Allylic strain
- 1. Organic Pedagogical Electronic Network Allylic Strain Created by Sophia Robinson Physical Organic Chemistry I CHEM 7240 (Sigman), 2015
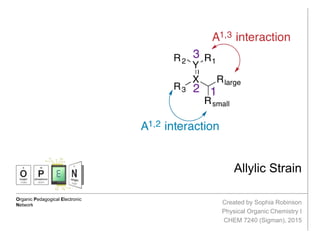
- 2. Allylic Strain http://isites.harvard.edu/fs/docs/icb.topic93502.files/Lectures_and_Handouts/05-Conformational_Anal-2.pdf Hoffman, R.W. Chem Rev. 1989, 89, 1841 Allylic strain arises from eclipsed conformations when Z allylic substituents and Z substituents at the 2- or 3-positions are large enough to create an unfavorable nonbonding repulsion. Strain between the allylic and 2-position substituents is called A1,2- strain (2.7 kcal/mol). Strain between the allylic and 3-position substituents is called A1,3-strain (3.9 kcal/mol). A1,2-strain and A1,3-strain affect the diastereoselectivity of reactions. The allylic position is the atom bound to a double bonded atom. The substituents on the allylic carbon and the doubly bonded atoms can result in allylic strain.
- 3. Allylic Strain http://isites.harvard.edu/fs/docs/icb.topic93502.files/Lectures_and_Handouts/05-Conformational_Anal-2.pdf Hoffman, R.W. Chem Rev. 1989, 89, 1841 Consider the general structure where X and Y are C, N, or O: The allylic center and its substituents often lead to a significant steric bias toward reactions occurring at the double bond.
- 4. Allylic Strain Hoffman, R.W. Chem Rev. 1989, 89, 1841 Consider rotation about the bond of the two substituted allylic systems shown: These values are from ab intio calculations performed by Houk. A and B are both minima in conformational energy while C is not an energy minimum. When the C(3) substituent is a methyl rather than hydrogen, conformational equilibrium strongly favors D. With the methyl present, E is destabilized by allylic 1,3- strain and is an energy maximum. F is an energy minimum but is far less energetically favorable than D.
- 5. Examples of Reactions influenced by Allylic Strain Diels-Alder Hoffman, R.W. Chem Rev. 1989, 89, 1841 The diastereoselectivity of a Diels-Alder reaction is increased by presence of an additional substituent on carbon 2 as the substituent destabilizes transition state B through A1,3-strain. Two transition states for the Diels-Alder reaction are shown in which opposite faces of the diene are shielded:
- 6. Examples of Reactions influenced by Allylic Strain Hydroboration Oxidation The alkene aligns itself as shown such that the smallest of the substituents is staggered with the alkene double bond to prevent A1,3-strain. The borane then approaches from the side of the medium-sized methyl substituent rather than the larger R1 substituent: Houk, K.N.; Rondan, N.G.; Wu, Y.D.; Metz, JT; Paddon-Row, M.N.; Tetrahedron, 1984, 40, 2257
- 7. Problems Hoffman, R.W. Chem Rev. 1989, 89, 1841 Fleming, I and coworkers, Chem. Commun. 1985, 318;
- 8. Solutions 1.) R = Me, A is major diastereomer (87 A : 13 B) R = Et, A is major diastereomer (80 A : 20 B) R = CHMe2, B is major diastereomer (40 A : 60 B) 2.) B. B is the most stable, C is the least stable 3.) B (> 95% ds) (A ds = 50 %) 4.) B. B is major diastereomer. If SiMe3 substituent is not present, no diastereoselectivity is observed 5.) C (ds = 90%)
- 9. This work is licensed under a Creative Commons Attribution- ShareAlike 4.0 International License. Contributed by: Sophia Robinson, (Undergraduate) Physical Organic Chemistry I CHEM 7240 (Sigman), University of Utah, 2015