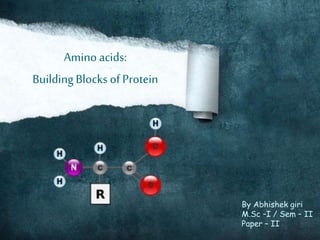
Amino acids- the building blocks of proteins
- 1. Amino acids: BuildingBlocks of Protein By Abhishek giri M.Sc –I / Sem – II Paper – II
- 2. Points to consider 1. Structure 2.Classification 3.Properties 4.D-amino acids
- 3. Amino acids are building blocks of proteins. Proteins are composed of 20 different amino acid (encoded by standard genetic code, construct proteins in all species ). Their molecules containing both amino and carboxyl groups attached to the same a-carbon (L-a-amino acids). Their chemical structure influences three dimensional structure of proteins. They are important intermediates in metabolism They can have hormonal and catalytic function. Several genetic disorders are cause in amino acid metabolism errors What are Amino acids?
- 4. Louis-Nicolas VauquelinPierre Jean Robiquet History of Amino acids Isolated Asparagine in 1806
- 5. Basic structure The basic structure of amino acids differ only in the structure of the or the side chain (R-group). L-isomer is normally found in proteins. L-isomer
- 6. Classification Amino acids are classified into different ways based on : A. Structure B. Polarity C. Nutritional requirement D. Metabolic fate
- 7. A. Structure Based on Structure amino acids they are divided into 7 distinct group 1. Amino acid with Aliphatic side chain: 2. Amino acid containing Hydroxyl group(-OH): 3. Amino acid containing Sulpur: 4. Acidic amino acid : 5. Basic amino acids: 6. Aromatic amino acids: 7. Imino acids:
- 8. 1.Amino acids withaliphaticsidechains: Alanine(Ala) Valine(Val) Leucine(Leu) Isoleucine(Ile) Glycine (Gly) Branched Chain Amino acids
- 9. 2. (–OH)amino acids: 3. Sulfur amino acids: Serine (Ser) Threonine (Thr) Tyrosine (Tyr) Methionine Cysteine (cys) Cystine
- 10. 4.Acidic aminoacid Asparagine (Asn) Glutamine (Gln)Glutamic acid (Glu) Aspartic acid (Asp) Lysine (Lys) Arginine (Arg) Histidine (His) 5.Basic amino acid Dicarboxylic monoamino acids
- 11. 6.Aromatic& Imino aminoacids Tyrosine (Tyr) Phenylalanine Tryptophan (Trp) Proline(Pro) (=NH) Imino group instead of Amino group (-NH2)
- 12. B. Polarity Based on Polarity amino acids they are divided into 4 distinct group 1. Non- polar Amino acids: 2. Polar Amino acid with no charge on ‘R’ group: 3. Polar Amino acid with positive ‘R’ group: 4. Polar Amino acid with negative ‘R’ group:
- 13. 1.Nonpolar(Hydrophobic)R Groups Proline (Pro) Tryptophan (Trp) Phenylalanine (Phe) Methionine (Met) Valine (Val) Alanine (Ala) Glycine (Gly) Leucine (Leu) Isoleucine (Ile)
- 14. 2.Polar (Hydrophilic)R Groups Serine (Ser) Cysteine (cys) Glutamine (Gln) Asparagine (Asn) Tyrosine (Tyr) Threonine (Thr)
- 15. 3.PositivelyCharged R Groups Lysine (Lys) Arginine (Arg) Histidine (His)
- 16. Aspartic acid (Asp) Glutamic acid (Glu) 4.Negatively(Nonpolar) Charged RGroups Two amino acids with negatively charged (i.e. acidic) side chains - Aspartate (Aspartic acid) and Glutamate (Glutamic acid). These amino acids confer a negative charge on the proteins of which they are part.
