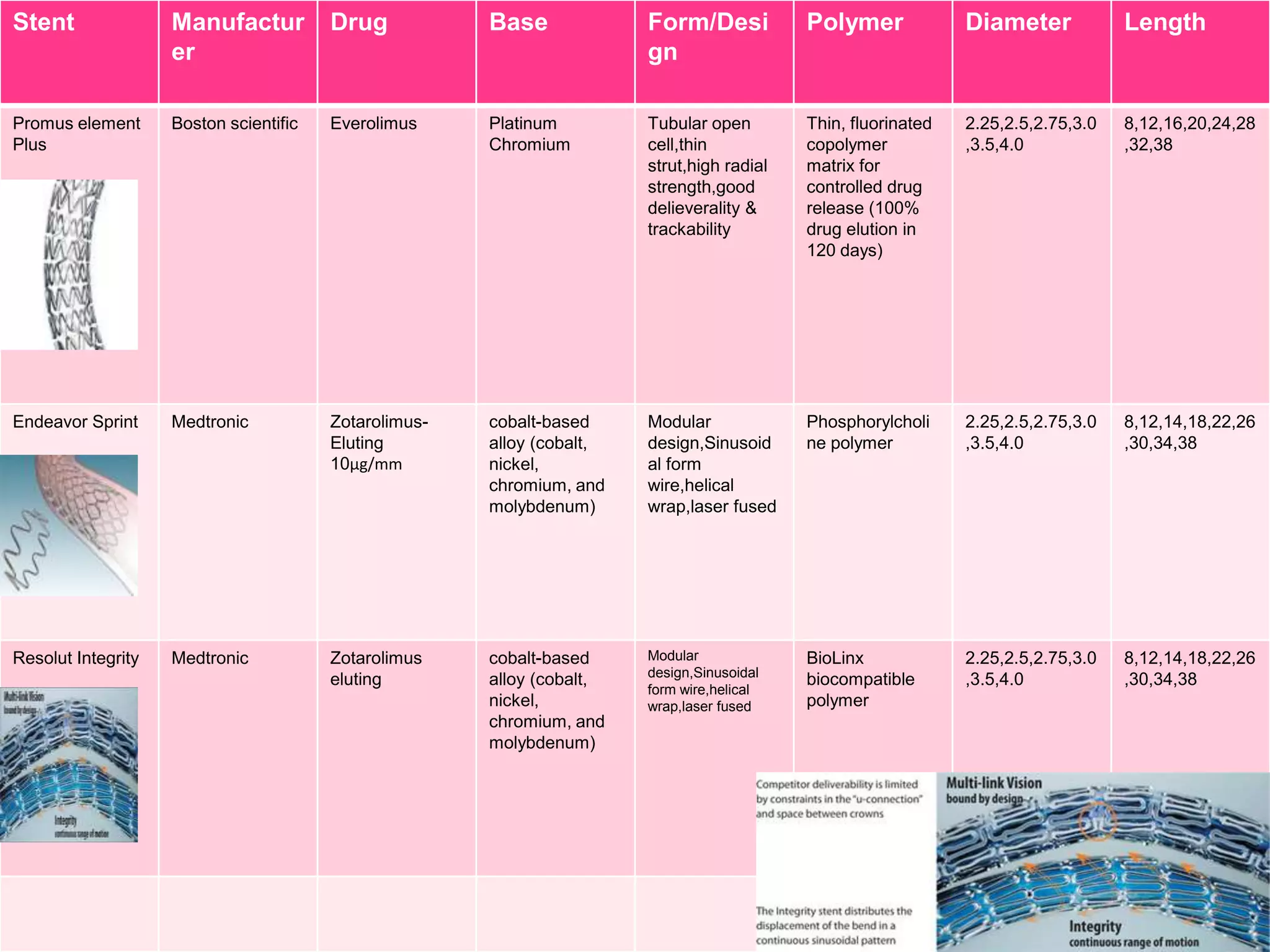
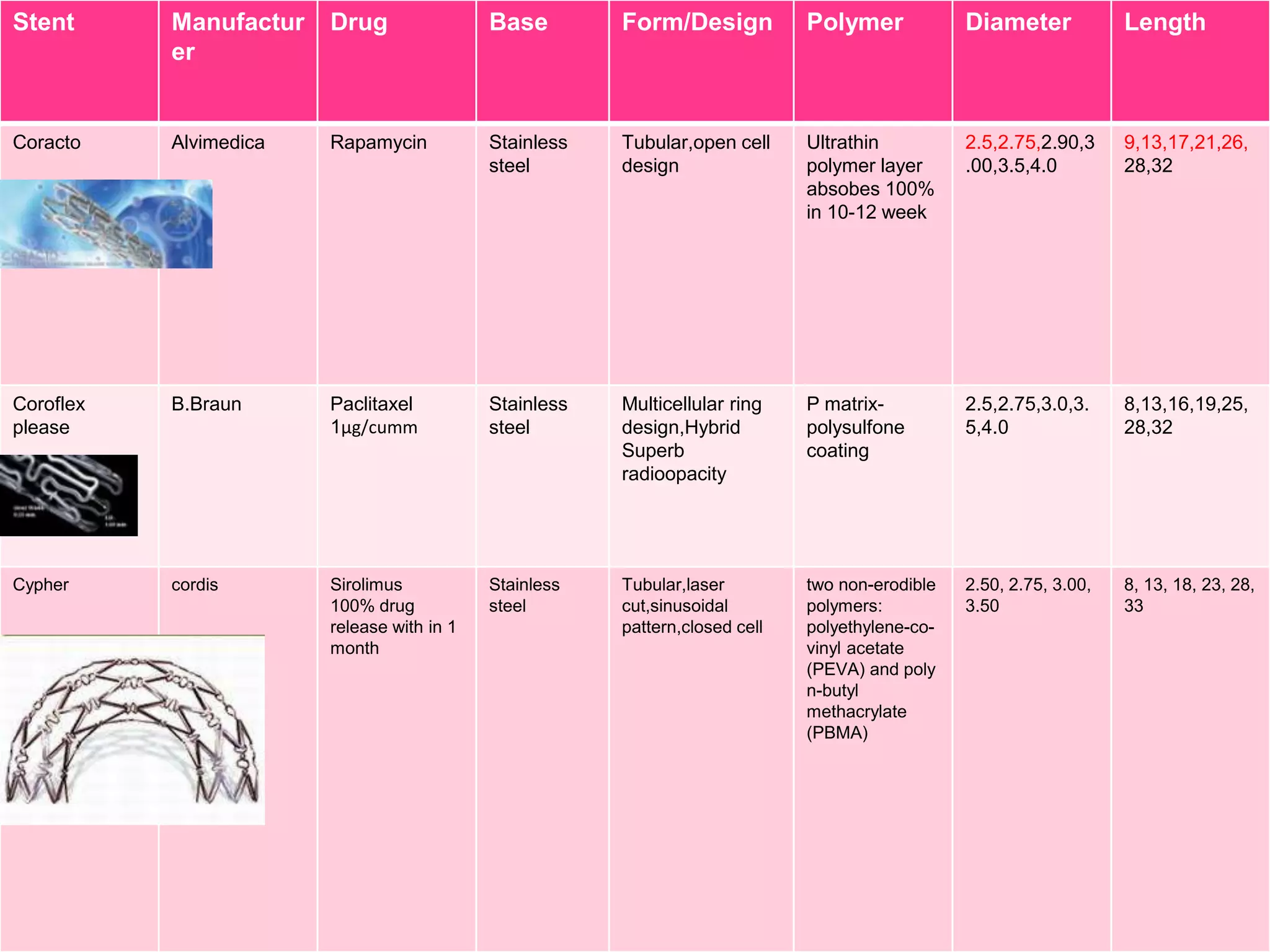
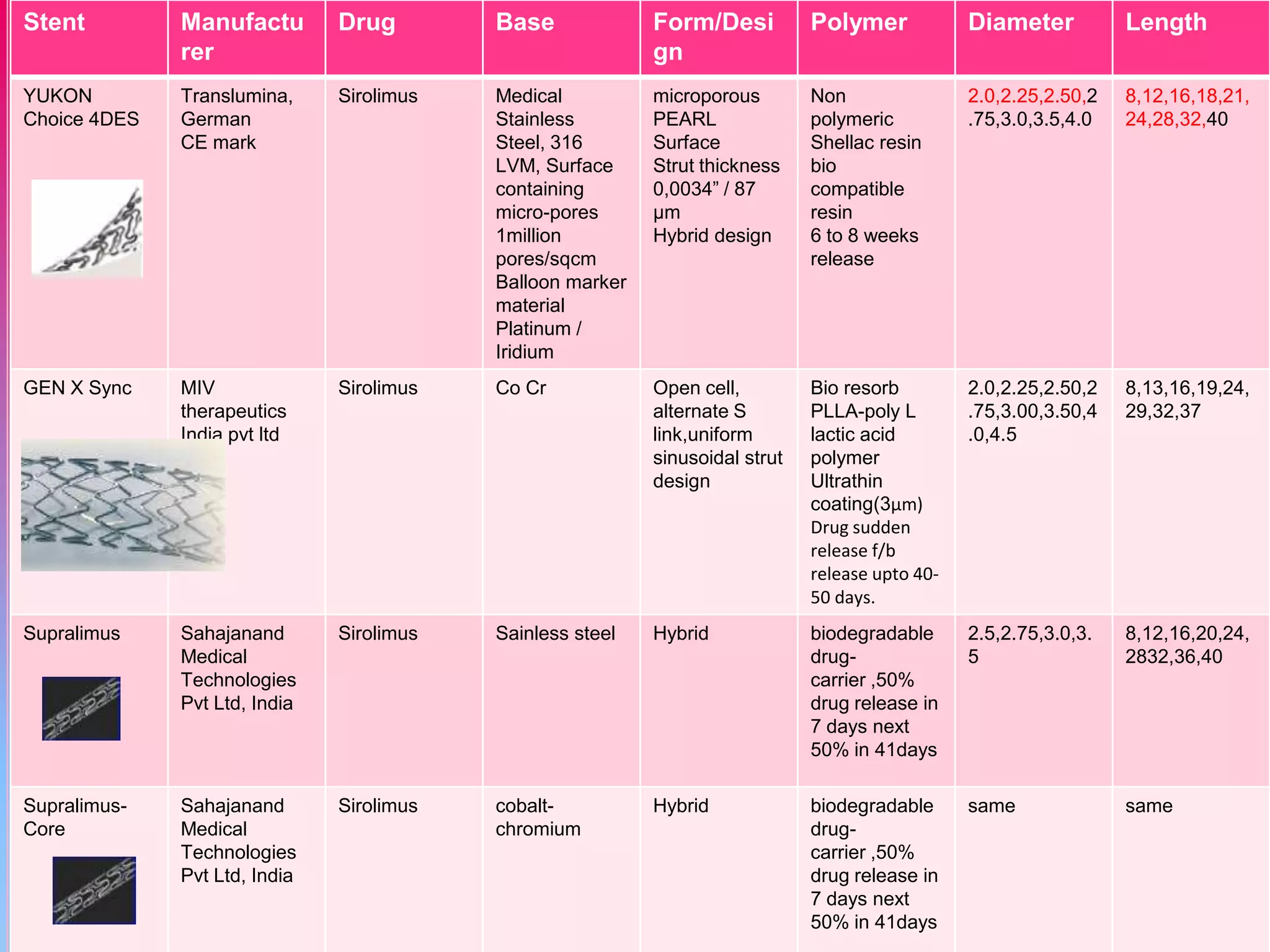
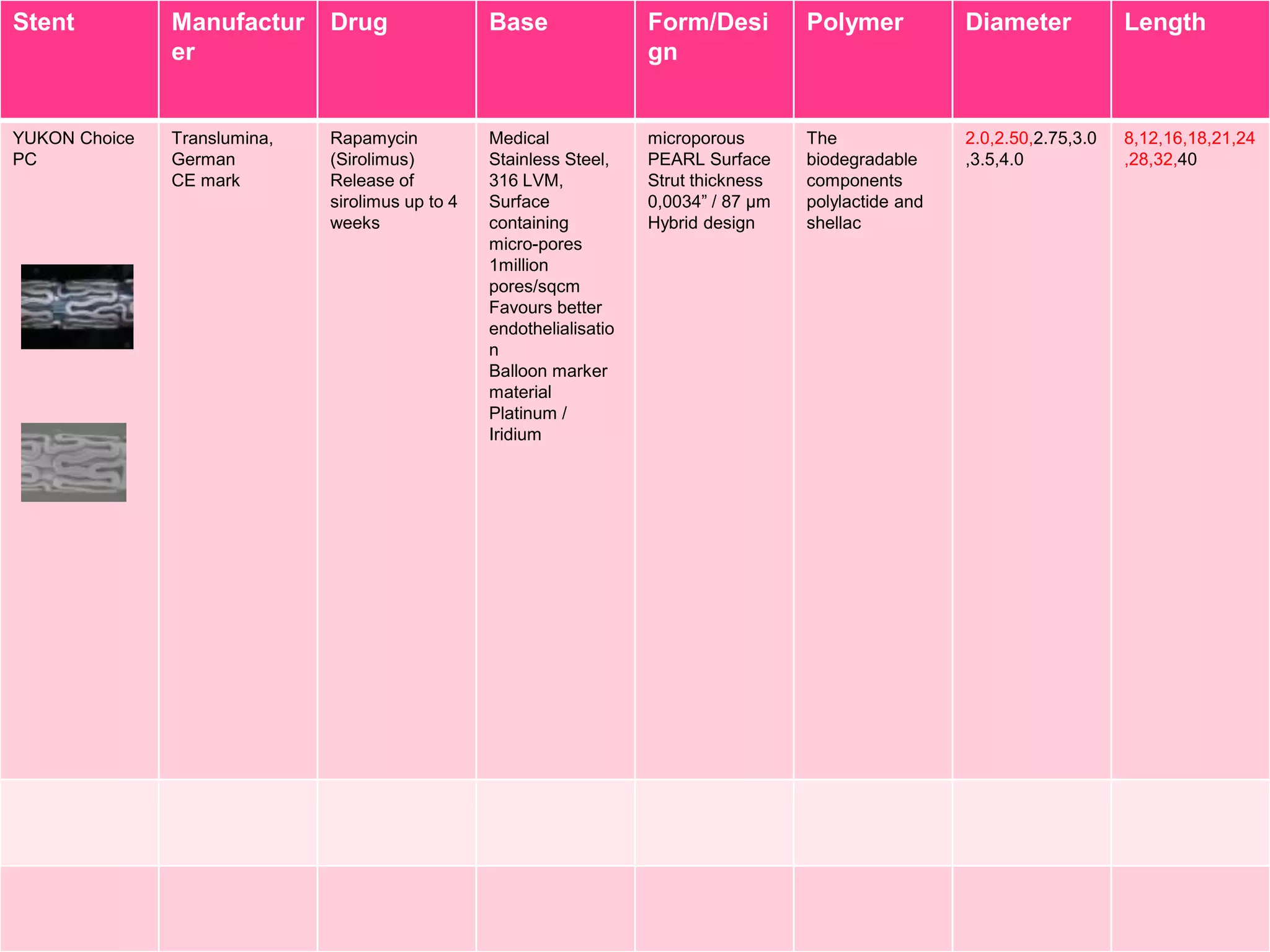
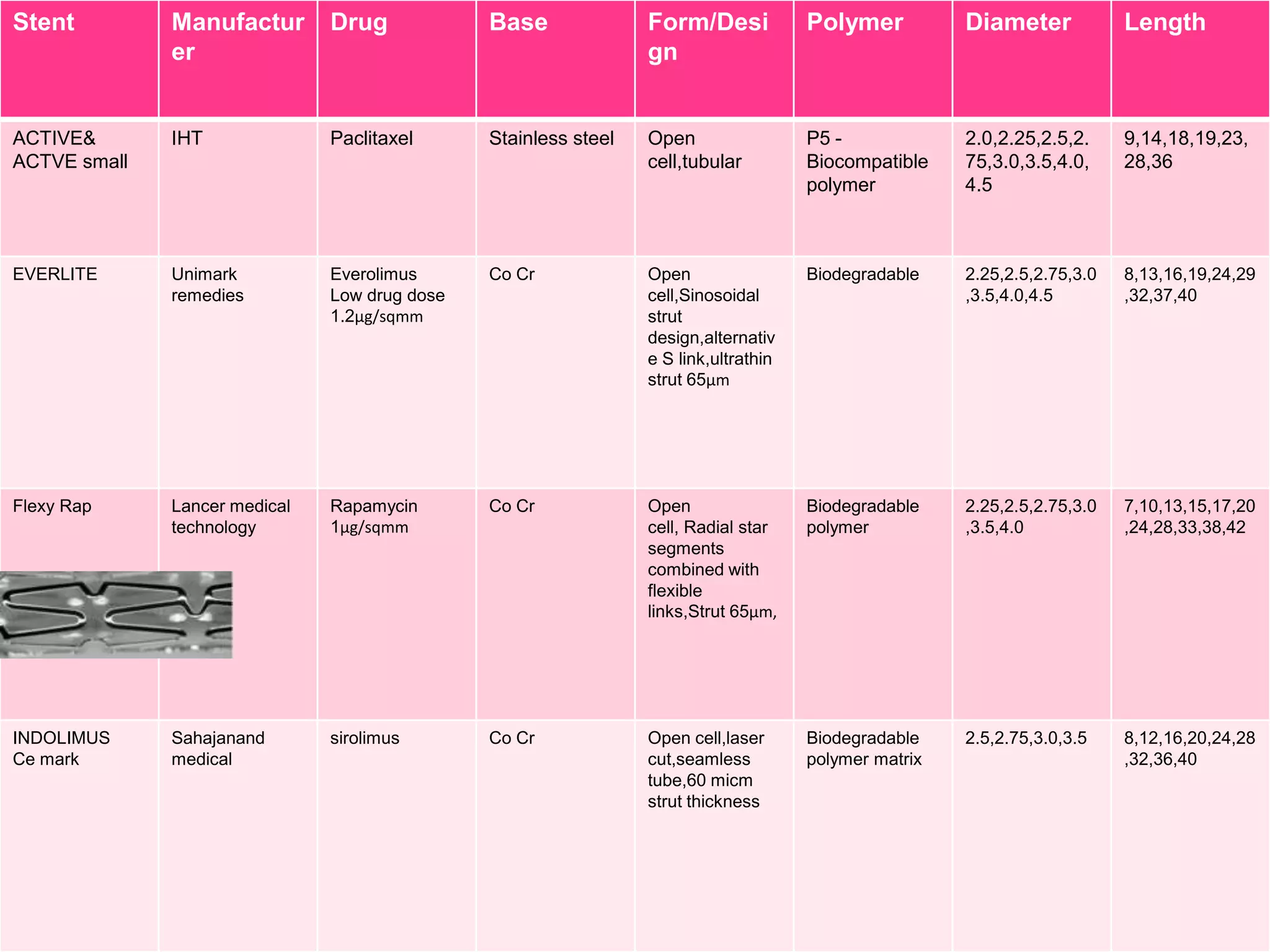
![Stent
Manufactur
er
Drug
Base
Form/Desi
gn
Polymer
Diameter
Length
Taxus Liberte
