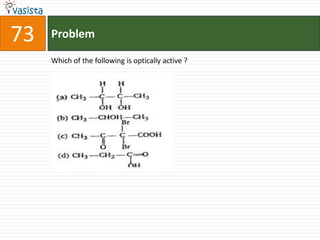
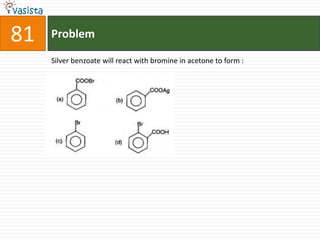
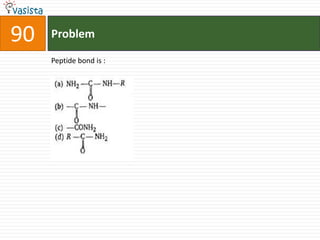
The document contains 59 multiple choice questions from a chemistry exam. The questions cover topics such as:
1) Identifying elements and compounds based on their electronic configurations and quantum numbers.
2) Calculating quantities like moles, molarity, and vapor pressure from chemical equations and experimental data.
3) Understanding concepts like ionization energy, electronegativity, periodic trends, chemical bonding, and acid-base chemistry.




























![27 Problem
Which of the following electrolytes is least effective in coagulating ferric
hydroxide solution ?
a. KBr
b. K2SO4
c. K2CrO4
d. K4[Fe(CN)6]](https://image.slidesharecdn.com/1999-111214014733-phpapp01/85/UPSEE-Chemistry-1999-Unsolved-Paper-29-320.jpg)