- 17. C. Nutritional Based on Nutritional requirement amino acids are divided into 2 distinct group 1. Essential or Indispensable Amino acids: 2. Non-essential or dispensable Amino acids:
- 18. Required in diet Humans incapable of forming requisite carbon skeleton Arginine* Histidine* Isoleucine Leucine Valine Lysine Methionine Threonine Phenylalanine Tryptophan * Essential in children, not in adults 1.Essential AminoAcids in Humans Semi-essential amino acids
- 20. PVT TIM HALL
- 21. Not required in diet Can be formed from a-keto acids by transamination and subsequent reactions Alanine Asparagine Aspartate Glutamate Glutamine Glycine Proline Serine Cysteine Tyrosine 2.Non-EssentialAmino Acids in Humans
- 22. D. Metabolic fate Based on C-Skeleton of amino acids serve as a precursor for the synthesis of Glucose or Fat or both. Form metabolic point of view they are divided into 3 groups 1. Glycogenic Amino acids: 2. Ketogenic Amino acids: 3. Glycogenic & Ketogenic amino acids:
- 23. 1. Glycogenic amino acids Alanine (Ala) Glycine (Gly) Methionine (Met) Aspartic acid (Asp)
- 24. 2. Ketogenic aminoacids Leucine (Leu) Lysine (Lys)
- 25. 3. Glycogenic & ketogenic Amino acids Tyrosine (Tyr) Isoleucine(Ile) Phenylalanine Tryptophan (Trp)
- 26. Properties ofAminoacids Amino acids differ in their physico-chemical properties which ultimately determine the characteristics of proteins. A. Physical properties B. Chemical properties
- 27. A. physical PROPERTIES Solubility Soluble in water but Insoluble in organic solvents. Melting point Melts at Higher Temperature. Taste Sweet (Gly, Ala, Val), Tasteless (Leu), Bitter (Arg, Ile), MSG. Optical properties Optical isomers due to Asymmetric C-atom. Amino acids as Ampholytes They can donate or accept a proton.
- 28. Nonionic and zwitterion forms of amino acids The zwitterion predominates at neutral pH Weak acid Weak base Zwitterion = in German for “hybrid ion”
- 30. Isoelectric pH Defined as pH at which a molecule exists as a Zwitterion & carries no net charge. Thus, the molecule is electrically neutral. pH = pK1 (COO⁻) + pK2 (NH3⁺) 2 pl = 2.34 + 9.60 = 5.97 2
- 31. Titrationof Amino Acids Dipolar ionIzoelectric point At the midpoint – pK=9.60. there is equimolar concentration of proton donor and proton acceptor At the midpoint – pK1=2.34 there is equimolar concentration of proton donor and proton acceptor.
- 32. B. Chemical Reaction is mostly due to Functional groups namely Carboxyl (-COOH) group & amino (-NH2) group. REACTION DUE TO (-COOH) GROUP : REACTION DUE TO (-NH2) GROUP :
- 33. 1.Amino acids forms Salts (-COONa) with bases & esters (-COOR’) with alcohols. 2. DECARBOXYLATON : amino acids undergo decarboxylation to produce corresponding amines. 3. REACTION WITH AMMONIA : + NH3 + NH3 Aspartic acid (Asp) Glutamic acid (Glu) Asparagine (Asn) Glutamine (Gln)
- 34. 4. The amino acids behaves as bases & combine with acids (e.g HCL) to form salts (-NH3⁺Cl⁻) 5. Colour reactions of Amino acids : S.No. Test Significance 1) Ninhydrin reaction Given by all Alpha amino acids 2) Xanthoproteic test Given by aromatic amino acids 3) Millon’s test Confirmatory test for Tyrosine 4) Ehrlich’s reagent Confirmatory test for Tryptophan 5) Nitric acid test Given by throsine & tryptophan
- 35. 6.Ninhydrin Reaction This strong oxidizing agent bring about the oxidative decarboxylation of amino acid. Purple, Blue or Pink colour complex is formed (Ruhemann’s purple) Quantitative estimation of amino acids & proteins
- 36. 7.Oxidative deamination : The amino acids undergo oxidative deamination to liberate free ammonia. 8.Transamination : Transfer of an α-amino group from an amino acid to an α-keto acid to form a new amino acid and a corresponding keto acid.
- 37. Uncommon Amino Acids Cell wall (plant) Collagen (connective tissue) Collagen Myosin Prothrombin (blood clotting) Ca2+ binding proteins Elastin A few proteins Incorporation during translation
- 38. D-Aminoacids Major amino acids are L-category, but certain D- amino acids are also found in the antibiotics. Antibiotics like Actinomycin-D, Valinomycin, Gramicidin-S. D-serine D-aspartate D-Glutamic acid D-alanine Brain Tissue Bacteria cell wall
- 39. Reference Biochemistry – U. Satyanarayana Advanced organic chemistry – Bahl n Bahl Principles of biochemistry – David L. Nelson A textbook on biochemistry – MN Chatterjee en.wikipedia.org/wiki/Amino_acid www.nlm.nih.gov/medlineplus/ency/article/002222.htm www.aminoacidsguide.com/ www.imagerynet.com/amino/classification.html
Editor's Notes
- To view this presentation, first, turn up your volume and second, launch the self-running slide show.
