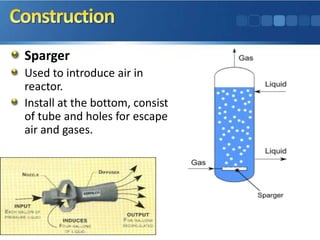
This document provides an overview of slurry reactors, including their types, construction, operation, start up and shut down procedures, troubleshooting, advantages and disadvantages, applications, and an incident involving a slurry reactor explosion. Slurry reactors can react solids, liquids, and gases simultaneously using a solid suspended in a liquid with gas bubbled through. Common types include bubble column reactors and Fischer-Tropsch reactors. The document describes the typical components of a slurry reactor and procedures for operation and maintenance. Potential problems that can occur and their solutions are also outlined. Applications include methanol production and waste water treatment.