Boston Scientific
Paclitaxel
1 μg/mm2
paclitaxel in a
slow release
(SR)*
316L surgical
grade stainless
steel
Sinusoidal ring
modules linked
via curved link
elements
SIBS
[poly(styrene-bisobutylene-bstyrene)], a triblock copolymer
(trade name:
Translute)
2.50, 2.75, 3.00,
3.50, 4.00
8, 12, 16, 20, 24,
28, 32
TAXUS Express
Boston Scientific
Paclitaxel
1μg/mm2
paclitaxel in a
slow release
(SR)
316L surgical
grade stainless
steel
modular ring
strut pattern
consists of two
separate module
designs: short,
narrow
sinusoidal Micro
elements linked
via straight
articulations to
long, wide
sinusoidal Macro
elements
SIBS
[poly(styrene-bisobutylene-bstyrene)], a triblock copolymer
(trade name:
Translute)
2.50, 2.75, 3.00,
3.50
8, 12, 16, 20, 24,
28, 32
Taxus Element
Boston Scientific
Paclitaxel
1.0 μg/mm2
Platinum
Chromium
Sinusoidal ring
modules
consisting of
alternating long
and short
SIBS
[poly(styrene-bisobutylene-bstyrene)], a triblock copolymer
2.25,2.50,2.75,3.
0,3.5,4.0,4.5
8,12,16,20,24,28
,32,38](https://image.slidesharecdn.com/coronarystentdesignppt-131213121014-phpapp02/75/Different-Coronary-stent-design-PPT-71-2048.jpg)