Organic Pedagogical Electronic Network Inductive Effects
- 1. Organic Pedagogical Electronic Network Inductive Effects Colin Swenson Tewoderos Ayele University of Utah
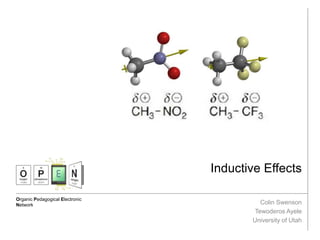
- 2. Inductive Effect Eric Anslyn and Dennis Dougherty, Modern Physical Organic Chemistry, University Science Books; pp 15-18 Overview: An inductive effect occurs when the electrons shared between two atoms or groups of different electronegativities are withdrawn to the more electronegative atom or group through the σ bond resulting in a separation of charge, or dipole. This effect can be felt a few atoms away affecting the reactivity of other groups on the molecule. The yellow arrows show the dipole moment of the molecule, indicating the direction of electron density.
- 3. Inductive Effects on Acidity Reush, W. Carboxylic Acids. https://www2.chemistry.msu.edu/faculty/reusch/VirtTxtJml/crbacid1.htm (Accessed October 2015). The increased acidity of carboxylic acids can be attributed to electrostatic effects, such as induction. More electronegative groups will greatly change the distribution of electron density in the molecule by withdrawing electrons. This effect will make the proton more acidic. The relative distance of the electronegative group also affects the acidity of carboxylic acids. The closer the electron withdrawing substituent is to the reactive proton, the more acidic the molecule becomes. Acidity increases as more electronegative substituents are added to the molecule and withdraw electron density from the acidic proton Compound pKa CH3CO2H 4.74 BrCH2CO2H 2.9 ClCH2CO2H 2.85 FCH2CO2H 2.65 CH3CH2ClCO2H 2.89 CH3ClCH2CO2H 4.05 ClCH2CH2CO2H 4.53
- 4. Case Study Bell, R., Mellot-Draznieks, C., Torrisi, A. J. Chem. Phys. 130, 194703 (2009) Inductive Effects on Aromatics for CO2 Adsorption Carbon dioxide, a side product in the production of hydrogen gas for fuel applications, needs to be separated from the gaseous mixture at very low pressures. An economic approach involves the use of Metal-Organic Framework materials. The quadrupole of CO2 interacts with the delocalized pi- system of aromatics. Substituents supplying inductive effects have been studied for enhancement of the affinity for CO2. Methyl substituents supplying a positive inductive effect are shown to stabilize the CO2-aromatic interaction, whereas halogen substituents supplying a negative inductive effect are shown to destabilize the CO2-aromatic interaction. Delocalized charge on aromatic carbons as a function of the binding energy (BE) to carbon dioxide of different substituent groups.
- 5. Problems Bell, R., Mellot-Draznieks, C., Torrisi, A. J. Chem. Phys. 130, 194703 (2009) 1. Rank the following in increasing acidity (1 is most acidic) a. 1 b. 1 c. 1 2. Explain how induction effects differ from resonance effects. 3. Provide an explanation for the trend seen in the delocalization of charge on the aromatic carbons presented in the case study. 4. Which molecule would you expect to have a greater affinity for CO2 , C6H5NO2 or C6H5NH2 ? Explain.
- 6. Solutions Bell, R., Mellot-Draznieks, C., Torrisi, A. J. Chem. Phys. 130, 194703 (2009) 1 a. b. c. 2. Resonance effects are directly involved with the movement of electrons, whereas inductive effects are a result of a change in electron density. 3. There is less delocalized charge on the aromatic carbons as halogens are placed on the ring because they withdraw electron density from the aromatic system. 4. C6H5NH2 because there will be more delocalized charge on the aromatic system due to the amine group being less withdrawing than the nitro group. 3 2 1 2 3 1 3 1 2
- 7. This work is licensed under a Creative Commons Attribution- ShareAlike 4.0 International License. Contributed by: Colin Swenson and Tewoderos Ayele (Undergraduates) University of Utah, 2015