Promiscuous enzyme
- 1. Promiscuous Enzymes Presented by- Vikrant Rana
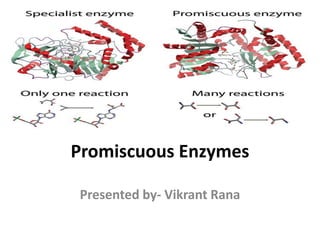
- 2. What is Enzyme Promiscuity? “The term ‘promiscuous’ is used to describe enzymes that catalyze more than one reaction” “Enzyme promiscuity is an ability to catalyze secondary reactions that are physiologically irrelevant”
- 3. • Enzyme promiscuity is the ability of an enzymes to catalyse a fortuitous side reaction in addition to its main reaction. Although enzymes are remarkably specific catalysts, they can often perform side reactions in addition to their main, native catalytic activity. • These promiscuous activities are usually slow relative to the main activity and are under neutral selection. Despite ordinarily being physiologically irrelevant, under new selective pressures these activities may confer a fitness benefit therefore prompting the evolution of the formerly promiscuous activity to become the new main activity.
- 4. Physiological function: Lactonase: Hydrolysis of oxidized lipids Secondary Function: OP hydrolase Physiological function: Lactonase: Vitamin C biosynthesis Secondary Function: OP hydrolase
- 7. • The biocatalysts normally do not require high temperature or other conditions which involve high consumption of energy. The biocatalysts are believed to be fairly specific, which would mean less number of side reactions. Side reactions lead to side products which lower the atom economy of the reactions. These side reactions hence lower the yield of the desired product (making the catalysis less efficient) and necessitate complicated down. • The results of these studies show that enzymes often do not accept just a single substrate, but instead accept a range of substrates Mechanism:
- 8. • In a growing number of enzymes a feature termed catalytic promiscuity has been identified, which can be defined as the capability of an enzyme to catalyze a chemically, and often mechanistically, distinct transformation in addition to its biologically relevant one. • Already in the 1970s, it was postulated that if a catalytically promiscuous activity could provide a selective advantage for the host organism, such a promiscuous activity could be the starting point for the evolution of a new enzyme.
- 9. • A low-level promiscuous activity can be amplified by accumulation of mutations, preceded or followed by gene duplication. The promiscuous activity of the parent enzyme then becomes the primary activity of the newly evolved enzyme. This concept is nowadays widely accepted and worked out in more detail by a number of key studies on enzyme promiscuity. • Second, the characterization of catalytically promiscuous activities of enzymes can aid in the identification of active site residues as catalytically important. Moreover, insight can be gained into the mechanistic roles of active site residues. Often it is found that the same set of catalytic residues is involved in both the enzyme’s native and promiscuous activity, but that these residues fulfill a different mechanistic role.
- 10. • Third, the presence of catalytic promiscuity shows the chemical versatility of an enzyme’s active site. In fact, catalytic promiscuity might even be predicted based on knowledge of the chemical and mechanistic properties of active site residues. Here lie formidable challenges and possibilities: the use of mechanistic reasoning to discover new promiscuous activities in existing enzymes, which could be exploited to generate novel biocatalysts.
- 12. Degree of promiscuity • Enzymes are generally in a state that is not only a compromise between stability and catalytic efficiency, but also for specificity and evolvability, the latter two dictating whether an enzyme is a generalist (highly evolvable due to large promiscuity, but low main activity) or a specialist (high main activity, poorly evolvable due to low promiscuity). Examples of these are enzymes for primary and secondary metabolism in plants.
- 13. Types of enzyme promiscuity:
- 14. • Enzyme condition promiscuity: Shown by enzymes with catalytic activity in various reaction conditions different from their natural ones, such as anhydrous media, extreme temperature and pH. • Enzyme substrate promiscuity: Shown by enzymes with relaxed or broad substrate specificity. • Enzyme catalytic promiscuity: Shown by enzymes catalysing distinctly different chemical transformations with different transition states. It can be 1. Accidental- a side reaction catalysed by wild-type enzyme 2. Induced- a new reaction established by one or several mutations
- 16. Application in pharmaceutical industry • Dr. Reddy’s Laboratory in production of mintop 10 solution which is a bioactive serum. • In production of Biocon’s immunosuppressant and statins • Insulins and analogs • In food products, used in USA by Dr. Trust in production of digestive enzyme. • Also used in production of some flavour compounds.
- 17. Conclusion • Enzyme promiscuity is a property shown by many enzymes, nowadays exploited in applied enzymology .It is classified in to conditional , substrate and catalytic which results into many industrial applications. • Over last few years, it have been observed that other classes of enzymes(hydrolases, oxidoreductases, transferases etc. are being capable of showing catalytic promiscuity.
- 18. References: • Gupta MN, Raghava S: Relevance of chemistry to white biotechnology. Chem Cent J 2007, 1:17 • Ulber R, Sell D: White Biotechnology. Berlin: Springer-Verlag; 2007 • Kazlauskas RJ: Enhancing catalytic promiscuity for biocatalysis. Curr Opin Chem Biol 2005, 9:195–201 • Khersonsky O, Tawfik DS: Enzyme promiscuity: a mechanistic and evolutionary perspective. Annu Rev Biochem 2010, 79:471–505
Editor's Notes
- either because they are too inefficient to affect fitness or because the enzyme never encounters the substrate
